Question 33: What is the philosophy or criteria for optimizing catalyst bed grading material to prevent high reactor pressure drop from feed containing significant amounts of Fe (iron)?
LIOLIOS (DuPont Clean Technologies)
Certainly, identifying the sources of the iron coming in – whether organic, iron oxides, iron sulfides, or just scale from tanks – is very critical to understanding your best strategy for mitigating pressure drop. Ultimately, when you form iron sulfide, it creates deposits on the bed and coke deposition, and certainly leads to reduced catalyst life.

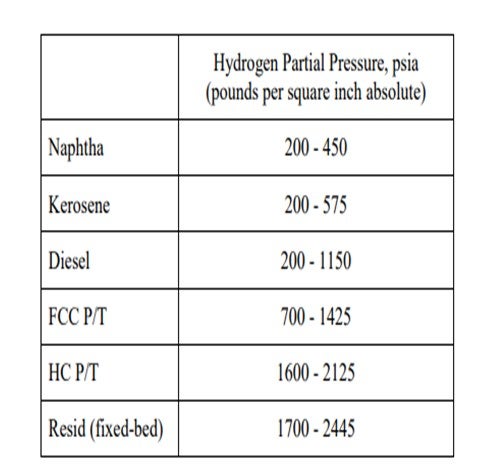
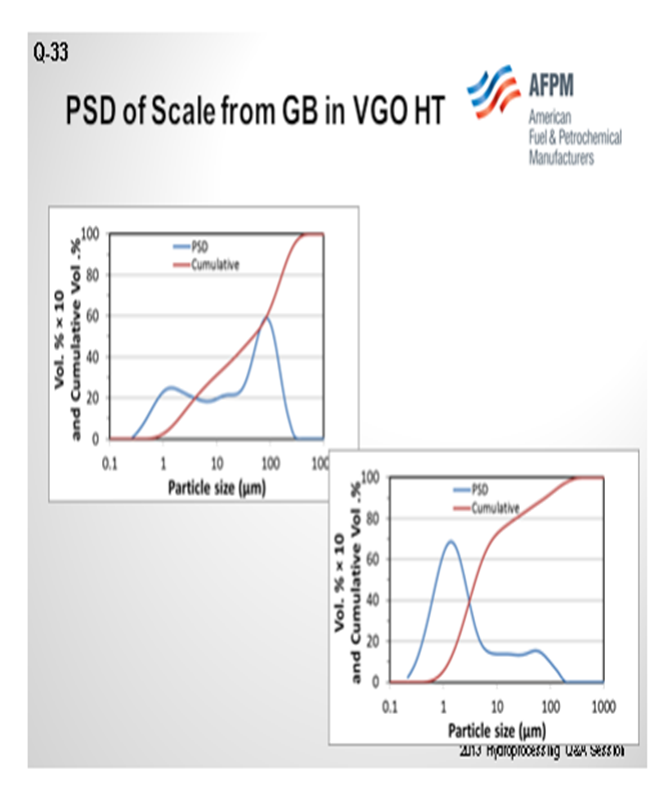
The next slide shows the analysis of the scale that was collected from a guard bed in a VGO hydrotreater. The graph on the left is the first pass on the analyzed scale. About 40% of it was under 25 micron. The analysts took that same sample and re-circulated it. I cannot get into it, but basically 80% of the sample after recirculation was under 25 microns. Also, the small particles will come into your bed under hydrotreating conditions. They will grow and increase in pressure drop, and they will take off on you. Understanding why iron sulfide is coming in and how it is coming in is very important to setting up a strategy, which is shown on the next slide. So, identifying the root cause is important.
The first phase of the reduction strategy should be feed filtration. Either cartridge or backwash-type filters are most commonly used down to about a 25-micron size. Next, look at size grading, which is a high-void area, in order to capture this material in the void area. Crystaphase CatTrap® or other specialized topping materials are very effective at capturing some of these iron materials then some active grading because some of the iron content will actually form iron sulfide at these conditions. So, after the active grading, you will need additional trapping material with large pore structures in order to capture the iron.

WATKINS [Advanced Refining Technologies (ART)]
I basically agree with a lot of what Glenn just said. The goal is to identify the origin of your iron and determine if there is a way to handle or stop it. If it is corrosion, we can certainly take care of it elsewhere. Characterization is the key. The bottom three pictures on the slide simply show iron and how it is sitting in the reactor or on various places in exchangers or furnaces or on the catalyst. Some can be filtered out; for others, you must use a catalytic mechanism. The one on the left is absorbed iron on catalyst.

Again, 25 microns is the target range for iron-particulate scale and other contaminates that are going to come into the hydrotreater using a grated bed system to mitigate any pressure drop. The goal is to dramatically increase the actual void fraction so you have somewhere to put this incoming iron. Top-bed materials with high internal void space are important. Sock loading: If you know that a prior cycle had a problem, the goal next time may be to change the diameter of your catalysts. Sock-load a portion of the bed so that you have some space, and do not shut down on pressure drop. If you have enough void space in your reactor, the contaminates can sometimes actually pass through. This will not really be a problem in your catalyst beds if you have too much void space.

The third slide shows an example you may have already seen. On the top left is a Crystaphase disk, which we can see has a very large void space. You can actually fill up quite a bit of that with iron and other particulates. A similar example is using a ring molecule at the bottom comparing fresh and spent samples so you can pick up coke, iron, and other contaminates. So, it is all about creating a void space.

MUKESH PATEL (Reliance Industries Ltd.)
What is understood from some literature is that soluble and insoluble Fe, of course by filter and cat (catalytic) trap, can capture the particles and also that some of the iron is emitted by heightened crude processing. Is there any temperature correlation for Fe falling?
SALVATORE TORRISI, JR. (Criterion Catalysts & Technologies)
I think the answer is: Yes, there is a temperature correlation. In general, when you have organic iron, as soon as it sees an H2S environment, it will start to recreate the iron sulfide. Typically, many of the hydrotreaters do not have H2S coming in until you first begin to desulfurize. It is very important to activity-grade to generate H2S in very small quantities as a start so that you do not deposit all of your iron at once. You tend to want to go through a very long activity grading, as well as a size grading on top of the activity, to try to disperse and deposit that iron in a large amount of pore volume. Of course, there is a temperature component to that, but perhaps the more important component is controlling the activity during that deposition.
I have a follow-up from the first question about arsenic. Mukesh, I think you asked that as well. I received a question for the panel after that about whether it is possible to put in a scavenger. You can soak up arsenic, which is the scavenger. That, of course, takes up space, but maybe not necessarily activity. Over the years, we have begun to put include multiple functionalities. You are not only picking up arsenic, but you also lose activity with arsenic because it will kill your ∆T. If you have a unit that is ∆T-limited, particularly at end-of-run, you become furnace-limited. I think the industry has begun to develop more catalysts, as opposed to traps, in an effort to accommodate the arsenic and compress it into as small a space as possible. That is a late response to Question 1.
Back to the panelists: More along the lines of Questions 1, 2, and 3. When you have multiple contaminants together, what is your philosophy on how to handle it? I do not think you have nickel, arsenic, or iron just by themselves. Does anyone want to comment on handling trapping systems? It seems to be a relevant enough topic to have made the top three questions about when you get into multiple contaminants.
WATKINS [Advanced Refining Technologies (ART)]
Generally, it will be dependent on the type of service: a gas oil hydrotreater, FCC pre-treat, or hydrocracker pre-treat, for example. Your major contaminants are nickel plus vanadium, so you will start putting more demet-related products at the top of your reactor. The convenience of using an HDM guard catalyst is that it generally contains nickel, so you do get that combined benefit of picking up arsenic plus a little nickel and vanadium (Ni+V). If your process has more arsenic than Ni+V, you will generally work on your larger molecules first because you need space to put contaminates. If you put in guard material that is designed for arsenic and naphtha up at the top, it will get plugged-up rather quickly with nickel, vanadium, and contaminates like that.
In diesel hydrotreating, the use of guard material really depends on the concentration of your limiting factor. Most of the time, it will be silica, arsenic, or maybe phosphorus. We often have a combined trap, which really is defined by your concentration. We are not worrying about nickel or vanadium in diesel and naphtha products.
GLENN LIOLIOS (DuPont Clean Technologies)
Literature shows that a mixed iron sulfide and coke deposits are common in higher temperature equipment which processes sour crudes and its processed components1. Hence, more information may be obtained from the 2011 study by Wang and Watkinson who determined that iron sulfide and coke formation were the two main culprits of fouling in HP catalyst beds. Catalyst bed grading targets the capture of particles, iron scale, and other contaminants that may cause fouling. Use of proper guard catalysts that can capture iron should be part of the solution supplementing catalyst bed grading. A more detailed explanation may be found below.
Preventing reactor pressure drop with feeds containing significant amounts of Fe typically involves a multi-faceted strategy including feed filters, bed grading, and the use of iron traps. Depending on the form of the iron (organic, iron oxide, or iron sulfide, etc.), it may also include a strategy for converting the iron to the form most readily trapped. Iron present as iron sulfide (FeS) in the feedstock, due to corrosion in upstream units, may be too small to be trapped in feed filters. To this end, guard bed material with ultra large pore structure iron traps can be used to trap iron sulfide inside the guard element. If the iron is particulate in nature, the particle size distribution will also impact the strategy.
Postmortem analysis of a hydrotreater scale includes a particle size distribution (PSD) analysis as one of the multiple tools used to troubleshoot iron contamination. In the diagram shown in Figure 1, at least 30% of the scale was FeS in nature from chemical analyses. The initial PSD plot shows multimodal particles centered at about 2 µm (micrograms), 20 µm, and 80 µm with bigger particulates being agglomerates of smaller ones. On this initial plot, one can also notice that particles under 25 µm account for 40% of the total. However, upon recirculation to allow some of the agglomerates to breakdown in primary particles, the most predominant particle size is around 2 µm with some particles up to 150 µm. In this case, particles under 25 µm account for more than 80% of the total particles. This is a very simplified analysis but shows the complexity of the problem to address not only the nature of the particles, but also their size distribution.
Feed filters, cartridge, or backwash type are usually used as initial mitigation for iron scale down to 25 µm particulates. Then size grading, along with activity grading, is used to mitigate iron source foulants depending on their type. It is also advisable that refiners identify the source of the iron and consider root cause analysis to potentially eliminate that source. For example, if the iron is a result of serious upstream corrosion, this may be managed through corrosion mitigation measures. However, if iron is naturally present in the feed (such as in naphthenic crudes), a combination of guard grading, shape, and activity is to be taken into consideration for mitigation of pressure drop strategy. In short, a combination of feed filters, graded size, and active material with iron traps can mitigate the onset of a rapid pressure drop increase; however, it should be tailored taking into account the nature and/origin of the iron contamination in the hydrotreater feedstock.
Finally, in situations when iron deposits have accumulated and resulted in significant pressure drop, the use of iron dispersants (such as provided by Baker Hughes) has resulted in varying degrees of success in partially mitigating the problem.
BRIAN WATKINS [Advanced Refining Technologies (ART)]
Iron works its way into hydrotreater feed as rust and iron scale from corrosion of upstream equipment and piping, as well as unfiltered particulates present in the feed. Iron naphthenates can form from piping corrosion due to naphthenic acid in the feed, and the iron readily precipitates out in the presence of heat and H2S. These iron particulates fill the interstitial spaces in the catalyst bed which will result in a higher than expected pressure drop. To help mitigate the pressure drop associated with iron, ART uses a series of grading materials which have high void space to accumulate and ‘store’ these particulates. Use of a specialized iron trapping material (GSK-9)
If iron is known to be the cause of the pressure drop issues, then changes to larger diameter catalysts can also be used in an effort to allow for additional void space in the reactor. Sock loading a large portion of the top of the reactor will also greatly increase the effective void space allowing the smaller iron particles to move through the reactor.
Another option is to use materials that are specially designed to dramatically increase the void fraction in the top bed of the reactor and which are very good at trapping iron, as well as using other particulates and scale. These measures are helpful for delaying pressure drop buildup, but they do not prevent eventual pressure drop buildup. Effective feed filtration to remove particulates (at least 25 microns) provides a longer lasting solution in helping mitigate pressure drop buildup from these sources, as well as providing a way to identify the sources of iron that are present in the process.
CHRIS CLAESEN (Nalco Champion Energy Services)
With regard to the Fe content of the feed, you can often control it by looking at the upstream process. Possible treatments are iron removal in the desalter and corrosion inhibitors for the upstream equipment or the installation of feed filters. In some cases, the catalyst bed pressure can be reduced with a reactor bed cleaner.
SERGIO ROBLEDO (Haldor Topsøe, Inc.)
Fouling of a catalyst bed can be caused by a number of different factors or sequences of event in the refinery; however, in most cases, one of the most commonly encountered contaminants is iron. Organic iron can be present in refinery feedstocks; but more often than not, the iron originates from corrosion within the refinery itself. This is especially true for refineries using naphthenic type crudes. Processing these high TAN (total acid number) crude types accelerates corrosion in plant piping, tankage, etc., due to the presence of naphthenic acids. This iron can have a wide range of particle size distribution ranging from sub-micron up to flakes or lumps that can be one-quarter to one-half inch, or even larger.
When grading for iron, the graded bed must be able to handle the iron in all of its size possibilities. Haldor Topsøe’s TK-25 TopTrap™ is designed to load with an interstitial void of 61% and holds an additional 25% void in the pore system, for a total void volume greater than 85%. This gives TK-25 TopTrap™ the ability to trap not only larger quantities of deposits, but also the ability to trap the smaller particles of less than 10 micron size. Larger-sized material deposits in the interstices between the individual quadralobes. Fines or smaller-sized materials enter the TK-25 TopTrap™ pore system and are trapped within the structure of the particle itself. Not all grading is created equal, and it is not just a question of creating a high interstitial void fraction. Porosity and surface area are very important functionalities for graded bed material for FeS, as well as other inorganic or organic contaminants. TK-25TopTrap™ has proven to be very effective for trapping the small micron-sized inorganic contaminants in the pore size, thus, minimizing the buildup.

This specialty trap would be loaded right under the high-void, high-density topping layer, such as TK-10 or TK-15. This is because when grading for iron, the objective is to keep the material near the top of the vessel where you have the highest amount of void. If iron migrates through the graded bed and gets to the main bed catalyst, it will hit the pinch point where interstitial void is at its lowest. This would then result in the pressure drop building quickly.
AUSTIN SCHNEIDER (Crystaphase Products, Inc.)
Iron is a ubiquitous foulant across all refining service types and is commonly the primary element involved in compound formation, which causes pressure drop. Delta P issues caused by iron tend to persist even after heating train lines have been pigged and upstream feed filter elements have been put in place. Although iron contamination comes in many forms, including both solids and solutes, when considering pressure drop, iron contamination should primarily be examined as a solid. In this form, it is most commonly encountered as the compound iron sulfide.
Upstream feed filters remove a very large portion of solids moving into the heating train and reactor system. A common action taken to reduce reactor fouling is to implement a filter with a tighter mesh, increasing the particle size range it can capture. This usually results in more frequent filter changes/washes, which operators do not enjoy doing at increasing intervals, with the end result being that the fouling rate is rarely impacted.
Pigging removes any residual iron sulfide crust that may have formed during the previous cycle. While this procedure removes fouling buildup and improves the fluid characteristics and heat transfer capabilities across the heating train, it does not do much to change the fouling rate. In fact, pigging can serve to increase fouling generation initially as removing the corrosion products from the inner piping exposes fresh steel to corrosion pathways.
Although necessary, neither of these proactive maintenance processes serve to keep dissolved iron from passing through the feed filters, nor do they prevent corrosion reactions from occurring on the hot skin surfaces of the heat exchanger or furnace tubes.
Much of the dissolved iron will contribute to catalyst deactivation, and there are catalysts in the industry specifically designed to trap these types of organometallics to protect the main bed catalyst. An example of this is shown in Figure 1, which is a SEM (scanning electron microscopy) image of a precious metal catalyst extrudate that has been poisoned by incoming iron contamination, which is identified in red. This limits the catalyst life but has little effect on the pressure drop.
Solid iron typically takes the form of iron sulfide as it enters the reactor via feed filter bypass, scale/corrosion generation in the heating train (Figure 2), or in-situ particle precipitation followed by agglomeration (Figure 3). Assuming the feed filters are run responsibly and continually, we can focus on the latter two: corrosion and precipitation. This is the material responsible for crust formation in the catalyst bed or ring grading layers, resulting in pressure drop.
Incoming iron of this nature is a direct result of energy input on the way to the reactor driving the reactions for corrosion and precipitation. The only option left for the engineer is to deal with the filtration issue raised by iron sulfide generation and to work with top bed technology suppliers to improve the filtration capabilities of the reactor. The volume for particle storage should be increased to improve the resistance to pressure drop. There are materials available that can greatly expand the reactor’s filtration capacity while minimizing the ∆P. These materials work in a uniquely different manner than standard grading and are able to filter without forming a pressure drop. They are differentiated by their sponge-like structure made up of individualized elements. These types of systems can be designed to handle the equivalent volume of several crust layers.
Additionally, characterizing the foulant is also of high value. It can help identify the source of the foulant and the type of filtration or catalyst system needed to trap the iron. Tools to help with characterization include Total Suspended Solids, Particle Size Analysis, Digital and Electron Microscopy (SEM, which is shown below), and some form of elemental analysis. Engineers should consider using all of these tools to separate iron foulant from catalyst fines and other significant particle sources to resolve problems both inside and outside of the reactor.
PAUL CECCATO (Criterion Catalysts & Technologies)
Iron is introduced to a reactor as either solid particulates from scaling or soluble iron from corrosion, which precipitates as solid iron sulfide in the presence hydrogen sulfide. In both cases, a reactor bed grading system distributes the particulates over a volume of catalyst to prevent surface caking and a rapid pressure drop increase upon restriction of the hydraulic channel. Installing layers of grading materials from high void fraction to lower void fraction above the main catalyst bed creates the desired distribution and filtering effect.
For solid iron particulates, large-shaped materials are utilized at the top of the grading package to prevent accumulation and crusting at the inlet of the reactor. For soluble iron, adjusting the activity of the grading materials from very low at the top of the bed to moderate above the main catalyst slows the generation of hydrogen sulfide to allow precipitation of iron sulfide over the full depth of the grading materials. To assist with control over iron sulfide precipitation, Criterion manufactures OptiTrap[MacroRing] 8.0 mm hollow cylinders. The mild activity and macro sized pores of the OptiTrap[MacroRing] allow for precipitation and retention within the substrate for an overall increase in iron capacity of the grading system.
Question 34: When processing cracked naphtha, what is done to ensure that polymerization of the diolefins/olefins will not result in pressure drop problems in a reactor or upstream equipment?
GATES (Motiva Enterprises LLC)
Again, we are trying to prevent the polymerization of the olefin/diolefin. The primary concern is trying to prevent contact with oxygen because that will ultimately lead to gum formation. So the preference would be, if possible, to feed this hot to all the downstream units and avoid intermediate storage. If you will have to put it into tankage, the preference would be to use an internal floating roof tank and add some type of chemical stabilization. So typically, some kind of oxygen scavenger would be put in there. The length of time that material will be staying in tankage may dictate the level of treatment that will be done.
The other concern is that if this material contains diolefin/olefins, as it goes into your feed/effluent exchangers, prior to entering the reactors, it could potentially foul those exchangers. Often, you want to add soak hydrogen or hydrogen upstream of that exchanger. It may need to be considerably upstream to ensure that as the material is going into the exchanger and getting hot, there will be some hydrogen present to prevent that gum formation.

If you are actually going to try and treat cracked naphtha, you will typically want a separate diolefin reactor that will be run at lower temperatures to avoid the polymerization. You will use catalytic hydrogen to eliminate the diolefin and operate it in a regime such that you will not saturate too many of the olefins. You are ultimately trying to put the naphtha into gasoline, so you want to minimize your octane loss for the gasoline pool. As far as the catalyst, look for a catalyst with sufficient surface area and large enough pore volume – going back to Sal’s comment – to manage any silicon or arsenic that might be coming with that cracked naphtha.
WATKINS [Advanced Refining Technologies (ART)]
Two of the reasons we are worried about olefins and cracked naphtha in our naphtha hydrotreater are that they are a large consumer of hydrogen and also because the reactions occur quickly. Once the reaction with olefins starts and we have enough temperature, it is hard to stop. We do not want excess temperature to coke up at the top of our hydrotreater. Like David said, with coker feeds, we would like to see them brought in straight from the coker while still hot. They should not be sent to tankage; and if they are, you should blanket the tank somehow and keep it protected from oxygen.
The top of the hydrotreater is where we are worried about excess exotherm. We want to spread out that heat and do the reaction somewhat slowly, if possible. We do not want to have it occur all at once. Generally, we will recommend a graded bed to mitigate that pressure drop and start olefin saturation to avoid worrying about anything else like HDS and HDN or aromatics. In that case, we will be using a low-metals guard material at the top of our hydrotreaters, which also gives us the ability to pick up silicon.

If your system has a high amount of diolefins, again, a dedicated diolefin reactor is important. At a much lower temperature, you can actually control just the diolefin reaction and avoid doing everything all at once by either controlling how much hydrogen goes to the hydrotreater or by keeping the temperature fairly low.
In the top of the main reactor of a coker naphtha unit, again, low metals, a guard material, is recommended. We generally then recommend avoiding the placement of very high-metals, high-active catalysts at the top, if at all possible, so we can try and do the reactions one at the time.

MUKESH PATEL (Reliance Industries Ltd.)
When we have to enable storage of naphtha, if necessary, which is the parameter we should monitor or what type of analysis should be carried out to determine if any gum formation has happened?
GATES (Motiva Enterprises LLC)
Off the top of my head, I do not know the ASTM (American Society for Testing and Materials) methods for that, but there are gums and then there are potential gums a
We run diolefin saturation tests, and I will put the ASTM method in my Answer Book response. We typically shoot for 60 to 80% diolefin saturation.
SHARPE (Flint Hills Resources, LP)
We run diolefin saturation tests, and I will put the ASTM method in my Answer Book response. We typically shoot for 60 to 80% diolefin saturation.
BRIAN WATKINS [Advanced Refining Technologies (ART)]
Processing coker naphtha can have several undesirable effects on the performance of the hydrotreater and the catalyst if the system was not properly designed to handle it. In general, coker stocks have a higher level of olefins present from the coking process. Once in the hydrotreater, these olefins will quickly get saturated (Figure 1), resulting in high hydrogen consumption and generation of a lot of heat. As a general rule of thumb, one mole of hydrogen is required per mole of carbon-carbon double bonds, or between five and 10 times the Bromine number reduction in standard cubic feet per barrel (scfb) of hydrogen. This additional heat [130 Btu/scf (British thermal unit per standard cubic foot) to 160 Btu/scf hydrogen consumed], if not managed properly, will initiate additional reactions, quickly creating a very high temperature rise. The high temperatures can accelerate coking and can lead to olefin polymerization, resulting in a dramatic increase in pressure drop. This can set an upper limit on how much coker naphtha can be processed based on the need to limit the heat rise or hydrogen consumption.
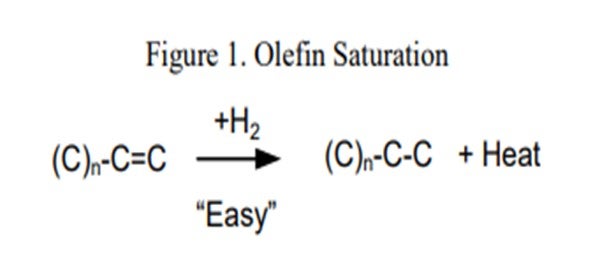
A system that is properly size and activity graded is extremely important when processing coker naphtha. ART recommends utilizing a grading system to help mitigate pressure drop buildup. A large inert hold down ring (GSK-19) with a very high void fraction used for trapping large particulates is placed at the top of the catalyst bed. A smaller diameter macroporous ring (GSK-9) that traps iron, as well as other fine particulates, is typically used in the next layer. After that, two types of smaller rings are used as active grading. These materials have a low level of active metals which help begin olefin saturation reactions, as well as provide additional particulate space at the top part of the catalyst bed. Avoiding the use of any highly active catalyst at this point is also recommended. Below the grading system, it is recommended to use a layer of larger size (1/10” or 1/12”) catalyst which provides activity for olefin saturation and additional void space for pressure drop mitigation. This layer is often a catalyst that is suitable for trapping silicon (and arsenic), which is another concern when processing coker naphtha.
An additional recommended practice to help prevent fouling and pressure drop buildup in coker naphtha units is to avoid contact of the coker feedstock with oxygen. It is preferred to bring the feed to the processing unit directly from the coker and avoid use of tankage. If this is not an option, then the alternative is to use a floating roof tank and purge the system to keep a nitrogen-blanket over the feed in order to keep oxygen out. Use of a diolefin treating reactor can also be considered. A diolefin reactor is operated at a much lower temperature in order to selectively catalyze the diolefin saturation reaction and avoid any excess heat generation from sulfur and nitrogen removal.
RAJESH SIVADSAN (UOP LLC, A Honeywell Company)
When processing cracked naphtha, plugging in feed exchangers and heaters may be caused by coking or polymer formation. Polymer formation can be caused by oxygen in the feed with cracked feedstocks. It is preferred to have cracked feeds fed to the hydroprocessing units directly from the upstream unit. Cracked feedstocks that do not come directly from the upstream unit should be stored in nitrogen-blanketed tankage. For units that process straight-run feeds from tankage, as well as cracked feeds, this straight-run feed should also be stored in nitrogen-blanketed tanks as this feed could provide the source of oxygen for polymer formation.
Processing these streams by themselves usually requires a two-reactor system with the first reactor operating at lower temperature to saturate the diolefins and a small portion of the olefins. The second reactor completes the olefin saturation and removes the sulfur and the nitrogen.
DENNIS HAYNES (Nalco Champion Energy Services)
Some issues may arise regarding polymerization. Polymerization in the feed stream may lead to fouling of heat transfer equipment and plugging of the reactor bed. A first step would be to minimize any potential for oxygen contamination as oxygen would act to accelerate fouling reactions. In addition, consideration may be given regarding the implementation of additive chemistries to inhibit polymerization rate (antioxidants) and minimize the potential for foulant (dispersants).
CHRIS CLAESEN (Nalco Champion Energy Services)
There are special low temperature reactor designs utilized to saturate the diolefins before they can thermally polymerize. Chemical programs with antipolymerants and dispersants can also help control polymerization and ∆P (delta P; pressure differential) increase. Reactor bed cleaning programs have been used successfully to reduce reactor ∆P caused by fouling with polymerized olefins and corrosion products.
RAJ PATEL (Haldor Topsøe, Inc.)
Designing a unit for processing coker naphtha can be challenging. The challenge comes from the silica in the feed which can poison the catalyst, high nitrogen in the feed which may be difficult to remove in low pressure units, and high quantity of olefin which can cause large temperature rises in the reactor, as well as very reactive molecules which are the conjugated diolefins. In the presence of small amounts of oxygen, or at elevated temperatures above 450°F, these molecules will radially polymerize to form gum that can foul exchangers or reactors causing poor heat transfer, as well as high rector pressure drop. If the feed contains a significant quantity of coker naphtha, then these diolefins must be removed at low temperatures to prevent gum formation.
The coker naphtha should preferentially be sent directly from the coking unit to the hydrotreater to prevent oxygen contamination. Even straight-run stock, which may be part of the feed component, must be prevented from contacting oxygen by storing the feed in a nitrogen-blanketed storage tank.
Even with strict adherence to avoiding feed contact with oxygen, the diolefins in the coker naphtha can polymerize at elevated temperatures. A dedicated diolefins reactor operating in the range of 300°F to 450°F will ensure that these highly reactive species are removed from the feed before polymerization can take place. Once the diolefins are removed from the feed, then the feed can be heated to the required temperature for silicon and/or sulfur/nitrogen removal. Topsøe has designed a large number of coker naphtha units with dedicated diolefin saturation units to mitigate the polymerization issues.
MIKE ROGERS (Criterion Catalysts & Technologies)
Hydrotreatment of cracked naphtha derived from FCCU, coker, or thermal cracking is a common refinery requirement. In some cases, refineries process these naphthas in a downstream catalytic reformer; while in others, hydrotreatment is necessary to meet gasoline blending sulfur requirements. Treatment of cracked naphtha can pose special challenges due to the presence of diolefins that are produced by the cracking reactions.
Diolefins in cracked naphtha streams from the FCCU and the coker will polymerize when heated, producing gums that foul the pre-heat equipment and main hydrotreater catalyst. To prevent polymerization, these naphtha streams are treated in a diolefin reactor operating at low but sufficient temperature to hydrogenate the diolefins. Typically, the diolefin reactor operates in a down-flow bubble phase with an LHSV of 2 hr-1 to 5 hr-1 and within temperatures between 180°C and 220°C (350°F to 420°F).
The effectiveness of diolefin saturation and protection against polymerization is monitored by measurement of the diene number in the reactor effluent. Typically, method UOP 326-08 (Diene Value by Maleic Anhydride Addition Reaction) is used to generate a MAV (maleic anhydride value) number representing approximately double the diene content in weight percent. Conversion of MAV in the diolefin reactor is in the range of 90 to 95%, and a target MAV number of less than 1 in the product stream from the diolefin reactor typically ensures protection against polymerization.
Catalysts loaded into a diolefin reactor should have a moderate but stable activity to selectively convert diolefins at low temperatures. For units processing coker naphtha containing silicon, a large surface area and high pore volume provide resistance to poisoning. Lastly, for units processing FCCU naphthas, catalysts with a low hydrogenation activity are favorable to minimize olefin saturation and octane loss.
Question 35: When processing tight oil crudes, are lower bed pressure drop problems in VGO/resid hydrotreater reactors a concern? If so, what mechanisms explain this issue?
LIOLIOS (DuPont Clean Technologies)
The highly paraffinic nature of the tight crudes and the destabilization of asphaltene molecules can cause precipitation and agglomeration. One of our customers with a gas oil mild hydrocracker switched feedstock to increase amounts of black wax crude. This was a five-reactor system. A guard bed reactor was first, followed by four other reactor beds. In the polishing reactor bed, this customer saw an increase in pressure drop. It was theorized that this pressure drop was caused by asphaltene precipitation and polymerization in the bed.

The following graphs show some of what was happening at this unit. It is a constant feedstock. They raised the temperature to get some additional cracking. You will notice an elevated pressure drop in the last bed shortly after they increased the severity of the unit. If you look at the next chart, you can see where they decreased the severity of operation of the unit and the pressure drop recovered. Our theory is that there was a recombination of those asphaltenes.

SHARPE (Flint Hills Resources, LP)
We have had no second and third bed ∆T problems when running high rates of Eagle Ford crude. When there were high bed ∆Ps in the lower treating beds, they were usually a result of coke fouling due to hydrogen starvation, and low hydrogen partial pressure.

GLENN LIOLIOS (DuPont Clean Technologies)
The highly paraffinic nature of tight oil crudes, and the potential increase in asphaltene precipitation when these crudes or cuts of these are mixed with polar asphaltenic oils or cuts, has been well documented. The increase in paraffin content can lead to destabilization of the asphaltene core which can then agglomerate to form larger macromolecules that may precipitate out under hydrotreating conditions.
A number of published documents2 detail the causes and reactions behind this phenomenon and outline methods to determine which crude type and cuts are compatible and what ratios are required to minimize the chance of this phenomenon occurring.
Much of the industry experience indicates that asphaltene precipitation and fouling in process units normally occurs in regions of high heat flux when agglomerated asphaltenes easily crack or dehydrogenate leaving coke-like deposits such as feed/effluent exchangers or where hydrotreater reactions are initiated; i.e., the top bed of a hydrotreating reactor. However, it was observed that a gas oil mild hydrocracking unit experienced a noticeable increase in pressure in a final polishing reactor after the feed to the unit was switched to process a feed that had been mixed with an increased percentage of highly paraffinic (black wax crude) feedstock. At the same time, the severity was increased by lowering the throughput without reducing inlet temperatures. The polishing reactor was the last in a series of five reactor beds, the bed being a separate bed reactor. During the observed increased pressure drop in the polishing reactor, no appreciable pressure drop was observed in the guard bed or main reactor beds. It is important to point out that after the space velocity and feedstock to the system were normalized, the pressure drop decreased almost to the baseline range prior to the event.
It is theorized that the observed bed pressure drop increase in the last bed was a result of asphaltene precipitation and polymerization on the bed that occurred after increased severity reactions cracked the smaller molecules that kept the increased asphaltenes in solution. According to work conducted by Wiehe on asphaltene precipitation3 , asphaltenes are maintained in solution in oil by a micelle type of configuration. This theory has been also explained by other authors4 . The asphaltene core is surrounded by a solvated shell that consists of resins. Resins are molecules with aromatic and naphthenic rings.
Under high severity conditions such as those experienced in this mild hydrocracker operation, the resins can crack into smaller molecules. This can disrupt the micelle type configurations at which asphaltenes are kept in solution, and the asphaltenes can precipitate upon cooling.
Analytical tests carried out on the hydrocarbon feed samples indicated that the asphaltene content (heptane insolubles), although low in comparison with a heavy residue5, was found to be approximately three times higher than the one on the sweet GO FCC feed sample that was being recirculated to the unit and the regular GO sample fed to the GHC.
This theory explains why the upstream reactor beds did not experience a corresponding increase in pressure drop. If it were due to deposits, catalyst fines, or simply rust from upstream units, the first two reactors should have acted as filters preventing the last bed from getting plugged-up.
JUAN ESTRADA (Criterion Catalysts & Technologies)
Two primary mechanisms for pressure drop in bottom beds are coking and asphaltene precipitation. Coking results from operation at elevated temperatures and hydrogen deficiency. Asphaltene precipitation results from a reduction in liquid solvency. The design of VGO hydrotreaters with elevated pressure, low space velocity, and high treat gas rates helps minimize coking; however, elevated saturation of aromatics reduces the solvency of the oil, increasing the potential for asphaltene precipitation in the catalyst bed.
Processing tight oils in the crude diet reduces the aromatic content of the gas oils. For this reason, the coking potential of the feed is lowered, but the potential for asphaltene precipitation increases. With lower feed aromatics and severe hydrotreatment, the solvency change may be sufficient in the lower catalyst beds to precipitate asphaltenes introduced with the other gas oil components from conventional or synthetic-derived crude sources.
The mechanism of asphaltene precipitation from a reduction in liquid solvency has been connected to many historical pressure drop problems involving changes in operation and feedstock qualities such as aromatic and C7/C9 asphaltene contents and the distillation tail. Applying this accepted mechanism to lower bed pressure drop problems in units processing tight oil derived gas oils logically explains recent pressure drop problems in a few VGO hydrotreaters. Refiners continue to learn compatibility limitations of co-processing tight oils in the crude diet, including impacts on VGO reactor pressure drop growth has become a consideration.
Question 36: Has the increased use of tight oil crudes and western Canadian crude been correlated with increased metals and solids in diesel, gas oil or vacuum resid?
SHARPE (Flint Hills Resources, LP)
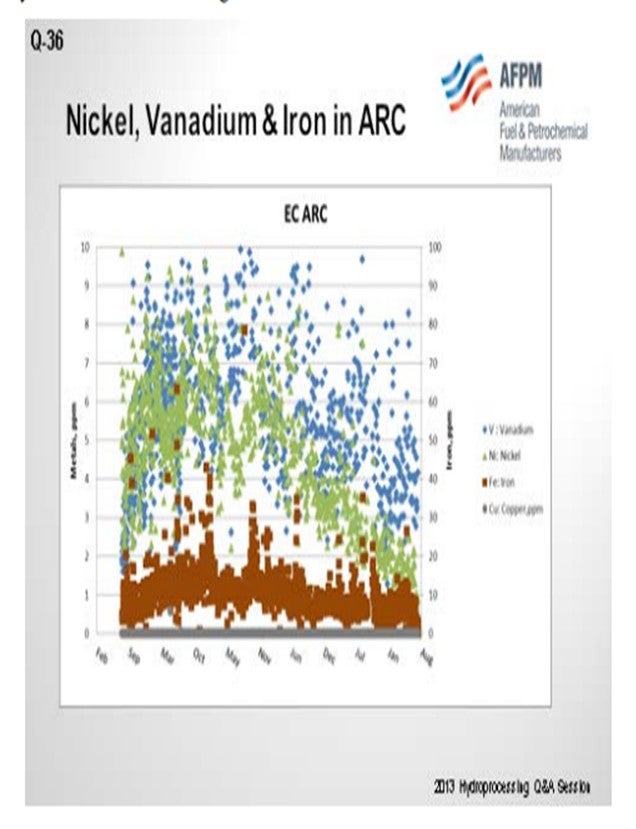
The FHR CC (Corpus Christi) refinery has not seen an increase in raw crude filterable solids. With the increased domestic crude rates, we typically run about 20 to 50 pounds per thousand barrels with these occasional spikes to 100. We have recently started monitoring the filtrate from the 0.45 micron test with 0.1 micron paper. We see loadings in a similar range, but we do not have much historical data for comparison.

We have seen an increase in copper and barium on the FCC catalyst. Vanadium and nickel were decreased by about 50% in the ARC (atmospheric residue) from our tight oil crude unit. The crude units we have been running, with a mix of domestic and some foreign crudes, have not shown substantially decreased metals.

The slide of our atmospheric resid shows that the vanadium and nickel have come down substantially since we started running a lot of domestic.
VICHAILAK (Marathon Petroleum Corporation)
The question is about tight oil and Western Canadian crudes. The two types of crude are on totally opposite sides of the pole. Tight oil is normally light sweet, low in metals, and paraffinic in nature. The prevalent areas where you get this type of oil are Bakken, Eagle Ford, Permian Basin, Niobrara, and Utica, Ohio.

Because tight oils are very low in metals and do not impact the hydrotreaters very much, I learned from my queries that the industry has not seen any increase in the metals from tight oil crude. Nickel and vanadium are usually low and may contain phosphorus, but that is due to the general fracturing agent they add in artificially, not from the crude itself. As I said, we do not see any impact on the processing of tight oil. We have seen barium in one of our reactors that processes Eagle Ford crude, but the barium is mixed in and hard to separate. Phosphorus can be a potential problem, both in foulings and deactivation. I think Brian discussed this a while ago.

Now I will talk about Western Canadian crudes. These are heavy asphaltic diluent bitumen blends. The bitumen portion has very high metals and will contain solids. It depends on how it was processed: either having been mined or produced by SAGD (steam-assisted gravity drainage). This type of crude contains high nickel-vanadium, so the gas oil and Bakken reserve will have very high nickel-vanadium as well.

Marathon has experienced asphaltene precipitation in the pre-heat circuit of our crude unit, but nothing downstream of it. We have not had any issues linked to precipitation in a downstream unit besides the pre-heat exchangers. However, our refinery that processes this type of crude separately has one train treating process only for tight oil and another for heavy crude. The product from the crude unit then gets mixed in between and goes to the same hydrotreater, for example. We can see the potential for issues when this happens. But as far as asphaltene precipitate, we normally do not put asphaltene to fixed bed hydrotreaters anyway. We try to prevent having asphaltene going in, so we do not really see any problems processing these feed streams.

MONTRI VICHAILAK (Marathon Petroleum Corporation)
Tight oil and Western Canadian crude are very different in nature. Tight oil refers to the light, sweet, low metals paraffinic oil from regions utilizing fracturing and directional drilling. The most prolific tight oil regions are:
• Bakken: primarily North Dakota, tremendous growth, two-thirds of Bakken now being railed out due to economics and pipeline constraints;
• Eagle Ford: Texas;
• Permian Basin: West Texas, combined into the WTI (West Texas Intermediate) stream;
• Niobrara: northeastern Colorado, White Cliffs condensate; and,
• Utica: eastern Ohio, volumes very low, mostly >50 API (American Petroleum Institute) condensate
These oils are very low in metals. Except for the phosphorus associated with the phosphorus-based gel fracturing agents.
\Marathon’s experiences from processing Eagle Ford crude are fouling in the lower section of crude tower where the foulant has been identified as barium that had come from drilling mud. We also see a significant amount of barium in fines retrieved from the distributor tray coming from the lead beds of FCC pre-treat hydrotreater. So far, we have not had much problem with processing tight oil in hydrotreaters.
The Western Canadian crudes are mostly heavy, asphaltic, diluent bitumen (dilbit) blends. The bitumen portion has very high metals contents and can have solids/processing issues depending on whether it is mined or SAGD produced. The heavy dilbits with high metals (Ni+V) will thus have gas oils and vacuum resids with high metals.
Marathon did experience asphaltenes precipitation in pre-heat circuit at crude unit when co-processing the tight oil with the Western Canadian crude. As the two opposite types of crude are mixed, the asphaltenes’ solvency reduced, causing it to precipitate out. We have not had issues linked to precipitation downstream of the pre-heat exchange.
For the refineries that have multiple crude units processing tight oil and the Western Canadian crude separately and where the heavy vacuum gas oil from both crude units is sent to a gas oil hydrotreater, there is a possibility that the asphaltene precipitation can occur in the hydrotreater. This problem may be manageable by optimizing crude/vacuum tower operation as initially, there should not be any appreciable amount of asphaltenes in the heavy vacuum gas oil.
GREG SAVAGE (Nalco Champion Energy Services)
High solids content in tight oil, typically iron and calcium salts, concentrate in the residual oil streams. Removal of the solids at the desalter, through optimization and solids removal agents, can minimize the amount of solids in the vacuum residual oil. Calcium and iron have been successfully reduced at the desalter using metals removal additives.
Question 37: How does the increased processing of tight oil (Eagle Ford, Bakken, etc.) affect hydroprocessing operations? With lighter feeds and less sulfur, how can the hydroprocessing reactors and catalyst systems be tailored to optimize performance? What other factors in economics replace volume gain when processing these lighter feeds (i.e., impact on FCC yields, gasoline blending, minimizing cetane giveaway, etc.)?
SHARPE (Flint Hills Resources, LP)
Tight oil crudes have impacted our hydrotreater operations in several ways. Catalyst lifecycles are extending due to the low severity required for treating low-sulfur feed. Low reactor severities have caused emulsion problems due to our not hydrogenating our surface-active compounds in the feed. We have had to set minimum reactor temperature limits on several of our hydrotreaters: every service from jet all the way through gas oil hydrotreating.

The colder hydrotreating temperatures can also result in downstream equipment running too cold. We had salt deposition in places we did not want it, as well as water condensing in unanticipated locations. So with the colder reactor temperatures, you end up having to pay more attention to cold and corrosion issues. Future hydrotreating catalyst evaluations will be considering the benefits of adding dewaxing catalysts. Several papers were presented on this at this past spring’s AFPM meetings.

WATKINS [Advanced Refining Technologies (ART)]
We have not seen any negative impact on the actual hydrotreating process itself. Much like Danna said, running these feeds has made the process significantly easier for the refiner. ON reason is that it gives you the ability to consider processing other opportunity streams, as well as more of those easier streams, in order to gain additional barrels of feed through your hydrotreater. You can also maintain volume swell, since that is what is important, so that more aromatic feeds can be used. Consider, though, that since it is highly paraffinic, you should use some type of dewaxing catalyst in your hydrotreater if you have an issue of selling your products while there is a cold flow property problem.
If your refinery is gearing towards running these lighter sweeter crudes in the long-term, consider changing your hydrocracking catalyst to a more distillate-selective material to actually maintain the diesel yields that you are used to or those you are looking to gain. As a side note, since speed quality varies quite a bit day-to-day, even across the same wells from where you may be pulling, we still recommend using demets or other trapping guard materials at the top of your hydrotreater to make sure that there are no other adverse effects through the poisoning.

VICHAILAK (Marathon Petroleum Corporation)
We operate similarly and have not seen any adverse impacts on our hydroprocessing units. Because the feeds are lighter and sweet, we have the opportunity to raise space velocity or process heavier crude coming into the refineries. As a result, you will start having lower catalyst life and/or lower hydrogen consumption; so it is possible that you might have to burn away hydrogen. However, easier feeds also provide the opportunity to reuse catalysts with extended cycle lengths in order to reduce operating costs. Cold flow properties are definitely a concern, as was mentioned by both Brian and Danna.

MUKESH PATEL (Reliance Industries Ltd.)
Actually, when we say tight oil does not have any impact on hydroprocessing, we are talking about how much time you have to process in the refinery. So, my questions are: How much time do you process, and how many months or years do we process until now?
SHARPE (Flint Hills Resources, LP)
We are increasing our run-lengths by 50 to 100%. That is the impact we typically see.
MUKESH PATEL (Reliance Industries Ltd.)
It is a combination of other fuel and tight oil, right? When we say 50%, we mean 50% tight oil and 50% the other fuel?
SHARPE (Flint Hills Resources, LP)
The percentage of tight oil varies at our two plant sites. But generally, it is a mix of products from tight oil and some of our foreign crudes. That is a complicated answer because of the way our feed systems are set up, so I cannot really give you the answer on that. Depending on which product stream you are observing, it could be diesel, jet, or gas oil. We have two plant sites, and the interconnections between the different units are rather complex.
STEPHEN PERRY (Motiva Enterprises LLC)
Does anyone have any experience with noble metal catalysts? If there is a little sulfur in the light tight oil, could you actually get a noble metal to work and increase hydrogen consumption and volume gain?
SHARPE (Flint Hills Resources, LP)
We do not.
VICHAILAK (Marathon Petroleum Corporation)
We cannot get to that level yet.
UNIDENTIFIED SPEAKER
Including the patent.
ROBERTSON (AFPM)
You better run out the door now. Wait. The government is closed. You cannot go to the Patent Office. If they get caught working, they will be arrested!
SHARPE (Flint Hills Resources, LP)
Keep your day job, Gordon.
ROBERTSON (AFPM)
If you live in Washington, you understand.
BRIAN WATKINS [Advanced Refining Technologies (ART)]
While tight oil trades at a discount compared to other sources, refineries with ready access to tight oil will be able to shift toward more hydrotreating, including demetallization and dewaxing, and away from a hydrocracking focus. This is still primarily limited to U.S. mid-continent due to transportation limitations. If a refinery is gearing towards structurally processing sweeter crudes, then there is also an incentive to tweak the hydrocracking catalysts toward the milder, more distillate selective part of ART’s hydrocracking catalyst portfolio. Since tight oil quality varies widely (even within the same area), it is advisable to continue to add additional demet and dewaxing capacity and continue to use advanced hydrotreating catalysts.
Looking at comparing a more naphthenic feed slate to one that is more paraffinic and understanding the impact on the various hydroprocessing units in the refinery is important. The use of these tight-oil more paraffinic type crudes (Bakken and Eagle Ford) will lower the required severity on the average U.S. ULSD hydrotreater. Understanding how these changes will directionally affect the various processes is important in order to provide some planning, as there are both positive and negative effects with this change. The lower severity will reduce the required hydrotreating catalyst volume needed in order to achieve the same sulfur specification thereby creating some additional catalyst volume which can be utilized to address some of the product property deficiencies resulting from these types of crudes. The use of tight oil crudes may also reduce the use and expense of using cetane improver to achieve the required cetane as, by nature, paraffins have higher cetane numbers (see Table 1). More paraffinic feedstocks will decrease the hydrogen consumption of a hydroprocessing unit as well.
In general, a feedstock that is more naphthenic will have a higher hydrogen consumption per barrel due to the increased concentration of aromatic compounds. With this increased aromatic concentration, the likeliness that the sulfur or nitrogen is bound up in one of these molecules is greater, making it more difficult to process in the hydrotreater. These feedstocks would require more aromatic ring saturation in order to remove the sulfur and nitrogen, hence increasing the hydrogen consumed. The increased aromatic content of these feeds also can influence the product cetane in ULSD (Table 1).
With higher aromatic concentration, however, there is a much greater potential for increased volume swell (Figure 1). Additional detail on volume swell with aromatic saturation can be found in the AFPM paper, “Understanding Catalyst Systems for Higher Yields of Diesel” AM 13-10.
Those feedstocks that are more paraffinic in nature, Bakken and Eagle Ford, are much easier on the hydrotreating operations in the refinery. The sulfur and nitrogen molecules in these feedstocks are not as likely to be bound inside an aromatic ring and can be removed via direct abstraction, which is significantly faster than having to complete a saturation step, as well as the sulfur or nitrogen removal. This will provide either additional cycle time, as the required temperatures assuming equal feed sulfur will be lower, or there will be opportunity to process additional barrels per day in order to make up the loss in product yields.
These feedstocks also indicate an increase in light gas make as well. With higher paraffin content comes less aromatic saturation opportunity, which will produce less volume swell per barrel than what is seen with a naphthenic feedstock. This also reduces the burden on the hydrogen system due to lower consumption. The high paraffin content of these feedstocks can also influence the cold flow properties requiring additional processing such as catalytic dewaxing or the blending of other valuable streams in order to meet these requirements. Another positive with the processing of a more paraffinic feed is that for ULSD service, an increase in paraffins will provide a much better cetane value than aromatic species of similar carbon content, as shown in Table 1.
MONTRI VICHAILAK (Marathon Petroleum Corporation)
We have not seen any adverse impact of increased processing of tight oil in our hydroprocessing operations. As mentioned earlier, tight oil crude is light sweet and easy to process. This gives refiners opportunities to either raise LHSV or process heavier crude without significant changes in operational requirements (i.e., hydrogen consumption, catalyst life, etc.). Minimally, processing tight oil gives an opportunity for catalyst reuse with extended cycle length.
HENRIK RASMUSSEN (Haldor Topsøe, Inc.)
Catalyst cycle lengths have gotten longer due to the lighter feeds and lower sulfur in the tight oils. Catalyst systems need to be tailored to match the cycle length with potential pressure drop issues due to longer cycle lengths. Also, refiners now have the option of processing more barrels or blending with even worse fossil crude and still maintaining the same hydrogen consumption and SRU capacity while meeting the minimum cycle length.
Challenges are that there will be more light products produced from a hydrotreater and the fractionation overhead section may not be able to handle higher rates. This would then limit the unit throughput which could mean a reduction in the feed rate.
The tight oils are also more paraffinic in nature and as such will reduce the possible volume swell compared to most fossil crudes. The paraffins will also contribute to worse cold flow properties of the diesel fraction but it will result in a higher cetane.
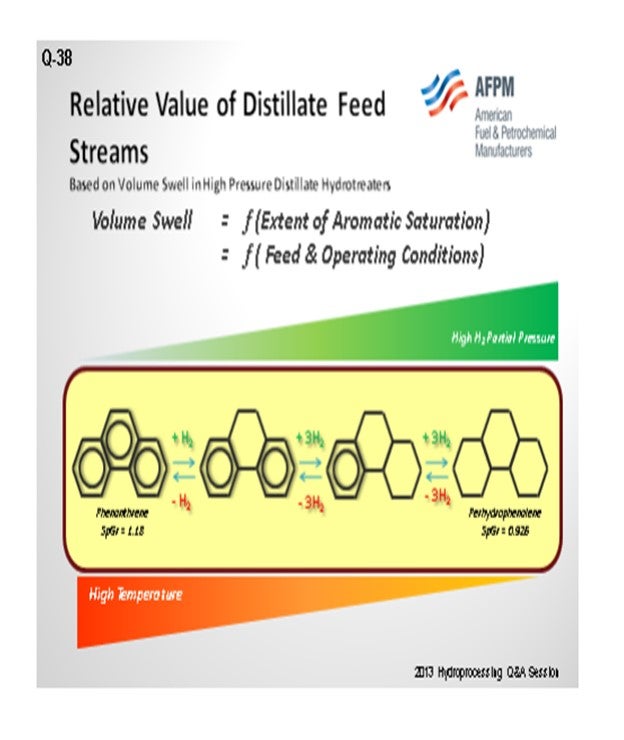
Question 38: Elaborate on the relative value of the various distillate feed streams in a refinery (i.e., straight-run diesel, light atmospheric gas oil, light vacuum gas oil, light cycle oil, coker distillate, kerosene, coker naphtha, heavy cat naphtha, and other) when considering maximum saturation and volume swell in high-pressure ULSD service.
VICHAILAK (Marathon Petroleum Corporation)
The question asks specifically about high-pressure ULSD service. ‘High pressure’ means the reactor has to operate around 1,000 pounds without any problem supplying hydrogen to these units. The relative value of the volume swell depends on how much you can saturate aromatics. This aromatic saturation is an equilibrium reaction, so you need high hydrogen partial pressure on the lower temperature side. If the temperatures are too low, you do not have the proper reaction rate; you have to be right at the peak. If the temperature is too high, then you will go the opposite way: Instead of saturating, you will de-saturate.
I am showing this graph from the 2011 ART CATALAGRAM® because I believe it answers the questions. You can see that the lower the API gravity, the higher the volume gain of a diesel stream. When plotted, you see that it is the same picture, just repeated. You want those two values. Heavy coker naphtha, which has a high API gravity, will show the smallest volume gain, followed by kerosene and heavy cat naphtha, and then other distillate feeds.
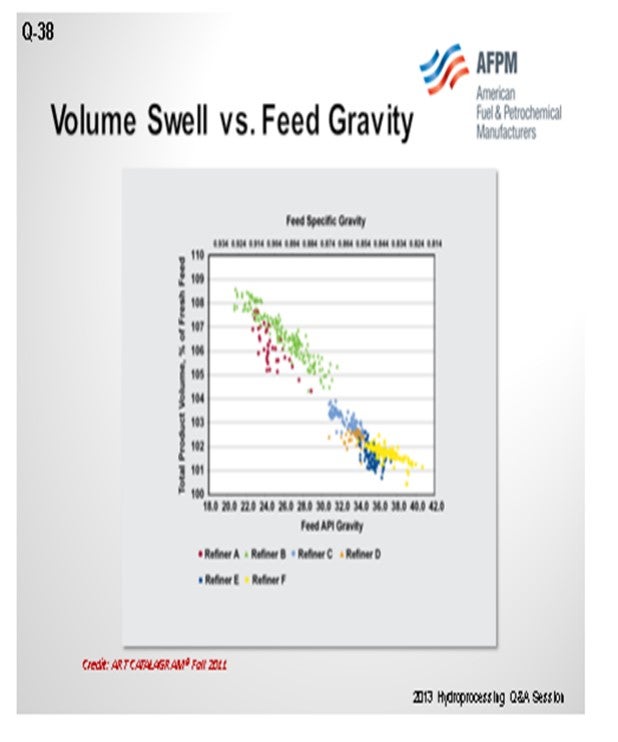
The feeds with a high API will have a lower volume gain. LCO (light cycle oil) has the maximum volume gain. Coker distillates do have a lot of cracked material inside, mostly olefins, but also contain a lot of sulfur and nitrogen, which, when removed, become volume loss.
LIOLIOS (DuPont Clean Technologies)
We agree with those comments. I want to add the information shown in bold at the end of the slide. As you consider what you are going to do with those particular feeds, you must look at cetane giveaway. As you do more saturation, you need to determine if you exceeding your cetane rating, resulting in some giveaway there. Although lubricity from oversaturation does not necessarily lead to reduced fuel lubricity, there is certainly a lot of discussion in that area. Oversaturation can lead to pour point and cloud point issues as you convert more of the compounds. Then depending on your feedstocks, you will end up making more light ends and less desirable products. Again, it is all economics, but the primary driver for volume swell really is the availability and cost of hydrogen.

SIVADASAN (UOP LLC, A Honeywell Company)
I do not have much to add on except one point: that the product qualities obtained when you are processing a cracked stock might be poor and may require further treatment or blending in the downstream units.

MONTRI VICHAILAK (Marathon Petroleum Corporation)
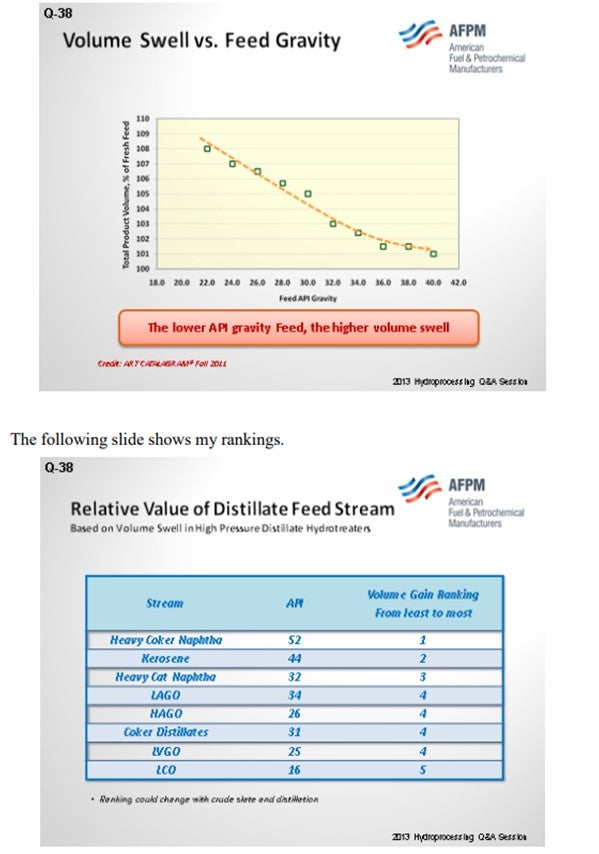
Volume swell in distillates is directly related to the extent of aromatic saturation. Aromatics content of distillates material is also directly related to its density (API). The lower the API gravity means the higher aromatics content, and vice versa. Below is the list of API for each of the distillates hydrotreater feed’s components.
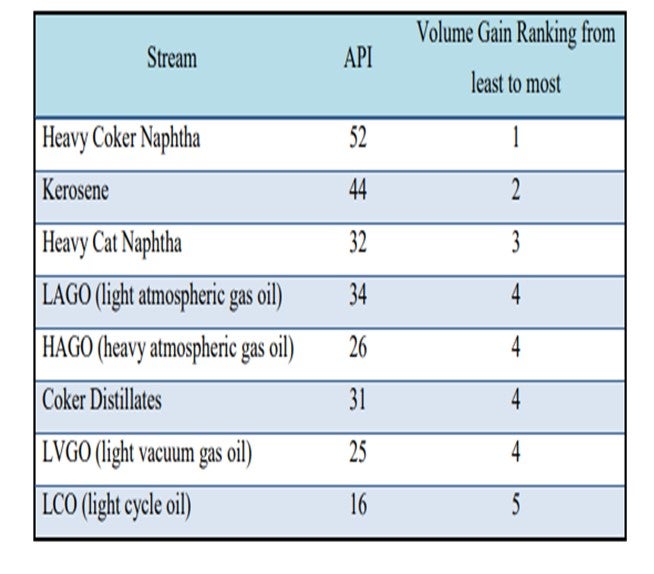
Saturation of olefins and diolefins also give volume gain in the range of 2 to 4%. Aromatic saturation generates at a minimum of 9 to 10% volume gain depending on how many aromatic rings get saturated. Therefore, streams that contain higher, polycondensed aromatics will generate more volume gain in high-pressure ULSD services. Removal of heteroatoms (sulfurs) creates mass loss, and that usually means volume loss.
Aromatic saturation reactions are equilibrium reactions. The ability to saturate multi-ring aromatics depends not only on operating condition, but also on feed composition. Higher saturates rings in feed will give less aromatic conversion than lower saturates rings in feed. Thus, straight-run material in feed will limit volume expansion potential. Therefore, straight-run material should be minimized to process in high-pressure units in order to maximize volume gain. The cost of hydrogen is also important. When hydrogen costs return to historical patterns, volume swell will be much less attractive.
Crude sources also impact the aromatic content in distillates cut. However, for a particular type of crude source, straight-run material normally contains the least amount of aromatics. Thermal cracking material (i.e., coker) contains significant amounts of olefinic and diolefinic materials. Light cycle oil (LCO) from catalytic cracking has the highest content of aromatics comparing to other distillates sources.
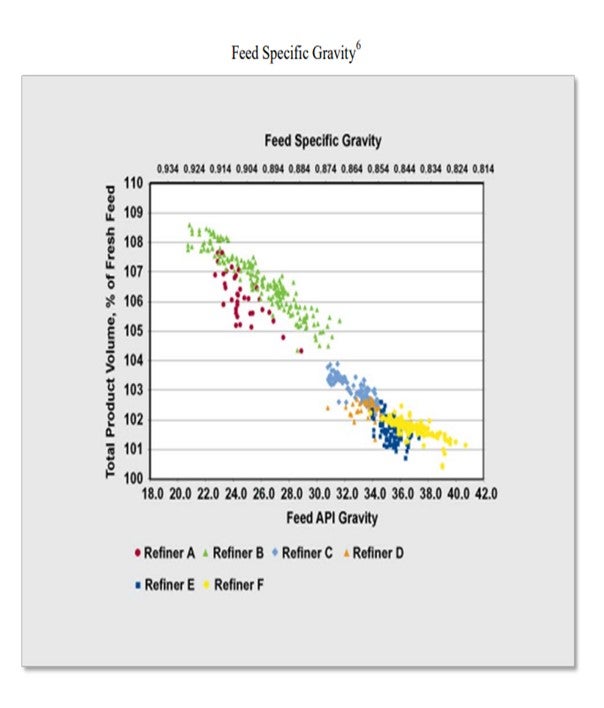
GLENN LIOLIOS (DuPont Clean Technologies)
DuPont agrees with the response from Marathon on the relative value of the different distillate feed streams related to volume swell. However, the availability of natural gas in North America has dramatically changed the hydrogen production economics and its impact on hydrotreating.
Cost of hydrogen and geographical location of the hydrotreater are also important variables to determine the economics of the volume swell. For example, in North America, the current price of natural gas is such that the manufacture of on-purpose hydrogen is a cost-effective means of increasing the volume of diesel. However, in other regions of the world, natural gas is often priced at parity with oil. In this case, volume swell using on-purpose hydrogen may not be a cost-effective option.
Other factors that need to be included in determining the optimal value of a particular feed stream when considering volume swell in the current low natural gas price environment in North American are:
• Cetane Giveaway: Saturation of aromatic and olefin compounds will result in an increase not only in volume, but also cetane ratings. While generally desirable, if cetane levels are higher than the distillate pool requirements, the operating cost or capacity restrictions may not justify either the volume swell or cetane boost.
• Lubricity: In general, oversaturation and the subsequent reduction in aromatic compounds, especially polynuclear aromatic and aromatic nitrogen compounds, can result in fuel lubricity issues. Lowering sulfur, nitrogen, or aromatics per se does not necessarily lower fuel lubricity; but in general, these compounds are known to have better lubricity than aliphatic compounds.
• Pour Point and Cloud Point Increase: Oversaturation may also increase pour and cloud points as aromatic compounds are converted to naphthenes or paraffinic compounds.
• Distillate Yield Reduction: The increase in reactor severity to achieve higher aromatic and olefin saturation can increase the conversion of distillate to lighter less valuable products such as naphtha and LPG. The higher severity can also increase catalyst aging and thereby reduce reactor run-length.
RAJESH SIVADSAN (UOP LLC, A Honeywell Company)
The biggest swell potential is in the cracked stock feeds. The streams constituting the cracked diesel (coker and LCO) will have the highest volume expansion and hydrogen uptake and will have the highest upgrade across the units. But product qualities from these streams are poor relative to processing straight-run, so it may be harder to meet tight specs. Consequently, refineries should really run an economic analysis.
DAVID VANNAUKER (Haldor Topsøe, Inc.)
Many sites have built new high-pressure units to meet the ULSD specifications for feed blend with lots of cracked stocks and high endpoints. The primary benefit is that the higher pressure improves the aromatic saturation and thus the kinetics for removing the difficult dibenzothiophene (DBT) sulfur species. The high-pressure units should process the heavy virgin distillates, such as the heavier component of the light cycle oil and the heavier component of the light coker gas oil. The wide range of virgin kerosene and diesel boiling material and coker and FCC diesel and kerosene boiling range molecules should be processed in the lower pressure units.
From the perspective of volume swell, the streams with the highest aromatic contents should be processed in the high-pressure unit. The highest volume swell is realized with FCC streams, followed by coker and then virgin streams. To maximize diesel production, one needs to consider the boiling range. Since converting an aromatic molecule to a naphthene reduces the boiling range, light aromatics upon saturation may move out of the diesel boiling range and into the naphtha range. For a site trying to maximize overall diesel production, this is another justification for having the lightest diesel boiling range cracked stocks in lower pressure units. When aiming for maximum volume swell, it is, of course, very important to have the required hydrogen availability to avoid hydrogen starvation.
LARRY KRAUS (Criterion Catalysts & Technologies)
When considering maximum saturation and volume swell in a high-pressure ULSD unit, hydrogen-deficient light cycle oil and coker light gas oil offer the largest opportunities for consumption of low-cost H2 to increase product volume. However, the ability to utilize these feeds is dependent on the availability of H2 at the unit, cetane constraints, and the desired cycle life. Optimizing cutpoint of the light cycle oil and coker light gas oil streams can improve volume swell within these constraints.
Straight-run diesel is valuable for its lower density and higher cetane characteristics; however, its low aromatic content limits its H2 consumption/volume expansion potential. Upgrading light vacuum gas oil has high value and increased potential for volume expansion potential versus straight-run gas oil when pushing the T-90 point to the maximum specification. Lastly, light feeds are valuable when they can be used to expand the distillate pool up to the flashpoint limit. Light products can have deleterious effects on ULSD operations by increasing H2 consumption through olefin saturation and reducing the hydrogen partial pressure through vaporization.
Question 39: There is a drive to target the highest endpoint possible on all distillate feed streams when maximizing overall diesel production. Are there feed streams that should be targeted first, considering operational impacts of such optimization, impacts to catalyst performance and life cycle, as well as cutpoint optimization between distillate units and the FCC?
VICHAILAK (Marathon Petroleum Corporation)
For maximum diesel, in general, we prefer getting the distillate out of the FCC. I think we had a discussion last year in the FCC forum during which we said that keeping some distillate in the FCC feed is a benefit. We still say that if you want to maximize the diesel, heavy-out the straight-run distillate of virgin gas oil because it will be the easiest to treat compared to the other two, which are coker distillate and LCO. Coker distillate is the second target because it has high sulfur and nitrogen. Light cycle oil streams are the last on which you want to increase the endpoint because most of the sulfur in there is very difficult to treat. Because if you are heavy on that part, you will probably have to back out a different easy stream to make room for it, keeping in mind that you must make the product spec.

Endpoint maximization depends on the capability of distillate hydrotreaters. Most of the time, if the easy stream (like light straight-run diesel) normally goes to one of the low-pressure hydrotreaters, while most of the cracked stock will then go to the high-pressure units as per design. Hydrogen availability at the site also dictates how you are going to maximize endpoints. You can upgrade low-quality diesel feeds like LCO and coker distillate, but the cetane of that cracked stock pool will normally be low. High paraffin content in crude may limit virgin feeds because you need a bit more aromatics in there. But for any cracked stock that you add to a unit, you will probably have to back out some of the easy stream. If you treat them together, our rule of thumb is: One barrel of coker distillate will replace three barrels of virgin feed.

WATKINS [Advanced Refining Technologies (ART)]
If you are looking for what you will consider for max diesel production, there are a couple options to consider. One is to get more barrels into your ULSD hydrotreater. The other is simply trying to get more barrels on the backend of your diesel hydrotreater. So, one is volume swell, and one is just putting in more barrels. This chart examines the initial understanding of increasing endpoint might do in terms of how much sulfur is coming into the unit. It is evaluating LCOs, coker gas oils, and straight-runs. So, if we want to just change up the endpoint of our feed regardless of feed stream, we will actually start increasing the amount of hard sulfur coming into the hydrotreater. So, we will want to pay attention if we have a hydrotreater that used to have a 650°F or a 750°F 90% point because we will now go to around 750°F to 780°F, which will have a serious impact in the terms of actually processing for your hydrotreater.
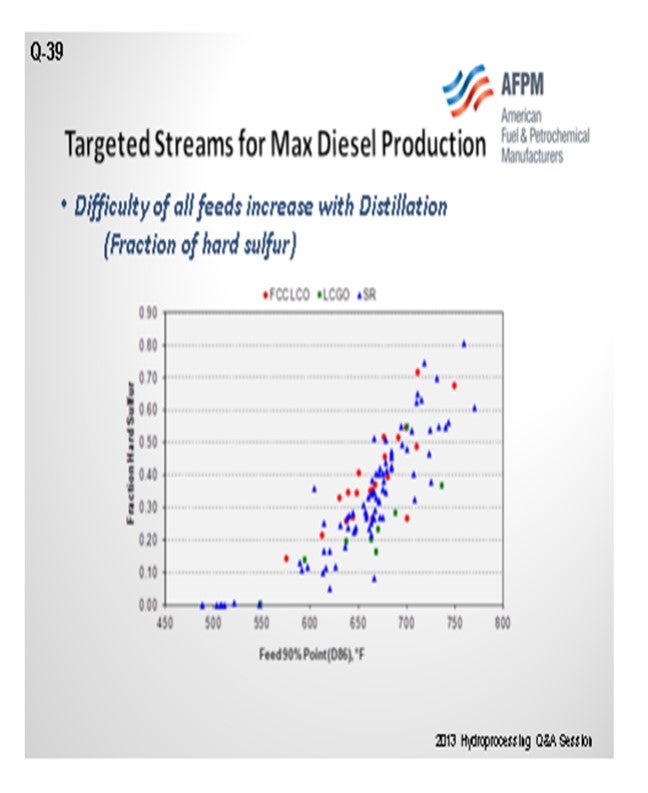
Virgin (SR) Feedstocks: If you are going to then want to think about what you will get from max diesel, if you can get more barrels into your units and are not constrained in that sense, then certainly bringing in a virgin feed stock is good. It will have minimal impact and will not change your hydrotreater cycle dramatically; so, the activation rate will not go up as much, nor will it consume as much hydrogen as your other two streams might; so, it is the easiest on your general operation.
Coker Distillates: It is kind of a one and a half choice. It is clearly a second, but it is almost interchangeable. It does have higher sulfur and nitrogen than a virgin feedstock, assuming the same distillation. As you mentioned, it has higher olefins so it will have more hydrogen consumption. If that is the feed stream on which you want to consider increasing endpoint, then make sure you have enough hydrogen in your system to handle that consumption because it will also require a higher weighted average bed temperature in order to get to the same ULSD targets.
FCC Light Cycle Oil: This feed would be your third choice. It will definitely have higher aromatics, PNAs (polynuclear aromatics), than a virgin feedstock of the same boiling range, a much lower API. Sulfur and nitrogen concentration will depend on your pre-treater or a lack of a pre-treater in front of the FCC. It could be very high sulfur or very low sulfur and nitrogen, depending on how that process is operated. This will definitely require higher temperature in your hydrotreater, so it will shorten your cycle length the most. It will also consume quite a bit more hydrogen in order to achieve that same level of product qualities you are used to.

My next slide shows that it could actually be your first choice, if you are looking for volume swell. That was the other piece of our puzzle. Is it more barrels in the front or more barrels in the back? If you have high enough pressure, you will have the incremental hydrogen to add to your process to avoid being limited. It could actually provide you with the maximum volume expansion or the actual largest cetane increase that you could see over any other feedstock. The chart compares the results when a refiner went from 100% light cycle oil to 100% straight-run during its process. You can see that the biggest cetane increase results from the inclusion of the most LCO. But obviously, it comes with the caveat that you're starting cetane is significantly lower if you run 100% light cycle oil. So, consider that you may not meet a 45- cetane specification because you started at 20.
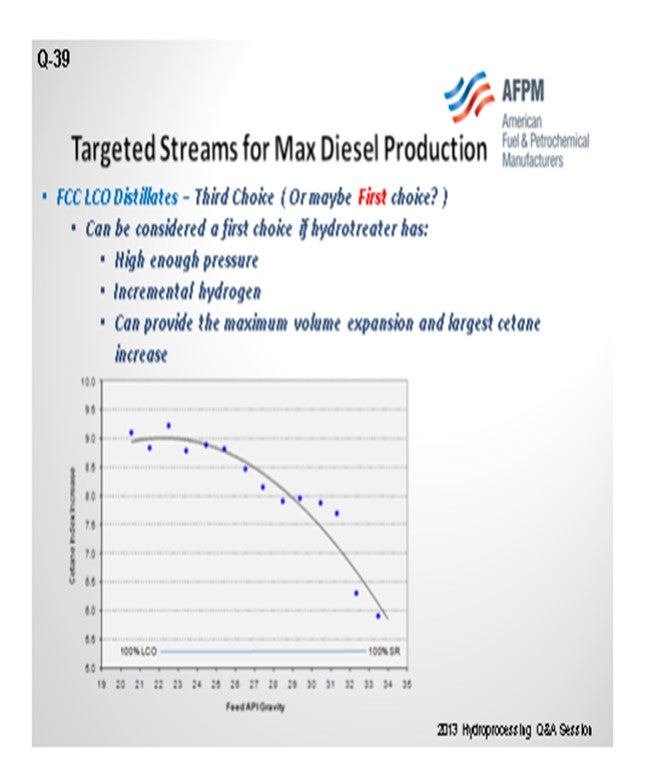
KEVIN PROOPS (Solomon Associates)
Those were all good comments. I want to particularly talk about Brian’s slide that showed the amount of hard sulfur in the streams as a function of TBP (true boiling point). To the panelists, if we are going to put more hard sulfur in the unit and start challenging the unit operation, does it make sense to start taking out some of the light material if you have a unit with that has a capacity constraint and in which space velocity is getting tough? Should we be taking the light cracked distillates in the front end of the light cycle oil or the front of the coker distillate out of the unit and putting them somewhere in a lower-severity unit, keeping the higher-boiling molecules in the unit that is better equipped to deal with them?
WATKINS [Advanced Refining Technologies (ART)]
Feed selection would really depend on the hydrotreating capabilities at your refinery. If you have another unit out of which you can pull those light barrels and continue to process, then it would make sense to do so in order to then pull that heavier diesel into your diesel hydrotreater. You will want some of that light material in your hydrotreater feed in order to give you some extra heat of reaction. It will easily be processed in your ULSD hydrotreater because that unit probably has more horsepower than if you had to try and bring it into your kerosene unit or some other light processing unit. It will really depend on the horsepower of your ULSD unit. If you want to take it out, it may just be going for the ride.
GEORGE HOEKSTRA (Hoekstra Trading LLC)
Every year in the industry, there are many cycles that are much shorter than expected. Sometimes that is certainly caused by pushing too hard on your feed. Other times, it is caused by the catalyst or by bad flow distribution, for example. So, I have a multiple-choice question: Do the panelists have any thoughts on which one of those three is the most likely cause of a short cycle in a diesel unit? Is it the feed, the catalyst, or the flow distribution?
VICHAILAK (Marathon Petroleum Corporation)
Before putting the catalyst in the reactor, you should evaluate what you plan to do for that cycle and try to protect it and preserve activity. Do you have a budget of how much temperature you will allow? If you have enough activity, then you can just heavy-up the feed and go further. That catalyst side is planned, but flow distribution of coke in the bottom of the reactor is not planned and could happen. So I would pick the last one to be the main reason. It will be your call to heavy-up the feed when you know, for certain, that there is not enough activity.
LIOLIOS (DuPont Clean Technologies)
One of our famous consultants told us, “A catalyst does not deactivate. It is always murdered.”
WATKINS [Advanced Refining Technologies (ART)]
I agree, because we see deactivation as being feed-driven. You start your cycle with one feed, and you end it with something completely different. Management is giving you opportunities every day, and you have to fight through it. Feed quality tends to be a lot of what shortens your cycle.
SHARPE (Flint Hills Resources, LP)
Yes: thinking, planning, and economics
SERGIO ROBLEDO (Haldor Topsøe, Inc.)
I have a general comment. With regard to diesel hydrotreating, most people look at D86 distillation. Often, fractionation is not that great. We have seen people who do SimDists (simulated distillations) where the tail is 100°F different from what you gain on D86. So, it is a good idea to look at D86 and your SimDist when changing your endpoints on a D86 basis.
SHARPE (Flint Hills Resources, LP)
When you increase your endpoint, you also increase nitrogen in the feed. The ULSD catalyst can be very sensitive to nitrogen, which forces you to raise your reactor temperatures to meet your sulfur targets. That can lead to more rapid deactivation. So, when you start working on high endpoints, you need to watch your nitrogen as closely as your sulfur compounds.
GATES (Motiva Enterprises LLC)
I will put the same caution on SimDist: You should always double-check to confirm you are looking at the final distillation quantity actually being reported. There may be a hidden tail that could get you, even on a SimDist, above and beyond the D86.
MONTRI VICHAILAK (Marathon Petroleum Corporation)
Straight-run diesel or atmospheric gas oil streams should be targeted first because they contain less sulfur and nitrogen and are the easiest streams to treat, compared with other cracked material with the same EP (endpoint). Maximizing the straight-run diesel endpoint also helps to debottleneck the heavy end processing units as well. Cracked distillates normally contain high sulfur and nitrogen. If the operating condition of ULSD processing is already limited, increasing the cracked material endpoint may require reducing throughput of the unit in order to meet ULSD product spec. Our approximation for tradeoff barrels between coker distillates and straight-run diesel on EP is a factor of 3.
Increasing light cycle oil endpoint increases the number of aromatics and heavy sulfur to the unit. For medium to high-pressure ULSD units with tight treat gas ratios, the concern will be significant increases in hydrogen consumption, which will reduce hydrogen partial pressure of the bottom beds. Also, the sulfur species in the heavy ends will be much more difficult to be treated, necessitating higher operating temperature. The combination of conditions required to meet product sulfur spec can lead to significantly higher deactivation rate due to coking.
BRIAN WATKINS [Advanced Refining Technologies (ART)]
There are several basic impacts that need to be understood when considering increasing the final boiling point of a particular feed stream in the refinery. With elevations in the final boiling point of the feed, the total sulfur and nitrogen, the concentration of hard sulfur, and aromatics content all increase. This is independent of the source of feedstock (Figure 1). In order to maintain the same product specifications, this will need to be compensated for by increasing the reactor temperature. Examining your available hydrotreaters and their processing capabilities, as well as the availability for an increased demand for hydrogen in one area or another in the refinery, is important to avoid unexpected turnarounds.
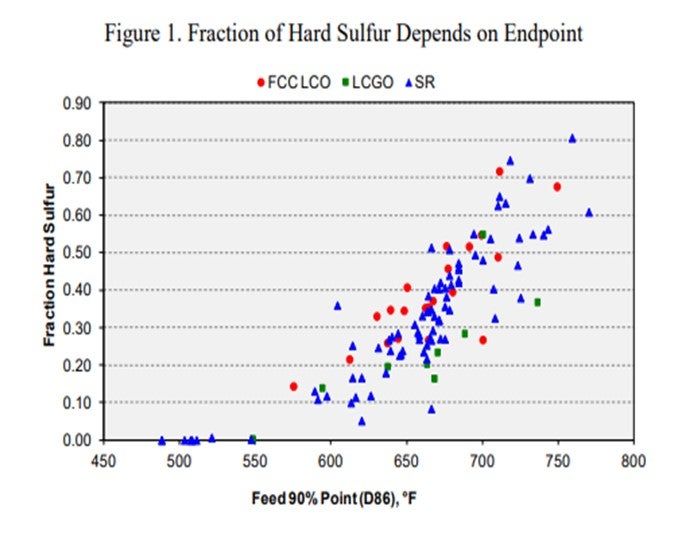
In order to rank which feed source to consider for increasing the FBP (final boiling point), a good understanding of the current situation in the refinery and the ultimate impact of the change is important. In general, cracked stocks (cokers and LCOs) are more challenging to hydrotreat and require a higher weighted average bed temperature (WABT) compared to a straight-run (SR) material at the same product targets. Coker stocks normally have higher sulfur and nitrogen content based on the nature of the feed to the coker and the coking process. These feeds also have a much higher olefin content (or Bromine number) than SR feeds. The higher concentration of sulfur, nitrogen, and olefins will produce a higher heat release when operating to the same product targets, giving rise to an increase in hydrogen consumption. When increasing overall diesel production by increasing feedstock endpoint coker stocks are probably the second-best option for catalyst longevity behind straight-run feeds.
Feeds from the fluid catalytic cracker (FCC) are much higher in aromatics than their SR counter parts and are especially concentrated in the polyaromatic content. This also gives these feeds a much lower API gravity relative to SR or coker stocks. The concentration of sulfur and nitrogen in these feeds is dependent on the presence of a pre-treater upstream of the FCC which can help in lowering the total sulfur or nitrogen content. However, the sulfur and nitrogen compounds in FCC are usually the more difficult refractory species requiring a much higher required WABT to achieve the same product specification (Figure 2).
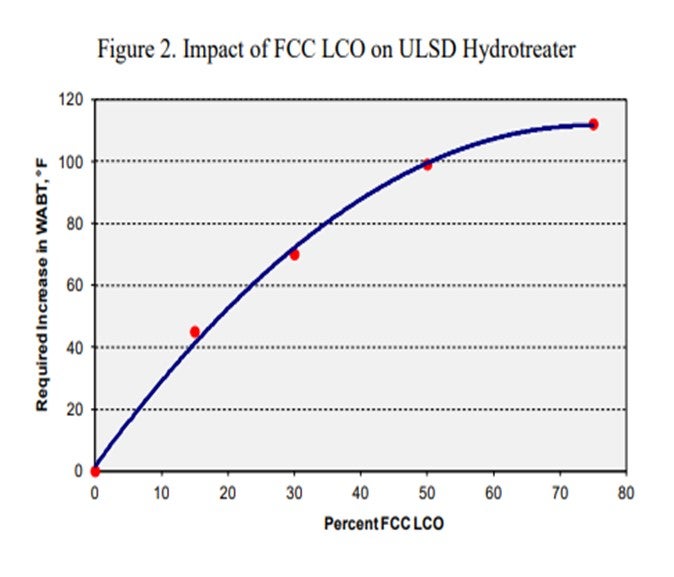
Not all feedstocks provide equal economic benefit. If your distillate hydrotreater has incremental hydrogen available and operates at high enough pressure, LCO may be the first choice for maximum economic return. LCO normally contains the highest amount of aromatics and therefore will provide the maximum volumetric expansion from hydrotreating. LCO will also undergo the largest increase in cetane across the hydrotreater as shown in Figure 3 below.
As always, ART is prepared to discuss these options and assist in making the best choice with the catalyst system already in use. We are also able to help you prepare for the next catalyst load in order to take advantage of these various paths.
DAVID VANNAUKER (Haldor Topsøe, Inc.)
The realized diesel feed endpoints depend upon the crude slate and overall refinery operations. From the hydroprocessing operational ease perspective, the virgin feeds should be maximized first, followed by the coker and then the FCC streams. In an economic environment favoring diesel production, keeping virgin and coker diesel boiling range molecules out of the FCC will maximize site diesel production and profits. There are additional considerations that impact the final realized feed endpoints. With waxy high paraffin content crudes, the virgin feeds may be limited by cold flow properties. The light coker gas oil typically has the same endpoint as the virgin feed, but sites typically have a lower endpoint for light coker gas oil due to coker drum cycles and the resultant coker fractionator instability. The LCO endpoint will be limited by unit operating pressure, hydrogen availability, and diesel pool cetane. In addition to very active distillate hydrotreating catalyst, Topsøe also makes distillate dewaxing catalyst, TK-932, which is added at the bottom bed of the reactor to isomerize the long normal chain waxy molecules to improve cold flow properties.
LARRY KRAUS (Criterion Catalysts & Technologies)
Stream value economics are refinery specific. In general, increasing DHT feed endpoint can increase DHT unit throughput and provide additional material for the diesel pool; however, feed endpoint has a critical impact on feed processing difficulty. Increasing the endpoint increases the multi-ring aromatic content along with the concentration and complexity of sulfur and nitrogen species.
Increasing feed endpoint makes achieving ULSD product specifications more challenging. Higher SOR temperature requirements, higher deactivation rates, and shorter cycle lengths are expected when increasing the feed endpoint. The benefit of increased feed endpoint is determined from the incremental value between the additional feed throughput and the cost of additional H2 consumption, the negative impact on product properties, and the reduction in cycle length. In general, the ability to increase feed endpoint is ultimately limited by the product T-90 specification.
Increasing the endpoint of cracked distillate streams, such as coker light gas oil and FCCU light cycle oil, typically offers the highest upgrading margin in the absence of a hydrocracker but the most processing difficulty in the hydrotreater. These highly aromatic streams contain sulfur and nitrogen species which require a high hydrogenation environment to meet ultra-low sulfur and cetane targets. Elevated hydrogen consumption and operating temperatures combined with additional coke precursors will shorten the hydrotreater cycle life. Maximizing the endpoint of the coker light gas oil and FCCU light cycle oil is an optimization between economic value and hydrotreater capability. Lower hydrogen partial pressure and higher space velocity units will be less capable of processing these streams having a higher feed endpoint.
Increasing the EP of straight-run distillate typically has less margin of improvement than for cracked stocks but increases available throughput. As a less aromatic stream, the endpoint is limited more by the T-90 specification than by the complexity of the sulfur and nitrogen species or cetane. Hydrotreater cycle life will be reduced due to higher required temperatures.
Question 40: Economic drivers dictate the need to process more difficult feedstocks, such as cracked stocks and diesel feeds with high distillation endpoints. What are typical guidelines regarding minimum hydrogen partial pressure for a given feedstock? (High-level answer with details will be discussed in the Hydroprocessing P&P.)
LIOLIOS (DuPont Clean Technologies)
We are treating feedstocks with higher endpoints and greater volumes of cracked feedstocks. Certainly, you are faced with refractory sulfur species that are harder organic nitrogen compounds, which then inhibits sulfur removal and polyaromatics. Higher hydrogen partial pressures are required to achieve diesel specifications and maximize catalyst life in a conventional treater.

The next slide shows some typical operating pressures on straight-run and cracked stocks, as well as typical hydrogen uptake. It is always important when you talk about hydrogen partial pressure that you ground yourself in what the meaning of hydrogen availability: the amount of hydrogen gas actually in the bed, which is makeup, recycle, and quench divided by the chemical hydrogen consumption.

The next slide shows some average ranges of hydrogen availability: the ratio on different feedstocks from straight run through cracked feedstocks to blends of the different ones, basically ranging anywhere from 2:1 up to 5:1 or greater on the more difficult feedstocks. So how do you get to higher hydrogen availability in an existing unit? Certainly, by increasing the purity of your makeup hydrogen, makeup gas rate, and purge rates. Then, if you have the availability, increase the hydrogen recycle rate. Higher pressures may be required. Of course, combinations of different catalyst systems (such as nickel molybdenum and cobalt molybdenum), at the higher pressures to remove nitrogen, will most likely be needed as they inhibit the refractory sulfur removal.

As you get farther along on whether or not to revamp, you will want to consider additional new reactors, new recycle compressors, and makeup gas compressors. We have a typical conventional unit revamp where you put in an additional reactor upstream of the older reactor. It generally involves an additional hydrogen recycle compressor and related treating of H2S before the recycle compressor.
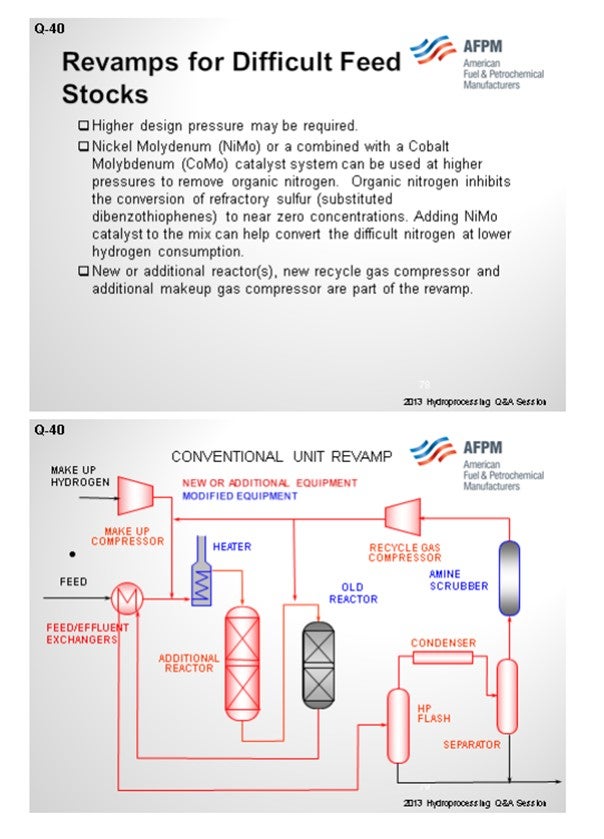
An alternative revamp scenario is using liquid hydrocarbon processing, IsoTherming®, which can provide some ways of getting to this in, hopefully, a more cost-effective revamp. The concept is that you would add a standalone reactor and a heater upstream of the existing trickle bed reactor. You can take out some of the simpler reactions and some of the harder sulfurs in a liquid phase reactor and not have to affect the recycle gas compressor or recycle gas treaters. Many of the studies we have done show this to be an economically viable revamp alternative.
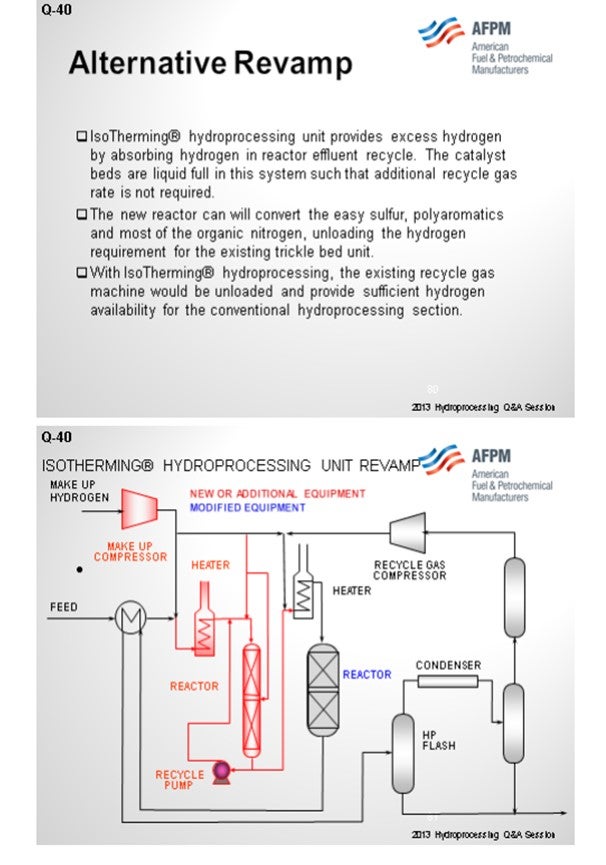
WATKINS [Advanced Refining Technologies (ART)]
In most units, hydrogen partial pressure has a moderate impact for temperature on your weighted average bed temperature. ULSD definitely has a large impact, so we will want to make sure that partial pressure is fairly high. I know that is not necessarily a quantitative answer. But as you said, it also has an impact on HDN. So, if you are allowing your partial pressures to drop, then it will have an impact on HDN, which will ultimately impact pre-ULSD; and also, for aromatics and PNA saturation, if you are using that for anything, in terms of volume swell or meeting targets for cetane and things like that.
It will also have an impact on fouling. If you are starting to run out of hydrogen and your partial pressure begins to drop, your fouling rate will go up. How low do you want that to go? The other question is: How short would you like your cycle to be? A rule of thumb is that cycle length is proportional to hydrogen to partial pressure, so it is one over that to a power. The range is generally two to three. You can see it has a decent impact, which is determined by reactor pressure at the inlets, total pressure, makeup hydrogen purity, and recycled gas rate. As Glenn asked, “How much can you bleed, and what is the degree of feed vaporization?” If you have a unit that was fairly heavy on the frontend and now you decide to feed co-processed naphtha in your ULSD unit, expect your hydrogen partial pressure to drop; because you have now created quite a bit more gas in your unit, and it will possibly have an impact on your ULSD.

The information on this slide gives you a feel for what the impact may be. First, we try to hold imminent pressure constant, and then we vary makeup purity. If you have a unit that starts at the bottom of the slide with 80% purity on your makeup, you can see that it has a fairly low outlet hydrogen pressure for this unit; so maybe you will expect a two-year cycle. If you can actually get up to using fairly high purity hydrogen, like 95 to 100%, you can see quite a big impact on your process, which might gain you 12 more months. So that might be important. The reason why it is going to do that is that it will give you a much lower start-of-run temperature. So that will extend your cycle length as much lowering your deactivation rate.
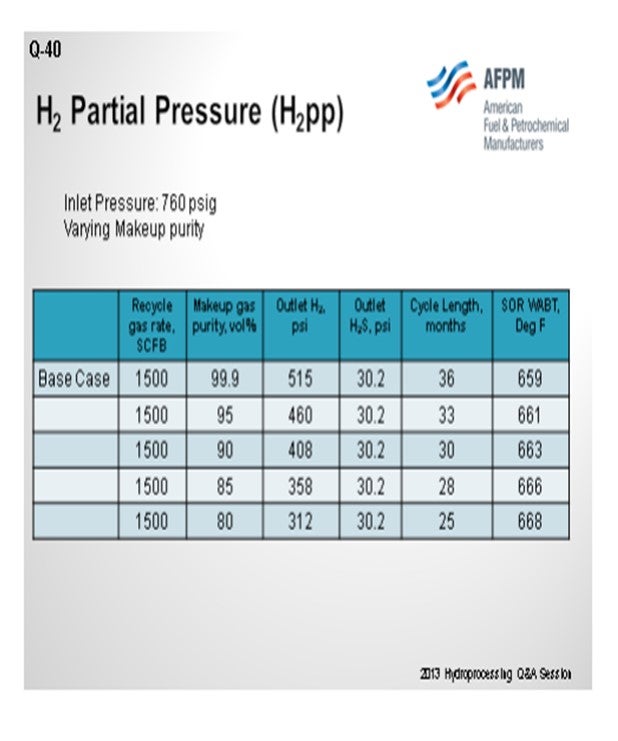
The next chart demonstrates another way to look at that same situation. If you work down the chart, you see that a unit that needs reduction of its recycle rate because of issues with hydraulics going through it. Every time you decrease that recycle rate; you will actually start killing off cycle length. So, you will have to raise the temperature in order to compensate for reduced hydrogen partial pressure, resulting in a higher fouling rate. So, in this case, going from 800 to 1500 scfb (in other words, doubling your recycled gas) will, again, give you your biggest bang for the buck. For this example, your cycle will potentially be 12 to 18 months. There is no true good answer of what is the lowest. It is really what you are willing to sacrifice, in terms of cycle.
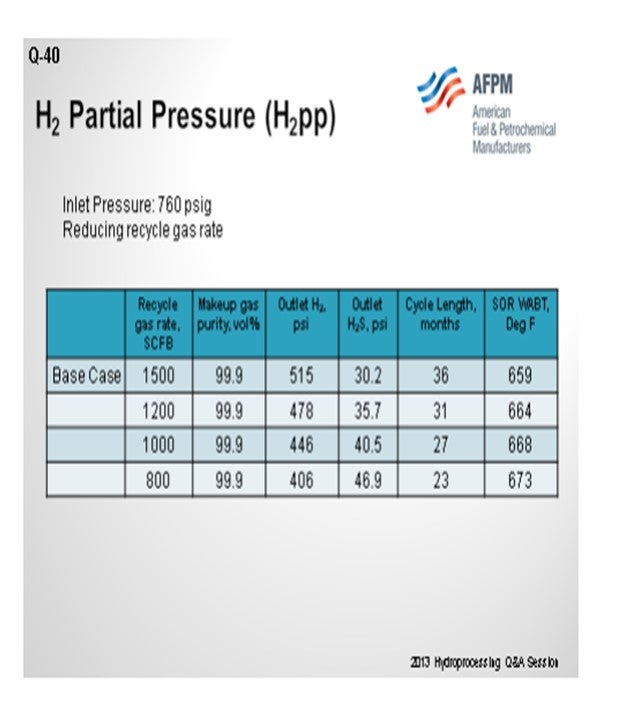
SIVADASAN (UOP LLC, A Honeywell Company)
The hydrogen partial pressure is mainly determined by the total pressure of the reactor system, makeup gas, recycled gas, hydrogen purity, and gas-to-oil ratio. All of these factors have a big effect on the cycle length of the unit. Recommending a minimum hydrogen partial pressure is difficult because it must depend on how much the refinery is going to forego on the cycle length.

MUKESH PATEL (Reliance Industries Ltd.)
What is the revamp scheme that was mentioned? Has anyone has gone for the parallel reactors to reduce that ∆P, split the feed, and then treat the feed?
R.E. “ED” PALMER (Wood Group Mustang, Inc.)
Yes, that has been done. There are some issues, as far as distribution, because you have two-phased flows. Ideally, you like to split the flow between two sets of combined feed exchangers, and maybe split the heater in two to go to two parallel passes or then to the heater; but that has been done.
JEFF JOHNS (Chevron Products Company)
You have all suggested that the choice of hydrogen partial pressure tends to be more of an economic one. Brian said that fouling was proportional to the inverse of the second or third power of the partial pressure ratio. I am interested in whether you have observed that the fouling can fall off a cliff if the partial pressure gets seriously way too low. Could you suggest a methodology on how you might determine that before you fall off that cliff?
SHARPE (Flint Hills Resources, LP)
I keep asking catalyst vendors that question, and they say, “It is complicated.”
JEFF JOHNS (Chevron Products Company)
That is why we are asking you.
SHARPE (Flint Hills Resources, LP)
I am working on it, but I have not yet figured it out. It is one of my current projects.
ALAN KERN (APC Performance, LLC)
I have a very similar question, but maybe once you are above the cliff. The hydrocracker I worked on for several years was maintained with one gas oil ratio for many different feeds. As long as they were above it, the wisdom was that coking would be okay. But based on the statements you have all made, it sounds like there is actually the possibility of a lot of optimization potential that can be seen when looking more closely at that gas oil ratio, depending on what you are trying to monitor, including your feed and cycle length. Would you agree? I would appreciate your feedback.
SIVADASAN (UOP LLC, A Honeywell Company)
I will agree with that rationale because they would assume there are some excess hydrogen numbers that we generally follow, like four or five times the hydrogen consumption. So, if you have more than that, you can optimize your gas oil ratio.
WATKINS [Advanced Refining Technologies (ART)]
I agree with that also. Depending on your gas oil hydrotreater, if it is a really high-pressure unit and you already have 90% hydrogen and a fairly high partial pressure, then maybe going to 95% hydrogen will not give you the economic return. Perhaps it will cost more to do that. Certainly on low-pressure units, such as low-pressure ULSD units, the ability to add more hydrogen will give you incremental benefits, in terms of cycle. That is really where it plays into effect. But usually for your high horsepower units, if you have already met a minimum requirement, adding more hydrogen will not give you any more performance.
ALAN KERN (APC Performance, LLC)
What I am hearing is there is not really a cliff on the high end. There is almost no point at which you have too much excess hydrogen. If it were more, it would not help your cycle length.
WATKINS [Advanced Refining Technologies (ART)]
Yes. On the high end, you may not get any benefit from adding more hydrogen. If you are already at four or five times what you think you will need, then adding six is just extra money that may not result in any benefit. If it is a low-pressure unit, you could probably get some benefit from that. I do not know your specifics.
VICHAILAK (Marathon Petroleum Corporation)
The concern of adding more gas is also with ∆P. If you have too much gas and then ∆P goes too high, that may not be good either. We only recommend a minimum hydrogen availability of 4:1.
GLENN LIOLIOS (DuPont Clean Technologies)
Treating diesel with higher endpoint, cracked feedstocks, or pre-treated feeds can be challenging due to the increased concentrations of refractory sulfur species, organic nitrogen species, polyaromatics, and contaminants such as silica and metals. Refiners generally recognize the need for increased hydrogen partial pressure with difficult feedstocks both to achieve the desired diesel product specifications and maximize catalyst life.
A distillate hydrotreater processing primarily straight-run diesel (SRD) from the crude unit might operate at 600 to 750 psi with a hydrogen recirculation rate of 1000 to 2000 scfb. Comparatively, a unit processing a feedstock with 50% cracked stocks (such as light coker gas oil, light cycle oil, or hydrocracked diesel product) could operate at pressure between at 900 and 1300 psi with a hydrogen recirculation rate of 2500 to 3500 scfb.
Refiners often reference hydrogen requirement as the ratio of hydrogen supply to hydrogen consumed. Hydrogen supply is the sum of the makeup gas, the recycle gas, and the quench gas for each bed, whereas hydrogen consumed is the chemical hydrogen consumption in that bed. When processing feed blends without cracked stocks, a typical target ratio is around 2/1 to 3/1. For high levels of cracked feedstocks, this ratio is increased to a typical 4/1 or higher. This ratio is commonly termed as the hydrogen availability. The required hydrogen availability is very dependent upon the feedstock and the product specifications. For ULSD applications, the minimum ratios are increased to 4.0 and 5.0 or greater for straight-run diesel and mixed feeds with cracked stocks.
Sometimes it is possible to increase the hydrogen availability for existing units – either through using higher purity makeup hydrogen, increasing the makeup gas rate, or increasing the recycle hydrogen rate, all of which result in higher operating costs.
For more severe feedstocks, a unit revamp at higher pressure may be required. A unit revamp could require additional reactor capacity, increased recycle gas compressor head and flow rates, and a new high-pressure amine scrubber. Higher design pressures allow the use of catalyst systems that are more suited to a reaction pathway for removing difficult sulfur species. Often organic nitrogen covers the catalyst sites and has to be removed before the difficult sulfur species can be converted to very low levels.
Nickel molybdenum (NiMo) catalyst typically performs better than cobalt molybdenum (CoMo) for removing organic nitrogen at 1000+ psi. (The use of NiMo catalyst also requires higher hydrogen partial pressure for stability to achieve an acceptable life.) Sometimes the catalyst types are loaded to target certain reactions. After the typical guard/grading catalysts, NiMo is used to reduce the nitrogen to near zero levels. CoMo is then used to convert the difficult substituted sulfur species in subsequent catalyst beds.
Generally, revamping a trickle bed unit involves adding a new trickle bed reactor, a new recycle gas machine, increased hydrogen makeup rates, etc. (See Figure 1).
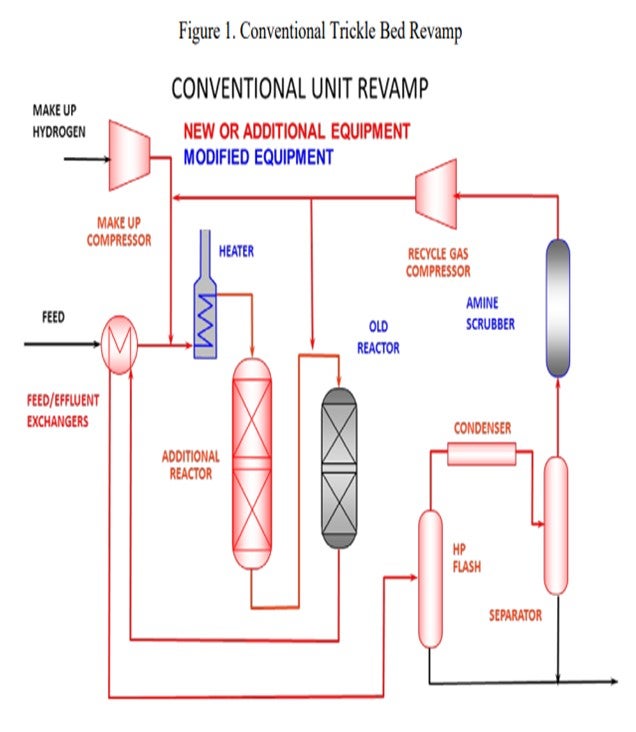
However, there is an alternative. A high-pressure IsoTherming® hydroprocessing reactor licensed by DuPont™ Sustainable Solutions can be installed and does not require a new recycle gas compressor (Figure 2). The IsoTherming® hydrotreating process is a liquid full catalyst system that recycles hydrotreated reactor effluent with absorbed hydrogen. The recycled oil delivers dissolved excess hydrogen to the reactor system by pumping reactor effluent that has been saturated with hydrogen. Hydrogen availability for the IsoTherming® hydroprocessing reactor is determined by the liquid recycle rate, hydrogen solubility in the oil, and the number of beds in the reactor. Makeup hydrogen is injected with the feed and mixed with the effluent from each bed to replenish the hydrogen consumed in the previous bed. This way the faster reactions (i.e., easy sulfur, olefins, polyaromatics, and most of the nitrogen) can be converted in the IsoTherming® hydroprocessing reactor. Adding the IsoTherming® hydroprocessing reactor provides sufficient hydrogen availability and undertakes the additional hydrogen consumption required due to an anticipated change in feedstock. The increased hydrogen consumption is completed in the IsoTherming® hydroprocessing reactor such that the existing trickle bed reactor does not require additional hydrogen recycle rate. This allows the conventional trickle bed reactor to operate at conditions similar to the hydrogen partial pressure enjoyed with the less difficult feedstock. As a result, the reactor product meets the distillate specification, and the reaction system maintains a similar catalyst deactivation rate. Hence, additional hydrogen availability may not be required such that the existing recycle gas compressor has sufficient capacity.
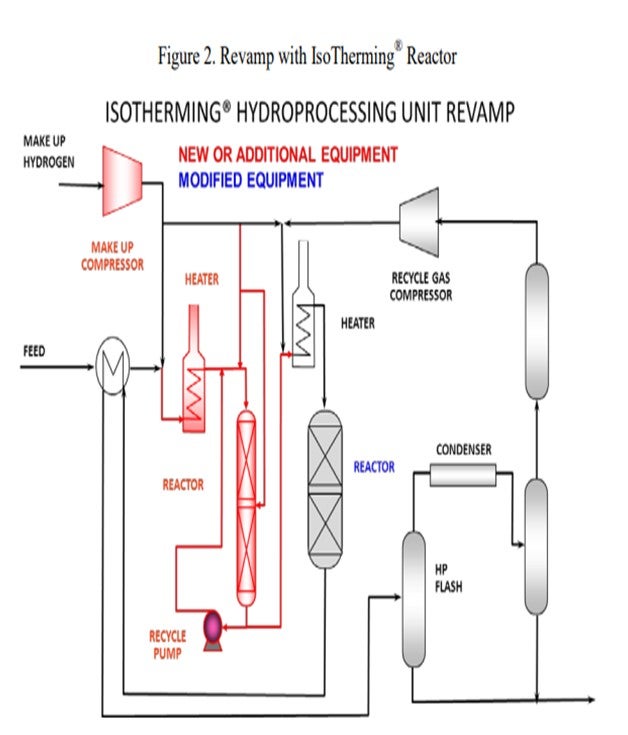
BRIAN WATKINS [Advanced Refining Technologies (ART)]
The question of “What is the minimum allowable H2 partial pressure for a hydrotreater?” has been asked frequently. The general answer is: It depends on how much the refiner is willing to sacrifice in cycle length. As background for this discussion, H2 partial pressure is set by the following parameters:
• Reactor design pressure,
• Makeup hydrogen purity,
• Recycle gas rate and purity,
• Bleed rate,
• Hydrogen consumption, and
• Degree of feed vaporization.
Typically, the reactor is operated at a constant inlet pressure near the design value and with a relatively stable feed quality. The hydrogen consumption and degree of feed vaporization are also essentially constant. Consequently, the variable parameters affecting H2 partial pressure are makeup hydrogen purity, recycle gas rate and purity, and bleed rate. Of these, the independent variables are makeup hydrogen purity and bleed rate which set the recycle purity, and the recycle gas rate. Since many hydrotreaters have amine scrubbers to remove H2S from the recycle gas, a bleed is not necessary and H2 partial pressure is a function of makeup purity and recycle gas rate.
The H2 partial pressure varies throughout the reactor with the highest partial pressure at the inlet and the lowest at the outlet. Since the operating conditions are the most severe at the outlet and strongly influence catalyst activity and stability, we will look at the impact of makeup purity and recycle gas rate on the H2 partial pressure at the reactor outlet.
The following tables show the effect of these parameters on unit performance for a diesel hydrotreater. The base case is:
Inlet pressure:
760 psig (pounds per square inch gauge)
Makeup gas purity:
99.9 vol% (volume percent) Recycle gas rate: 1500 scfb
Table 1 shows the effect of reducing makeup gas purity on reactor outlet conditions, cycle length, and start-of-run (SOR) weighted average bed temperature (WABT).
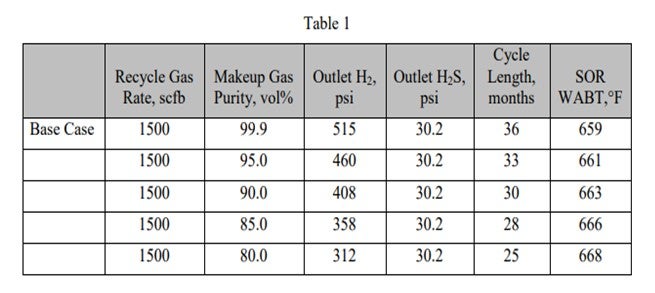
As the makeup gas purity drops, so does the outlet H2 partial pressure. This leads to reduced cycle length and higher required SOR WABT. The impact of reducing makeup gas purity is essentially the same as reducing inlet pressure. Note that as long as the recycle gas rate remains constant, even though the H2 purity is dropping, the outlet H2S partial pressure is constant. In Table 2, the impact of reducing the recycle gas rate is illustrated. Here the effect is more severe because as the outlet H2 partial pressure drops, the outlet H2S partial pressure increases. These further decreases catalyst activity and cycle length. The effect of the outlet H2S partial pressure can be isolated by comparing the 90% makeup gas purity case with the 800 scfb recycle gas rate case. The outlet H2 partial pressure is similar, 408 psi versus 406 psi; however, the cycle length is seven months shorter, and the required WABT is 10°F higher.
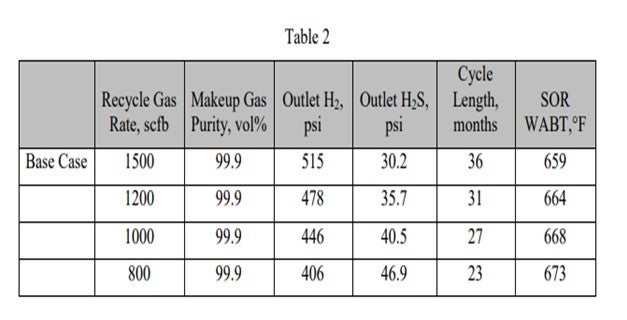
RAJESH SIVADSAN (UOP LLC, A Honeywell Company)
Hydrogen partial pressure has a major impact on cycle length from a catalyst activity standpoint. Cycle length is determined by the catalyst deactivation rate at the design space velocity and hydrogen partial pressure. Increasing hydrogen partial pressure corresponding to reactor inlet pressure decreases the catalyst deactivation rate; and hence, the longer predicted cycle length for a fixed quantity of catalyst. H2 partial pressure is mainly determined by the:
• total pressure,
• H2 purity/recycle/makeup purity, and
• gas/oil ratio.
The operating pressure is probably the most important variable. It directly affects the H2 partial pressure; therefore, the cycle length. Higher pressure drives reactions, especially HDN.
A higher treat gas rate can be used to increase the hydrogen partial pressure, but this is usually limited because of the associated increase in the reactor loop pressure drop and the corresponding maximum operating pressure of the various system components.
Hydrogen purity of makeup gas also impacts the hydrogen partial pressure. Lower purity makeup hydrogen requires higher hydrogen circulation rates to maintain the target hydrogen partial pressure and may even require a purge stream from the cold separator.
It is, therefore, difficult to recommend a guideline for the minimum hydrogen partial pressure as it depends on how much the refinery is willing to forgo on the cycle length.
SERGIO ROBLEDO (Haldor Topsøe, Inc.)
There is no practical minimum hydrogen partial pressure for specific feeds, per se. Temperature, hydrogen partial pressure, and LHSV (liquid hourly space velocity) are the three most important variables that will dictate whether specific product specifications can be met for a given feed. Therefore, the minimum hydrogen partial pressure for a given feedstock is also very dependent on the unit design. If a unit has a low hydrogen partial pressure and also a low LHSV, then it will have a very good chance at removing more difficult species in the cracked or high endpoint feeds. Besides, the unit conditions the activity of the installed catalyst is also an important factor as to whether a specific feed can be hydrotreated to meet the desired product properties. There are plenty of ULSD units loaded with Topsøe’s BRIM™ catalysts that treat cracked stocks at low hydrogen partial pressures of less than 500 psi and a number of units with an inlet hydrogen partial pressure of 200 to 300 psig.
Lower hydrogen partial pressures when treating more difficult streams will mean that a higher reactor temperature will be required to drive the reaction. Topsøe’s BRIM™ catalysts have proven to offer unmatched stability at these low hydrogen partial pressures. Also, cracked feeds and/or higher boiling range material also introduces more coke precursors which will increase the deactivation rate. This is especially significant at lower hydrogen partial pressures. Therefore, the unit will most likely be able to hydrotreat the feedstock, but this will come at a cost of cycle length.
This might still be economically attractive, as the refiner should not necessarily run his unit to meet maximum cycle length but to ensure the highest profitability for the refinery. Of course, if the hydrotreater is tied to another more profitable unit’s turnaround cycle, then meeting the cycle length is important and processing a harder stream might not be viable. It is true that as the feed is being treated deeper into the barrel, more hydrogen partial pressure will typically be required. Below are some general guidelines for operating units based on the service they are in: