

Question 28: The Clean Air Act required refineries to develop and implement a Leak Detection and Repair (LDAR) program to control fugitive emissions. What is the current status of this implementation and who is responsible for it in a typical refinery management structure: production, maintenance or EHS?
Greg Harbison (Marathon Petroleum)
Background/Regulatory Requirements:
Since the inception of the Clean Air Act of 1955 and multiple amendments through 1990, Leak Detection and Repair or LDAR regulations have been a part of air pollution control. Today’s LDAR programs are governed by Federal and State regulations and agreed orders (consent decrees) that provide the control of fugitive emission leaks from process equipment by requiring equipment inspections and leaking equipment repair. As such, the specific requirements can vary company to company or even between refineries operating in different states within the same company. Marathon complies with these regulations.
Equipment Inspections
Components that are LDAR applicable can vary by type and inspection or monitoring frequency. Generally, LDAR components consist of valves, pumps and compressors that are monitored on a quarterly basis. Monitoring requirements can be more stringent for units built or modified post November 2006 and can apply to flanges, connectors, fittings, hatches, and agitators (to name a
few). Process stream speciation determines the applicable regulatory requirements for streams. The typical streams requiring the most rigorous application of LDAR regulations include:
1. gas/vapor streams that are typically > 10% ethane and heavier,
2. light liquid streams are typically heavy naphtha or kerosene depending on specific stream properties, and
3. process streams containing greater than 5% hazardous air pollutants (benzene, methanol, toluene, etc.)
These monitoring requirements can be more or less frequent and have different leak definitions based on different applicable regulations. A leak definition is the threshold in parts per million that a component must reach to be considered leaking. LDAR monitoring is outlined in EPA Method 21, which states that a toxic vapor analyzer (TVA) must be used to assess total volatile organic compound (VOC) leaks from LDAR components. As LDAR regulations become stricter, the leak definitions are increasingly being lowered. With every change in regulation, the LDAR program becomes more challenging to manage since most facilities are required to stay below a facility wide leak percentage for leaking equipment (typically 2%). Thus, a rigorous and well-structured leak repair and maintenance portion of the LDAR program is vital to minimize emissions and maintain compliance.
Program Oversight
A practical LDAR program encompasses multiple people spread across many different job functions. Overall, it is our experience that a successful LDAR program can be successfully managed if several critical items are in place. These include dedicated personnel, a robust software database, good overall management system, well defined roles and responsibilities, and a comprehensive auditing system. At our refineries, it is typically the responsibility of the facility Environmental LDAR Coordinator (HES Professional) to manage and oversee all aspects of the LDAR program. We also use a contract company to conduct the emissions monitoring, and another contract company to make the initial leak repairs on valves (typically injection of a sealant into the valve packing area). Other LDAR applicable components such as motor operated valves (MOV’s), control valves, pumps and compressors are repaired when leaking by qualified individuals within the facility Maintenance Department. The requirements for completing the repairs are often sensitive to equipment and process functionality.
The LDAR Coordinator should have daily communication with the LDAR Monitoring Contractor to go over every open leak Work Order. This information is reviewed and an updated list of all leaks within the facility is given to the Contractor and facility Maintenance Department every day.
Overall, the regulations are complex and can generate an overwhelming amount of information based on the size of the facility and how many leaks are found above the leak definition. A large refinery could have upwards of 70,000 LDAR components governed by state and federal regulations as well as additional requirements from agreed orders. It is imperative to have a functional LDAR database that manages this information. The database should be capable of scheduling all monitoring and repair dates based on applicable regulations for the facility. The progress of the monitoring schedule needs to be easily accessible for all parties involved.
Question 85: What is the typical range that you employ for iron content on FCC equilibrium catalyst? What methods are available to determine how iron is accumulated on the catalyst surface? How does the distribution of iron on the catalyst surface impact the FCC operation, yield structure and emissions?
Jeff Lewis (BASF)
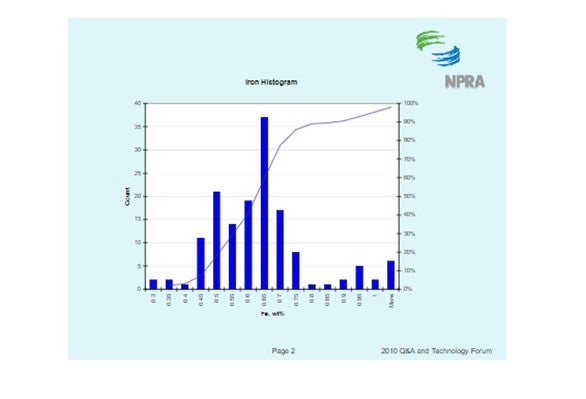
The histogram below shows the distribution of iron content for all ecat samples BASF receives. It should be noted that fresh catalyst has an iron content of about 0.55 wt%. The histogram shows that the median ecat iron concentration is approximately 0.62 wt%. This suggests the median contaminant iron level on ecat is 0.07 wt%.
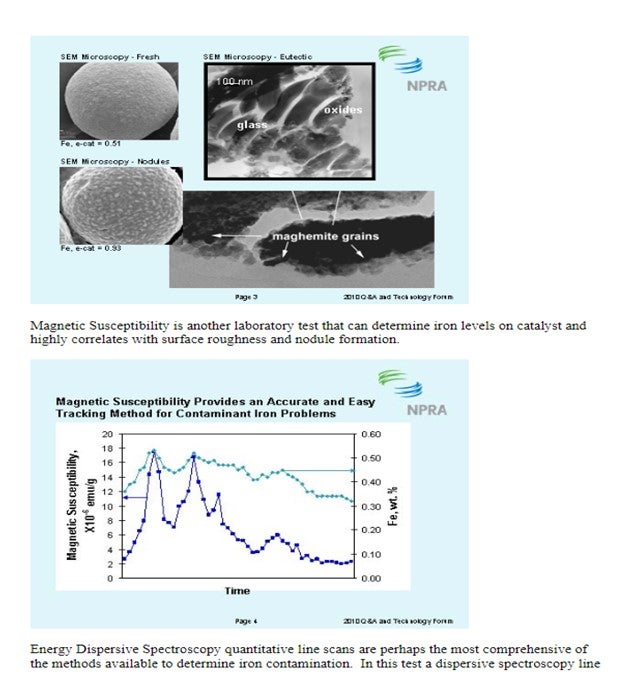
There are several methods available to quantify iron contamination on catalyst. Scanning Electron Microscopy (SEM) pictures are a valuable means to qualitatively assess iron laydown morphology on the catalyst particle. The three images below show varying degrees of iron contamination on a catalyst particle. The first picture shows a fresh catalyst particle that is free of contaminant iron on its surface. The second picture shows a catalyst particle with a significant concentration of iron nodulation on the catalyst surface. The third picture shows a low boiling eutectic formed in the presence of an alkali metal like Ca or Na and is the severest form of iron poisoning.

David Hunt (Grace Davison)
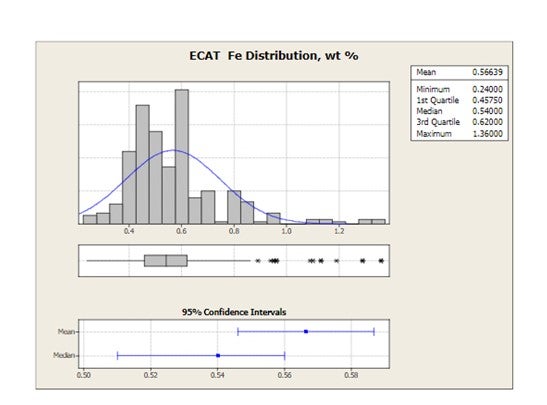
Grace receives E-cat samples for most of the FCC units operating worldwide. The figure below shows the distribution of average equilibrium catalyst Fe levels for 2010 for all FCC units that have provided E-cat samples to Grace. Mean Fe levels are 0.57 wt% and the highest Fe level in one unit is 1.36 wt%.
Iron can be detrimental to the unit in many ways including bottoms conversion, catalyst circulation stability and SOx emissions.
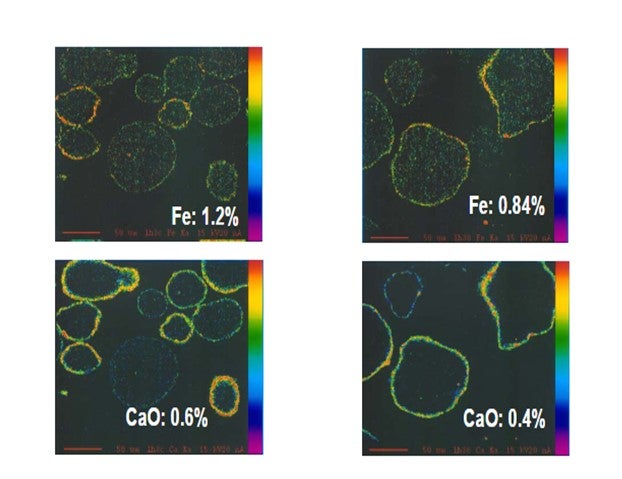
Yaluris (1) showed using an Electron Probe Micro-Analysis (EMPA) technique that iron from organic iron sources is primarily a catalyst surface contaminant. Yaluris also used scanning electron microscopy and optical microscopy techniques to confirm Fe is a surface contaminant. The figure below is an EMPA image of an FCC catalyst particle cross section. Warmer colors on the surface of the particle confirm that Fe and CaO are primarily surface contaminants. EMPA Image of Two FCC Catalyst Particles
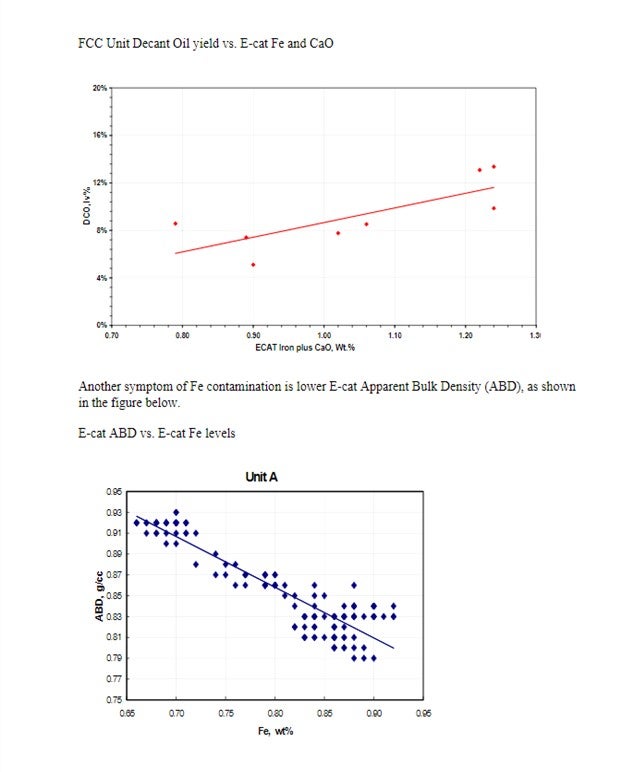
Yaluris (1) discussed how Fe contamination can lead to pore closure and nodule formation. The presence of Na and CaO can act as fluxing agents, aggravating the effect of Fe. The figure below shows Decant Oil or Main Fractionator bottoms yield vs. E-cat Fe plus CaO levels. Decant Oil increases at the higher contaminant levels due to the damaged catalyst pore structure.

Question 13: Severe fouling of diesel and gas oil hydrotreating preheat exchangers has been a growing problem. In your experience, what are the causes and how can these be prevented? Have you tried antifoulant injection in this service?
Dan Webb (Western Refining)
Fouling of the heat exchanger train is sometimes a problem particularly when processing cracked feed stocks. The fouling is often caused by polymer like compounds (gums) that form when petroleum distillates come in contact with air. When heated olefinic compounds react with absorbed oxygen to form gums that deposit in the preheat train.Iron scale and other particulates in the feed often adhere to these gums to produce severe fouling that restricts unit capacity and accelerates heat exchanger corrosion rates. Typically, every effort is made to avoid air ingress into any of the unit feed stocks. Fouling precursors may also be present in straight run feed stocks in the form of certain chemical contaminants that may be present in the crude or inadvertently introduced in an upstream process unit. Some precursors such as amines, carboxylic acids, and carbonyls form gums without air ingress into the feed. Antifoulants have been used successfully to mitigate fouling caused by these compounds in addition to mitigated fouling caused by oxygen contaminate cracked feed stocks.
Michael Chuba (Sunoco)
Typically distillate hydrotreaters exchanger fouling has been associated with cracked stocks that contain olefinic material and trace amounts of O2 coming in with the feed from tankage. In addition to oxygen-initiated polymerization, other impurities can lead to free radical formations that can promote polymerization reactions. These impurities include certain nitrogen and sulfur compounds well as some metal ions including iron, calcium, and magnesium.
In addition to free radial polymerization, condensation polymerization reactions can also result in fouling. In this route, two radicals can react to form a larger molecule. The new compound can continue to react and grow until it precipitates out of solution forming deposits.
What I would like to present here is an example of fouling we had on one of our units and how we have significantly reduce fouling via a simple jump over line.
Prior to conversion of this unit to ULSD the unit processed a mix of virgin and cracked distillate stocks. Historically this unit had exchanger fouling that was attributed to the presence of the cracked stocks. When the unit was converted to ULSD the cracked stocks were removed. The resulting feed was a 50:50 mix of direct rundown material from the crude unit and tankage. As a result of this change in operation it was anticipated that the fouling rate would decrease, however, during actual operation the fouling rate actually increased.
An initial program to address the problem included detailed analysis of the various feed stream followed by a targeted antifoulant chemical injection program. Results were somewhat effective but still left significant room for improvement. Continued investigation into the problem targeted O2 contamination coming from the material coming from tankage. The intermediate distillate tanks are cone roof design which would be relatively costly to convert to blanketed tanks. As a first step it was decided to install a jump over from the tank inlet line directly to the suction of the tanks’ transfer pumps. With this simple connection the average volume of material actually drawn from the tanks dropped dramatically.
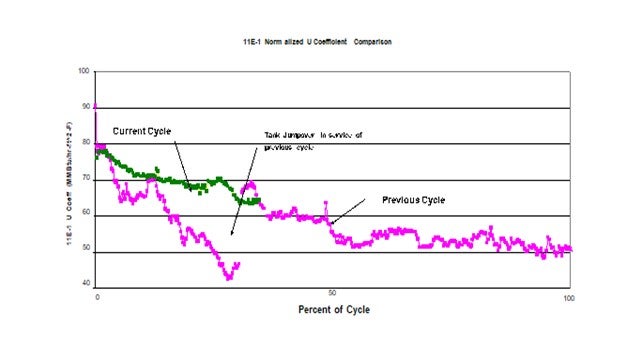
This plot shows the impact on the heat transfer coefficient of the feed effluent exchanger as a result of this simple jump-over. The pink plot represents the previous cycle. At about ¼ of the cycle the jumpover line was installed. At this point significant fouling had already occurred. The discontinuity in heat transfer coefficient a week or two later was the result of a power failure. It is suspected that the rapid depressurization dislodges some of the fouling material thereby improving the heat transfer when the unit is re-streamed. This same response has been seen in previous emergency shutdowns. The green plot represents the current cycle which started with a clean set of exchangers and operation of the jumpover in service from day 1 of the cycle. As can be seen this simple jumpover has significantly reduced the rate of fouling compared to previous cycles. Since the only change was the potential ingress of O2 from the tank, this project confirmed the impact O2 had fouling.
Gregg McAteer (Nalco Company)
Fouling can be a serious problem in hydro-desulfurization (HDS) units because of their importance in producing fuels that should meet environmental specifications. Fouling can limit a unit's ability to maintain a specific feed rate or meet an extended turnaround date. It can greatly influence product quality as well as energy consumption, and catalyst or equipment life. Stricter limits on sulfur and aromatic content of finished fuels make fouling control even more important today. To achieve today’s limits of 0.05 wt.% for diesel, refiners must increase severity of refining operations, which often worsen fouling. Fouling ultimately necessitates shutdown and extensive maintenance, a costly process, both in terms of maintenance expenditures and lost production. Causes of fouling in diesel and gas oil hydrotreaters are both organic and inorganic in nature. The organic foulants are primarily gums formed as a result of processing cracked material and accelerated if the material is exposed to oxygen at any time. Antioxidants and/or antipolymerants are used to reduce the formation of gums and dispersants are used to keep any gums already formed from growing in size.
In one case an antifoulant program utilizing both an antioxidant and a dispersant was applied to a gas oil hydrotreater that normally fouled enough to require a shut down after an average of 440 days. The antifoulant program started on a fouled system and showed
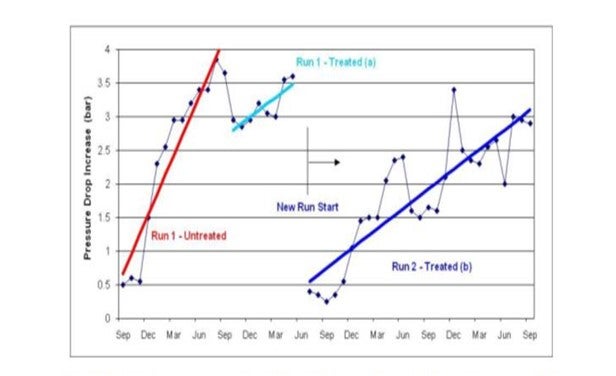
a slight recovery of pressure drop. After a shutdown they started again and achieved a 1300 day run (see graphic below).
“Run 1” is shown in red and light blue. The red trend shows the steep increase in pressure drop during normal operation (without antifoulant program). The light blue trend shows the antifoulant program started, saw a small decrease in pressure drop, and then the unit was brought down for a regeneration. “Run 2” is shown as the dark blue trend and shows a lower fouling rate and longer run length with the antifoulant program. Customer estimated the ROI to be between 400-500%.
Phil Thornthwaite (Nalco Company)
Foulants typically found on the feed side of the preheat exchangers include various gums or polymers, iron sulphide and salts.
The organic fouling due to gums & polymers results from the polymerization of unstable species in the unit feed. The problematic species include olefins (generated in cracking processes), organic acids, mercaptans, ketones, aldehydes, phenols, organo-nitrogen and organo-sulphur compounds. Therefore, in order to determine the risk of organic fouling for a particular feed stream, detailed analysis for the problematic species can be useful guide in evaluating fouling propensity and mitigation strategies.
A typical level for concern for each problematic specie is outlined below:
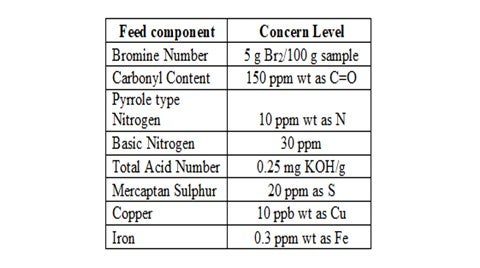
Another key factor to consider is the oxygen content of the feed stream as this can promote the polymerization of various unstable compounds, particularly olefins. Therefore, it is good practice to exclude oxygen from feed storage tanks by ensuring tank seals and vents are in good condition and through the use of a nitrogen blanket. However, this method is ineffective with streams already exposed to oxygen since the nitrogen blanket will have no effect on oxygen reaction products such as aldehydes, peroxides and hydroperoxides.
Inorganic fouling is mainly caused as a result of iron sulphide that can either be carried from upstream units or generated in-situ in the preheat exchanger network. However, the latter is not so common since refiners choose the metallurgy to mitigate against sulphidic corrosion in most cases.
In order to mitigate and control fouling in the preheat train, chemical dispersants and antipolymerants are used. The properly selected dispersant will act upon the organic polymers by keeping them finely dispersed within the feed stream thus minimizing the risk of deposition on the exchanger surfaces. Likewise, dispersants can also prevent deposition of FeS by keeping them dispersed in the feed stream.
Antipolymerants act by disrupting the propagation and chain extending stages of the free radical polymerization reactions and by increasing the rate of termination. This will limit the rate of polymer growth within the preheat system. They will also minimize carbonyl formation which will in turn disrupt condensation polymerization reactions.
The key to monitoring the program effectiveness is through accurate monitoring of the preheat exchanger network. If the fouling results in a limitation of heat transfer efficiency, then a temperature survey of the exchanger network is carried out and this data is entered into a rigorous thermodynamic process model, such as Nalco’s MONITOR® program. This model will then use the plant data to calculate actual and normalized exchanger duties and heat transfer coefficients plus it will calculate the normalized furnace inlet temperature (NFIT). A successful antifoulant program will limit the decay in the NFIT and will generate significant returns for the refiner by improved energy efficiencies and optimized unit operation.
Robert Wade (ART)
We have not had success reducing fouling effects by adding antifoulants. It is our experience that adding antifoulants at best treats the symptom of the problem, and at worst further contributes to localized and downstream fouling. We recommend that the source of the fouling contaminant be identified through analysis and addressed at the source. If this is not possible then we revisit the basic design of the heat exchanger in question and ensure that it is operating in a shear controlled flow regime so that fouling effects are minimized
Question 38: What measurements and criteria do you use to decide when to change your gas and liquid chloride absorber material? How do you determine the selection of absorber material?
John Clower (Chevron)
For both gas and liquid service, Chevron monitors the inlet HCL/Total Chloride and replaces the adsorbent/molecular sieve based on material balance loading of chloride on the adsorber media. Chevron does monitor adsorbent outlet HCL/Total Chlorides, but as a best practice will change the adsorbent material before vendor maximum loading if breakthrough has not occurred. Spent adsorbent will become acidic and pass chloride as organic chloride to the downstream processes. Organic chlorides are difficult to detect by conventional tubes in gas service and will form HCL in downstream processing units.
This performance-based approach is not without problems, e.g., the accuracy of both chloride measurements and represented adsorbent capacity, and therefore requires a trial-and-error
approach.
Represented capacity of any chloride trap material will have been set the vendor to minimize high acidity conditions that lead to organic chloride and polymer (red/green oil) production. Commercially there are four main types of chloride adsorbent material available:
•Alumina
•Modified/Promoted Alumina
•Molecular Sieve
•Metal Oxide
Each of these materials is used in Chevron Refineries and joint ventures. Each adsorbent type will have various properties that can be used in making a decision on application:
•Total chloride capacity (HCL and Organic)
•Reactivity – potential for organic chloride and red/green oil formation
•Interferences (e.g., Sulfur)
•Cost per pound of chloride removed
Also, the design of the vessel used is important (L/D for adequate flow distribution, contact time) and can result in shorten life versus predicted breakthrough. Selection of adsorbent versus service will usually be made on a cost per pound of chloride removed.

Janel Ruby (Johnson Matthey Catalysts)
Chloride can be removed from streams using various products. These chloride guard products can differ in the way they are manufactured and in the way they work in certain applications, so it is important to choose the right one for your needs. The most common products are chemical absorbents or promoted alumina adsorbents. Chemical absorbents remove chlorides by irreversible chemical reaction, meaning that the chloride is chemically bound within the absorbent. Chloride removal in promoted alumina is accomplished mainly by adsorption in which hydrogen chloride is adsorbed onto the alumina surface. Both types of beds are non-regenerable and require change-out at chloride breakthrough.
When determining which product is right for a particular service, it is important to evaluate the operating parameters of the chloride guard bed. The location of the bed in the reforming flow sheet, the operating temperature of the bed, and the normal and maximum inlet chloride levels are important factors to consider when selecting an absorbent type.
Promoted alumina products are available for liquid and gas services. Promoted alumina can work over a range of operating temperatures but chlorides that are adsorbed onto the material may desorb at higher temperatures which will decrease the effectiveness of the product in these regimes. These products also have a lower chloride capacity usually ranging from 12 to 15% wt/wt, and require a high change-out frequency. An area of concern when utilizing promoted alumina materials is the formation of undesirable side products. When the chloride binds to the alumina surface of the guard material, it creates surface acid sites. The acidic surface of the material can catalyze side reactions and lead to the creation of organic chlorides or high-molecular weight hydrocarbons called “green oils.” Green-oils not only foul equipment, but also the guard bed itself, which can cause difficulties in bed discharge (increased purge time) and disposal.
Chemical absorbents are the most favorable option for chloride removal. These products are available for use in liquid and gas services. Chemical absorbents work over a wide range of temperatures. These products have high chloride pick-ups, for example PURASPECJM 2250 is a mixed metal oxide chemical absorbent which can achieve a chloride capacity of 30% wt/wt in non-fouling, gas phase applications. As previously stated, these products remove chloride through an irreversible chemical reaction. The alumina structure present in these types of chemical absorbents acts only as a binder which minimizes the tendency for unwanted side reactions. PURASPECJM 2250 can commonly be employed with the use of just a single guard bed.
There a few other considerations surrounding chloride guard bed materials. It is important to avoid two-phase flow in these beds as this will affect the performance of the chloride guard. Both promoted alumina products and chemical absorbents have a higher pick-up in gas phase, non-fouling and non-wetting applications. In liquid applications, diffusion through the liquid film around the chloride guard particle is the rate limiting step and capacities are generally lower than gas phase duties because of the mass transfer effects. Chemical absorbent products, PURASPECJM 6250 and PURASPECJM 6255 were designed to address this concern. These products have a high capacity and specific pore structure to allow improved removal capacity. They are comprised of the same chemical formulation and micromeritic properties but represent two differing particle sizes; PURASPECJM6255 is manufactured as a smaller sized sphere. The smaller size provides better performance as this minimizes the liquid film through which the HCl must diffuse, reducing the depth of the mass transfer zone and leads to higher average chloride pick at the point of HCl breakthrough.
The presence of HCl or organo-chlorides (RCl) in the exit stream of the chloride guard bed will indicate it is time to change out the material. The life of the guard depends on how the bed(s) is configured and what type of product(s) has been installed. Unless the bed needs to be shut down for inspection or is involved in a larger turnaround plan, chloride breakthrough will be the main reason for a shutdown to replace product. Regular testing for chlorides in the exit stream will help to determine when change out is needed. In applications with longer life cycles (years) testing may only be needed monthly until the bed is getting closer to its expected change-out interval. In applications with shorter life expectancies (months), the frequency of testing should be at least weekly.
Throughout the life of the bed, it is important to measure the HCl and RCl levels both inlet and exit the chloride guard beds. It has been shown that when promoted alumina is used for HCl removal, it catalyses the conversion of HCl to organic chloride species that can then slip from the bed. If the operator is only measuring for HCl then this chloride slip can go undetected until downstream issues occur. Chlorides passing through the bed can cause corrosion of downstream equipment and formation of ammonium chloride that cause fouling and blocking of equipment e.g., stabilizer columns, exchangers and compressors.
Question 10: What causes metal-catalyzed coking (MCC) that obstructs catalyst circulation in CCR reformers? What actions do you take to mitigate MCC formation?
BILL KOSTKA (AXENS NORTH AMERICA)
Metal-catalyzed coke (MCC) formation typically occurs on 3d valence transition metals such as iron and nickel. Under CCR-like conditions of low hydrogen partial pressure (less than about 620 kpa), high temperature (more than about 480 °C) and low or stagnant flow, hydrocarbons can adsorb and completely dissociate on these metals. The resulting adsorbed, dissociated carbon can then dissolve into and change the metal structure. Once a nanosized portion of the metal becomes supersaturated with carbon, carbon begins to precipitate in a tubular crystalline form breaking the carburized-metal fragment away from the parent metal with the carbon nanotube continuing to grow between them. Despite their fragile appearance, these carbon nanotubes are incredibly strong and can readily damage equipment when present in sufficient numbers.
Mitigation of filamentous carbon growth is best achieved by reducing the possibility of hydrocarbon adsorption on the problematic iron surface. Two methods have been used to successfully achieve this goal in CCR reformers: 1) passivation of the metal surface with an adsorbate such as sulfur and 2) use of a more appropriate metallurgy.
Research done by HJ Grabke et al. has shown that very little sulfur, about 0.5 wppm in the naphtha feed, is required to adequately passivate the metallurgy of a CCR reformer. As a result, most CCR reformers are operated with roughly 0.5 wppm sulfur in the feed. Some refiners may rely on incomplete naphtha pretreatment to supply this sulfur, however, addition of a known amount of a sulfur-containing species to the feed ensures adequate passivation on a continuous basis.
Carbon steel is very vulnerable to MCC formation. Alloying carbon steel with increasing amounts of chromium and molybdenum reduces this vulnerability. These two metals tend to migrate to the steel’s surface and greatly dilute iron’s presence there. As a result, there are much fewer Fe-Fe neighbors necessary for hydrocarbon adsorption, dissociation and dissolution into the steel structure. A 9Cr-1Mo alloy steel greatly reduces MCC even at 650 °C. Utilization of this alloy with on-oil sulfur injection virtually eliminates MCC even at 650 °C.
DAVINDER MITTAL (HPCL Mittal Energy)
The catalyst circulation in CCR may be obstructed due to other reasons as well besides metal-catalyzed coking (MCC). However, the metal catalyzed coking presents a serious problem especially in low pressure CCR reforming units.
The processes of metal catalyzed coke formation will cause particles of the heater tube metal to break away from the tube surface. There is also an increased risk immediately following replacement of heater tubes. The coke formed in the furnace tubes may eventually migrate to the reactors and lodge behind the scallops or baskets. These coke deposits can grow until the scallops or baskets are deformed, affecting catalyst circulation, unit performance or even leading to an unplanned shutdown.
The recommended approach is to generally operate the Naphtha Hydro-treating (NHT) unit to remove essentially all of the sulfur in the feed. This will ensure that other contaminants (nitrogen, metals, oxygenates, etc.) are also removed from the feed to the extent achievable by the NHT. Organic sulfur is then added to the CCR reformer unit feed with a chemical injection system pumping in a specific and controlled amount of organic sulfur compound to achieve the target recommended by the licensor. This provides the refiner with independent control of the sulfur in the feed to the unit that can be changed as needed if feed rate or operating conditions change.
Our Continuous Catalytic Regeneration Reformer Unit was commissioned in May’2012. However, within one year of operation, the unit started experiencing several performance issues including restriction of catalyst flow in some of the spider legs of all 04 reactors , higher pressure drop and lower endotherm in reactors (more severe in 2nd Reactor, 60-70% of design value) and lower RON than design.
In view of the above issues, it was decided to shut down CCR during March-April’2014 and inspect reactors. Significant unexpected damage of reactor internals was found.

Picture-1: Huge quantity of coke in annular space between reactor grid and shell
Picture-2(a): Last panel of outside reactor grid found fully bulged with huge coke build up

Picture-2(b): Last panel of outside reactor grid found fully bulged with huge coke build up
Picture-3(a): Shiny coke between and inside scallops leading to bulging and fish mouth cracks

Picture-3(b): Shiny coke between and inside scallops leading to bulging and fish mouth cracks
A joint root cause analysis with Licensor confirmed presence of Fe and carbon graphite (high carbon content) in the coke samples. During cleaning of the scallops, presence of lot of hard shining coke (metallic coke) was observed along with soft coke. It was concluded that coke build up in reactors/scallops/grids may have taken place due to metal catalyzed coking considering problem with DMDS dosing pump during initial year of commissioning as well as due to other reliability issues like frequent trip of recycle gas compressor. The presence of metallic coke in reactors may have acted as nuclei and further catalyzed the coke growth during recycle gas failure.
The heater tube thickness measurements also indicated some loss of thickness indicating metal catalyzed coking in addition to other forms of coke. The level of thickness loss was fortunately not alarming to inhibit future operation.
Based on root cause analysis certain recommendations were made to minimize metallic coking and damage to reactor internals.
Metallic Coke:
Maintain sulfur level 0.3 to 0.5 ppmw on CCR feed to be substantiated by presence of detectable amount of H2S in recycles gas and 100-150 ppmw of ‘S’ on catalyst sample.
Operate Naphtha Hydro-treating (NHT) unit to remove essentially all of the sulfur and other contaminants in the feed. Inject DMDS in CCR feed through dedicated facility to maintain recommended range of sulfur.
No flame sweeping/scattering on the furnace coils.
Maximum Tube Metal Temperature (TMT) to be restricted below 620oC.
Operation of heater burners within the design regime (maximum allowable process absorbed duty per burner: 1.0 Gcal/h).
Perform positive material identification of tube metal to confirm P9 (confirmed).
Other Coke/ catalyst agglomeration due to coke:
Improvement in reliability of recycle gas compressor.
Check for cold spider legs and try to restore catalyst circulation
Check for quality and temperature of net gas from CCR to avoid condensation in reactor spider legs
Maintain recommended coke ( 4 -5 wt%) on spent catalyst
Stress build up in Reactor internals:
Carry out emergency catalyst circulation in case of unplanned trip of the Recycle Gas compressor to relieve the mechanical stress built up due to difference in the thermal expansion coefficient between catalyst and reactors internals.
Question 61: Have you found that you needed to install a methanator upstream of a chlorided catalyst isomerization unit to remove carbon monoxide (CO) from the feed? What is the source of the CO and how much of a difference has the addition of the methanator made to catalyst life? What is the expected payout for the cost of the methanator?
FERNANDEZ (Jacobs Consultancy Group)
This question is quite related to the previous one: the same type of system—the platinum on aluminum-based catalyst. The problem now comes from the oxygen ingress coming in with makeup hydrogen, with the culprit typically being CO and CO2. The problem is slightly different here. The amount of oxygen that you bring is not as high, obviously, because of lower mass flow of hydrogen in comparison to the feed; but it’s much more pernicious. It’s typically a problem when you have nothing and there’s not much you can do in the isomerization unit. So what you really have to look at here is the alternatives to solving the problem. Obviously, methanation is an alternative; but, as always, we would always recommend looking at where the problem is and seeing if you can address it there, rather than spending capital on additional equipment.
It’s important that we look at what may be the sources of CO and CO2 in the refinery hydrogen systems. Everyone traditionally thinks that CO and CO2 are formed in the steam reformer; that is true. Older type hydrogen plants have solvent extractions and a methanation unit to eliminate CO and CO2 in the hydrogen. So there you will see, at most, 10ppm CO plus CO2.
Instead of having this methanation system, modern hydrogen plants have PSA units that are pretty good at removing both CO and CO2; but there’s always a balance between the purity that you get in the PSA unit versus the recovery. So in some PSA units where the units are being pushed either in terms of capacity or on hydrogen recovery, you do have the potential of having CO leakage coming out into your hydrogen system.
A third source—and it’s sometimes forgotten about—is that there is CO coming out with the net gas from reforming units. Some of it might be residue from regeneration operations, but some of the CO actually comes by formation of CO in the unit, particularly in what we would call wet units or units that have a high moisture content in the recycle gas. In those units, CO is formed on a reverse water shift reaction. So this is an area that you have to look at because there will be a source of CO that you may be able to manage.
And lastly, we see there are many refiners today that try to recover hydrogen to the max from all their other services, including FCC units, Coker off gas, and off gas from the hydrotreaters. Those streams tend to be contaminated with all kinds of contaminants, including CO and CO2.
Regarding the solutions to the problem, unfortunately it’s not a very easy problem to track down. The reason is that many refineries have several sources of hydrogen coming together and mixing in a big hydrogen header system. Trying to find out where the CO is coming from is pretty difficult.
I recall that years ago, we were helping a customer that had a Penex unit in which they were having a very high rate of deactivation of the catalyst. This refinery definitely had what we would call a wet reformer. They were injecting large amounts of water and chlorides to try to keep catalyst activity. You definitely knew, and could measure, the amount of CO that was coming in that hydrogen stream.
Unfortunately, that was not the only hydrogen stream. When we tried to correlate the rate of the activation of the catalyst with the amount of CO that was in the hydrogen, the rate of deactivation was higher. One idea was to put in a methanation unit, but you’re only going to resolve part of the problem. And really, the problem is in the bad operation of the reformer. So methanation didn’t seem to be a very good solution there.
We’ve also talked with other refiners that actually had a major problem with CO and CO2, but these guys are talking in the range of 100 to 1000 ppm contaminants of the makeup hydrogen. They know very well what the source is. This refinery recovers hydrogen from FCC, cokers, and hydrotreaters, and they do that through a cryogenic unit. So for them, there really is no alternative. They had to put in a methanation unit and they’re very happy with it. Once they put in that methanation unit, their operation became very stable. They completely eliminated the catalyst deactivation that was being caused by their makeup gas.
In summary: If you cannot prevent or remove it, you’re going to have to methanate it. Methanation is the ultimate solution. It works. There are simple systems. It’s well known how they operate. It’s a relatively low capital cost solution, probably three-quarters to a million-dollar installation for a small hydrogen stream. We would recommend that before anyone commits to the methanation unit, they do two things: One, make sure you know the sources of the CO and confirm that you cannot solve them at the source, which is obviously always the cheapest solution. Secondly, make sure that it is the only problem with contamination in your isomerization unit, because you may be calculating a payback on that methanation unit based on large catalyst deactivation which might not be all caused by the CO.
HAZLE (NPRA)
Jay.
ROSS (Axens)
Yes, thank you, Pedro. We agree with all those points, but I would also like to take a minor issue with the CO contention. We naturally limit all oxygen. That is going to the Isom process because of the potential for H2O formation. This occurs with alcohol as a direct formation of water and with the CO, CO2 in the possibility of methanation. That’s typically limited to about 10 ppm in the hydrogen gas. However, these units do operate at very low temperatures and the conditions in the Isom—in our view—significantly promote methanation. A reversible poison by carbonyl formation with the platinum is probably a more significant concern, and thereby affects the metal activity of aromatic saturation preventing coking, etc. We’ll go to the next slide.
In one particular unit, it was a little different than the unit that Pedro showed. This is a two-reactor traditional chlorinated aluminum Isom unit, but with the lead benzene reactor, because there was a relatively high benzene in the unit. Here, they had a problem on a methanator on the hydrogen plant, so a fairly large concentration of CO passed through the Isom unit as a wave. The benzene reactor experienced a sharp increase in delta P and the exotherm from the saturation of the benzene was temporarily inhibited almost completely as the CO passed through.
And then there were some thermal effects causing the lag to be a little different, as you might expect; but effectively, the benzene was pushed downstream and had to be picked up, in this case, by the lag reactor. Over the course of about four hours, everything came back to normal. That’s not necessarily proof positive that there was no catalyst damage; but in our view, there was limited permanent catalyst damage due to methanation and water formation, and, rather principally in this case with CO, a temporary poison of the platinum metal effects.
HAZLE (NPRA)
Clever.
HATZEL (Tesoro)
Just a quick history of the Mandan methanator: We had a C5/C6 Isom unit installed in early 1981. After multiple catalyst poisoning episodes and catalysts regeneration or replacement, the methanator was installed in 1985 and did seem to work. Looking back to the files, we see that a CO-related poisoning pretty much ceased. That doesn’t mean that we didn’t find other ways to kill the catalyst in the years after that. We did, with alcohols and other things that got into the feed. As late as 1995, they partially bypassed the methanator inadvertently. They went out and found the bypass valve cracked open, but it was too late. Within just a few days, the catalysts had been poisoned. So that has kind of been our experience.
When talking to our Isom experts, I think we give pretty serious consideration, especially with the advent of some of the benzene regulations, to having methanators on future Isom units.
HAZLE (NPRA)
Those are the panel responses. The last question is on isomerization. There is one right here. Questions? Comments? I think I saw a hand over here.
OYEKAN (Marathon Petroleum)
Soni Oyekan, Marathon. I just have questions for the panel. In the case of the example for Mandan: Was the hydrogen recycled or once-through to Isom unit initially? Secondly, for Jay: In terms of the levels of concentration of CO and CO2 that you’ve suggested, I believe the upper limit is 10. We might have temporary relief after the carbonyl is formed initially. Should we worry if it’s above that or basically be satisfied that we’ll have this passivation of this carbonyl? So, there are two questions. One is about the effect of this carbonyl with hydrogen once-through units, and the second one will be max carbonyl and CO and CO2 level that we’ll be looking at in these streams.
HAZLE (NPRA)
Let’s start with Jay.
ROSS (Axens)
Wait until Soni gets his card out there. As with other, we traditionally recommend less than 10 ppm CO plus CO2. And as with all these things, even with the dryers, you recommend as dry as you can, but you recognize that there will, in fact, be some breakthrough and slow degradation. The example that I showed was a rather extreme one, where with the methanator failure, they had 3 or 4% CO. So it made a wonderful lesson and example, but it was perhaps a bit extreme.
HAZLE (NPRA)
Clever, once-through or recycle?
HATZEL (Tesoro)
You’re testing my memory a little bit. We actually shut the Isom unit down in 2000, but I believe that was once-through. The source of the hydrogen unit was from a cyclic reformer.
HAZLE (NPRA)
Other questions?
PROOPS (Solomon Associates)
Kevin Proops with Solomon. I was going to ask Clever where his hydrogen came from, because my experience has been with dedicated hydrogen from a cyclic reformer, not significant deactivation due to CO. Pedro gave a good overview of managing your hydrogen system; but I guess I would suggest that if you’ve got a complex hydro system, especially with hydrogen plants, you should consider taking the lowest CO sweet hydrogen stream direct to the Isom unit rather than trying to deal with any problems after you’ve blended your hydrogen streams together.
Refiners also sometimes have issues with having a high enough pressure hydrogen stream when you don’t really want to end up with another compressor if you don’t have to. So it’s nice if you have the pressure. One of the disadvantages a lot of times in older reformers is their higher pressure, because the yields [tend] to be worse. Maybe one of the advantages is that you can then take the hydrogen stream off of that unit when that unit is available directly to an Isom unit.
HAZLE (NPRA)
Other questions or comments? There would be another one back here.
DETRICK (UOP)
Kurt Detrick, UOP. The example Jay showed us is a good example of what we would expect with a high breakthrough of CO: a severe depression of the platinum activity. And certainly, once you take away the platinum activity, the catalyst does neither benzene saturation nor isomerization. Eventually, though, that effect will go away.
However, our experience in many commercial situations has been that the CO does also act as a permanent poison, probably through a methanation reaction in the reactor leading to water formation. We’ve seen this with low level CO, constant low level CO, and steady deactivation. Once the CO is removed, that deactivation goes away.
There is clear commercial evidence of permanent damage to catalysts due to CO, and with CO2 as well, although there does seem to be a difference. The CO does seem to be a little more potent poison. I think it’s because we’ve seen evidence that CO2 can actually break through the reactor. It doesn’t all get taken up in the reactor. We’ve seen a couple of suggestions of that. The data that we’ve seen is also consistent in that maybe the CO2 isn’t as strongly held by the catalyst or picked up as completely. I think you can pretty much figure that all of the CO is going to go to water. It may not be right on the top of the bed, but it’s going to all lay down on that bed and it’s all going to make water.
And again, Kevin’s commented that historically, the older reformers often had much lower levels of CO. Just the operating conditions led to lower CO levels and those have been very reliable sources of hydrogen for Isom units for years. Some of the newer units are operating at conditions that are going to generate 10 ppm CO—and maybe even a little bit more at times— and those are the ones that are concerning in the current situation.
HAZLE (NPRA)
Kurt.
DETRICK (UOP)
Sorry. Jay.
ROSS (Axens)
I think it’s clearly been reported previously that the CO will methanate more readily than the CO2. I believe it’s the first one to go. And certainly, we recognize that even though we are at relatively or very low temperature comparatively, there is certainly some methanation occurring. It’s just that others, in the past, have tended to treat it as quantitative. I just wanted to make the point that it wasn’t perhaps quantitative.
OYEKAN (Marathon Petroleum)
Soni Oyekan. This is just a question for most of the people in here. Is anyone working with dryers that have mixed absorbance in them that want to help you with moving to CO and others to dry down the water?
HAZLE (NPRA)
Panel? Pedro.
FERNANDEZ (Jacobs Consultancy Group)
One of the ways that people have said dryers in makeup hydrogen service can be made more productive is by adding multiple types of absorbance; but typically you can get something that can remove water and CO2. I don’t think that there is any good experience with absorbance that can effectively remove the CO. I would look for anyone that has done that.
HAZLE (NPRA)
Anyone else on the panel? Clever.
HATZEL (Tesoro)
Again, it’s been a while. I seem to remember, though, that we had some on the liquid feed belt system—not on the hydrogen coming in—and that we had some mixed absorbance on that same unit: sulfur on the top and water on the bottom.
HAZLE
(NPRA) Anyone in the audience? Fred.
HILL
Kurt, on the effect of the CO and CO2 going through the unit, did you see any difference between a once-through and a recycle unit?
DETRICK (UOP)
Kurt Detrick, UOP. I think with the CO2, possibly. Again, to get back with Jay, certainly I agree that the conditions are pretty mild. And with CO2 anyway, quantitative methanation is probably not happening. It doesn’t all get methanated, at least not the first time through. But in a recycle gas unit, a lot of it gets another chance to go back through. So I think you’ll see, with the CO2, that it could be more damaging to a recycle gas unit than it would be to a hydrogen once-through unit. Like I said, we did see some evidence of CO2 coming out of a hydrogen once-through reactor, even the second reactor. We’ve never seen that with CO. It may not be 100% of the CO gets converted to water, but it’s pretty close. So I don’t think there’s going to be much difference there between a recycle gas unit and a hydrogen once-through unit. In theory, that follows, too, because CO is much more easily methanated than CO2.
HAZLE (NPRA)
Other questions? Alright. I owe you a break. Emerson has bought you coffee this morning. It’s waiting for you in the hall. We are going to resume at about 10:40 and we will start with Naphtha Hydrotreating.
Question 43: What are your best practices when shipping ecat, fines, feed, and slurry to suppliers for testing? Please also comment on some best practices for sampling equilibrium catalyst.
TODD HOCHHEISER (Johnson Matthey)
When shipping ecat or fines, an appropriate sample container should be used. Catalyst suppliers will typically provide refiners with sample containers if needed. Catalyst shipping containers should be made of plastic or metal. Glass containers are not recommended due to potential breakage but can be used with appropriate packaging. A screw top lid is preferred over a snap on lid sometimes found on metal containers. Prying opening a snap on lid can result in personnel dust exposure. Catalyst samples should not be shipped in plastic sandwich bags or other containers not designed for catalyst service.
JM has found that metal sample containers with a screw top lid are best when shipping low vapor pressure hydrocarbon samples. A best practice is to place the sample container in a plastic bag containing adsorbent pads. These pads should minimize the chance of hydrocarbons leaking out of the box if the sample container leaks.
For hazardous catalyst and hydrocarbon samples, a GHS complaint label must be placed on the sample container. The safety data sheet must also be included with the shipment. Most catalyst suppliers prefer for a safety data sheet to be included even if the sample isn’t considered hazardous. Other regulations and requirements may apply especially for sample shipments between countries.
Common sense precautions are also recommended. Some examples are shipping only the quantity of sample that is required, packaging in strong boxes, and using labels with high quality adhesive. Our lab has received sample boxes containing multiple ecat samples and multiple labels that are no longer attached to the sample containers. Clearly identifying the date of the ecat samples is critical for unit monitoring.
For any sample that is shipped, it is recommended that a company representative certified under DOT or applicable regulations be involved in the packaging and shipping process. Carriers also have specific requirements for shipping hazardous material.
KEN BRYDEN AND LUIS BOUGRAT (W. R. Grace & Co.)
For all samples, it is important to provide a safety data sheet (SDS) when shipping the sample and to follow appropriate Department of Transportation (DOT) and International Air Transport Association (IATA) rules when packaging and sending the sample. Samples should not be sent by U.S. Mail or any service that transfers to U.S. Mail. Based on our experience receiving and testing thousands of customers Ecat and hydrocarbon samples each year, Grace has the following suggestions on best shipping practices.
Ecat and Fines Samples
For Equilibrium catalyst (Ecat) and fines samples, we have found that for routine testing a 500 mL screw top plastic container is an ideal size. Grace provides complimentary Ecat Express containers for this purpose. Screw-top metal containers are another packaging option for Ecat. Glass containers are unsuitable for Ecat since they tend to break in shipment. Containers with paint can lids are unsuitable since the lids tend to come off during shipment and spill catalyst. Bags are also unsuitable containers since they tend to leak. For any container, do not put any tag, string or wire between the cap and the container lid since they will compromise the seal and cause leaking. For large quantities of Ecat, we have found that five-gallon (or 20 liter) plastic screw top buckets are good containers.
For Ecat and fines samples, proper labeling is important in making sure the desired tests are done and reported. At a minimum, samples should be labeled with the following information:
-
Refinery or company name.
-
Refinery location. For example, city and state.
-
Unit Name: Especially important if there is more than one FCC unit at the refinery location.
-
Sample Date: The date that the sample was collected.
-
Sample ID: (Optional) A sample number or name, for your reference.
-
Sample Type: for example, Ecat, fines, purchased Ecat, non-routine, etc.
As part of the complimentary Ecat Express kits, Grace provides container labels that already have the refinery name and unit written and barcoded on the label.
Feed and Oil Slurry Samples
For routine analytical testing to measure the properties of feed or oil slurry samples, a 16-ounce (or 500 mL) sample size is preferred. In shipping hazardous materials, proper packaging and labeling is essential to ensure compliance with the appropriate regulations. This will prevent fines from the carrier and delays in your shipment. In addition, poorly packaged samples can leak, which results in the sample being compromised and thus unsuitable for analysis. There are many good packaging systems available from suppliers that may be chosen to meet the packaging requirements of IATA and CFR49. Which system to use has to be determined by each individual shipper for their samples. The most common system that we see customers use is a 4GV shipper where the hydrocarbon sample is packaged in a metal can, which is then placed inside a plastic bag with an absorbent sleeve. The entire assembly is then placed in a certified cardboard box. It is important to make sure that the lid is screwed on securely. We occasionally receive leaking samples where the container lid vibrated loose in shipment. In preparing containers, make sure tags and wires from labels are not in the thread area of a cap. A string or wire from a label tag put into the sample container, with the cap sealed over it, will act as a wick. This will always cause leaking. Container types that we have noted problems within the past are a) paint cans- the lids often pop off during shipment, and b) glass bottles- they have a tendency to break during shipment.
As with Ecat samples, labeling of feed and slurry oil samples is important. The container should be labeled with the material identity and the appropriate Global Harmonized System (GHS) hazard symbols. Additionally, the sample should be labeled with the following information:
-
Refinery or company name
-
Refinery location: for example, city and state
-
Unit Name: especially important if there is more than one FCC unit at the location
-
Sample Date: the date that the sample was collected
-
Sample ID: (Optional) a sample number or name, for your reference
-
Sample Type: for example, feed, oil slurry, etc.
Process Ecat Sampling
Routine and representative sampling of the circulating Ecat inventory represents a critical part of FCC performance monitoring and optimization. Samples of the circulating inventory should be collected from a fluidized and accessible section of the unit to enable representative sampling of the catalyst system. From a safety standpoint, regenerated catalyst represents an inherently safer sampling source than spent catalyst due to the lack of entrained hydrocarbons and the lower coke concentration along the surface of the catalyst. However, the process temperatures associated with regenerated catalyst are significantly higher than those of spent catalyst and should be mitigated accordingly.
The regenerated catalyst standpipe represents the most common sampling location due to the continuous catalyst flow and accessibility associated with this standpipe. Although the flowing catalyst is well fluidized within this type of standpipe, it is important to properly fluidize the sampling manifold as well when obtaining a catalyst sample. Plant or instrument air are the most common fluidization media for regenerated catalyst sampling stations, which can also be equipped with steam connections to serve as blast points for line plugging troubleshooting. An air or steam purge into the process should be maintained at all times across the standpipe sampling nozzle to prevent catalyst ingress and nozzle plugging. The fluidization medium should correspond to a reliably dry source to prevent potential catalyst agglomeration issues throughout long-term operation. The sampling outlet nozzle should be purged prior to lining up the sampling line to the process to ensure that the manifold is clear of fouling and to confirm that the sample fluidization medium is available and properly dry. The key considerations and best practices for the Ecat sampling process, among others, are as follow:
-
Field personnel should be equipped with all necessary PPE prior to collecting the Ecat sample. Contact your catalyst vendor if any additional feedback or specific PPE guidelines are required.
-
Any potential impacts on instrument readings or safety interlocks by the Ecat sampling process should be thoroughly identified. Ecat sampling activities should be communicated to the board operators prior to starting the sampling process to help ensure that instrument and safety interlock functions are not compromised while sampling.
-
Ensure that the sampling container or recipient is adequately rated for the normal process temperatures associated with the circulating Ecat inventory. The sample containers used for shipping are not typically rated for these elevated temperatures. Metallic containers are typically required to accommodate Ecat sampling.
-
The sampling valve and the sampling outlet nozzle configuration should, ideally, enable sample collection without exposing field personnel to catalyst and entrained flue gas at the high process temperatures. A remote point where the operator can operate a HIC (Hand Indicate Controller) valve to take the sample in line of sight of the sample station but a safe distance away is practiced by several refiners. The sampling recipient can be attached to a long metallic or high-temperature-resistant handle to help mitigate personnel exposure to high temperatures throughout the sampling procedure.
-
Sufficient sample flow should be established to enable collection of a representative Ecat sample. Insufficient purging of the sampling manifold with the flowing Ecat can lead to non-representative or compromised results due to the presence of stagnant Ecat from previous sampling rounds, or other similar contamination sources. Collection of a slip stream during continuous Ecat flow through the sampling line tends to yield a more representative sample than collecting a vial sample from a drum or (large container) of Ecat sample inventory.
-
Excessive superficial velocities through the sampling manifold should be prevented while sampling to help mitigate potential erosion and attrition issues. Excessive catalyst attrition through the sampling line can lead to false PSD profiles for the circulating catalyst inventory that can prompt unnecessary troubleshooting activities. Adequate velocities through the sampling nozzle also help reduce turbulence and dust as the flowing Ecat reaches the sampling container, thus preserving as much of the fines content present in the circulating inventory as possible.
-
A pint of Ecat sample is usually sufficient volume to accommodate routine lab testing for process monitoring purposes. Excess Ecat sampling volume should be properly handled and discarded via spent catalyst drums or disposal lines routed to the spent catalyst hopper, if available.
-
Ecat samples should be allowed to properly cool before filling the corresponding shipping containers. Windy or wet environments should be avoided for the cooling period to avoid altering the physical properties of the Ecat sample.
The guidelines and best practices previously referenced should be followed when shipping the Ecat sample containers. Board operators, unit engineers and other supporting staff for the FCC complex should visually inspect Ecat samples before the sample is shipped to the catalyst vendor. Visual inspection can help qualitatively gauge the health of the circulating catalyst inventory – especially with respect to coke on regenerated catalyst (CRC), drastic PSD shifts, and/or potential Fe poisoning contamination – well before the corresponding lab results become available.
Question 21: We are observing fouling of our feed/effluent exchangers that has impacted heat transfer and restricted feed. What are potential contributing causes and how can we mitigate?
ROBERT STEINBERG (Motiva Enterprises)
There are many things that can contribute to fouling of feed/effluent exchangers. Fouling can occur on either the feed or product side of the exchangers.
Possibilities sources of fouling on the feed side include:
• Dissolved O2. Oxygen can get into feeds if they come from a tank that is not N2 blanketed, this is especially likely if feeds have been imported from another site via a barge. Oxygen can also be present if a feed come from a vacuum tower with an air leak. Corrosion inhibitors or oxygen scavengers injected into the feed as far upstream as possible may help. The best method to remove oxygen is to add an O2 Stripper on the stream that contains oxyg
en.
• Caustic. Small amounts of caustic that was not water washed can lead to severe fouling.
• Particulates, scale, corrosion particles. FeS scale is often found in refinery streams. If the source is known, corrosion inhibitors may be able to reduce the amount of scale. Good feed filters may be able to remove some of the scale but FeS particulates can be small enough to pass through most feed filters.
• Dirty feed. Cracked feeds, especially coker gasoils, tend to be dirty and have small coke particles. Good filtering is essential. If not done at the upstream unit it needs to be done on the hydroprocessing unit. It is often a good practice to filter both places in case one of the filters is bypassed.
• Salt in Feeds. If crude oil is not properly desalted there can be salts left in heavy feeds. Salts from other sources can also be present at times. A water wash or a desalter can remove salts.
• High temperatures. High skin temperatures tend to increase fouling. High temperatures may be unavoidable when exchanged against reactor effluent, especially in the hotter shells. An exchanger design that increases velocity and promotes turbulence on the feed side will increase the heat transfer coefficient and reduce skin temperatures. Injecting hydrogen upstream of the exchanger will help.
• Low velocities. Lower velocities in the exchanger reduce pressure drop but lead to higher skin temperatures, make it easier for particulates to stick to tube surfaces and increase fouling. Injecting hydrogen upstream of the exchanger will help. Recycling hydrotreated product when the unit is turned down will maintain higher velocities in the exchangers.
• Cracked feeds. Cracked feeds have olefins and sometimes di-olefins which can polymerize and are more prone to fouling. Cracked feeds can be a particularly severe problem if dissolved oxygen is present. A selective hydrogenation unit or reactor can be used to saturate di-olefins at a relatively low temperature upstream of the main reactor before the feed gets hot enough for severe fouling to occur.
• Asphaltene precipitation. This is normally only an issue with resid units. Mixing different feeds, especially a lighter more paraffinic feed with resid, can create incompatible mixtures and cause asphaltene precipitation.
Reactor effluent is normally cleaner than reactor feed. Olefins get saturated and dissolved oxygen gets converted to water in the reactor. The reactor effluent will always have hydrogen which tends to keep velocities high. However, there are some possible sources of fouling on the reactor effluent side:
• Salt precipitation. H2S, NH3 and HCl are normally present. These form ammonium chloride (NH4Cl) and ammonium bisulfide (NH4HS) salts when temperatures fall below the salt formation point. The salt point is dependent on the operating pressure and concentration of H2S, NH3 and HCl. Salt point curves can be found in API Recommended Practice RP-932B Design, Materials, Fabrication, Operation, and Inspection Guidelines for Corrosion Control in Hydroprocessing Reactor Effluent Air Cooler (REAC) Systems. Typical precipitation temperatures are in the 300-400°F range for NH4Cl and around 100°F for NH4HS. In addition to fouling, these salts can be extremely corrosive if water is present. Dry salts are not corrosive but an intermittent water wash may be needed to remove them once fouling occurs.
• Polynuclear aromatics. This is normally only an issue with hydrocrackers, especially the 2nd stage of a two-stage recycle unit. If conversion is too high the PNA concentration can get high enough that they become insoluble in the oil. The lighter cracked products can cause PNA’s to precipitate in exchangers as the effluent cools and more of the naphtha range material condenses.
JOE RYDBERG (CITGO)
In our recent experience, fouling on the “feed side” of the feed/effluent exchangers in Naphtha units is due to corrosion products (Fe) entering with the feed, and processing recycled Naphtha’s particularly from LPG Caustic Disulfide separators. The recycled naphtha’s can have higher levels of Sodium and Salts (likely amine degradation products that build up in the caustic).
Other causes can be contamination of cracked stocks into the virgin stocks system. Exposure to oxygen will cause gum formation. Crude supply sources have unknown diluents. Refineries are now collecting more material from various refinery sources and rerunning as slops, for example introduction of flare gas recovery liquids, reprocessed as slop oil; re-processing/chemical cleaning liquids pumped to slop system.
Use of chemical additives (organic dispersant, antipolymerant, oxygen scavengers) can be used and are used within CITGO to mitigate fouling. Proper tracking of heat exchanger fouling is important and can aid in scheduling cleanings (requiring unit shutdowns) outside of turnarounds, during catalyst change-outs, etc. When dealing with especially challenging feeds and / or extending cycle length goals, installation of spare feed/effluent heat exchangers could be value added approach
Effluent side fouling typically is caused by inadequate water wash, presence of NH4Cl in addition to FeS. HCl can also react to create additional FeS in the presence of H2S.
ERIC LIN (Norton Engineering Consultants, Inc.)
In a hydrocracker with liquid recycle (could be single stage recycle or two-stage recycle), there exists the possibility of HPNA (Heavy Polynuclear Aromatics) buildup at the bottom of the fractionator. Although the overall conversion will decrease, the best solution is to have a dedicated bleed stream out of the unit (FCC is a typical destination) to prevent this buildup. High asphaltenes in the feed are usually a sure sign of HPNA production.
In a residue hydrocracker, the existence of sediment can cause similar fouling in these exchangers. Sediment can typically be mitigated with the use of slurry oil as a cutter (much easier to acquire for units that also have an FCC nearby).
SAM LORDO (Consultant)
Fouling in the circuit ahead of the furnace and furnace can be caused by inorganic solids, or polymerization of feed components (organic fouling). Mitigating fouling from inorganic solids, such as iron sulfide and other corrosion byproducts, sand and silt (from imported feedstocks) is primarily done using feed filters. The pore size is best at 1-5 micron. The filter can be cartridge style, sand filters. Some filter arrangement would have backwash capability.
Fouling downstream of the reactor may include ammonium chloride (NH4Cl). Typically, a well-designed Waterwash is used. The use of salt dispersants are also applicable where Waterwash is feasible
Organic fouling could be from:
• Stream that contain olefinic/diolefinic components which when exposed to elevated temperatures ass found in the hydroprocessing units
• O2 contamination of feed or feed component streams
Mitigation of this source of fouling can be done using an appropriate chemical additive, such as dispersant and/or antiploymerant.
MICHAEL PEDERSEN (Honeywell UOP)
Most hydroprocessing catalysts require a conditioning period at start of run to allow the active sites to stabilize. One aspect of this process is the common industry practice to avoid processing cracked feedstocks during the first few days of operation. Prior to conditioning, fresh catalysts have a high tendency to generate excessive coke when operated with reactive feedstocks or at normal unit operating severity. A short period of mild operating conditions can pay big dividends in overall catalyst cycle performance while high severity operation at start of run can have substantial negative impact on apparent catalyst activity and cycle length. In general, catalysts that are claimed not to require conditioning have been artificially inhibited prior to delivery.
Hydroprocessing catalysts encompass a wide variety of formulations, so a general set of conditioning guidelines is not applicable. For a specific catalyst system, instructions from the supplier should be followed.
SIMERJEET SINGH and RAJESH SIVADASAN (Honeywell UOP)
Fouling of feed/effluent exchangers in hydrotreating units is a common problem leading to throughput losses, increased energy consumption, unit downtime and maintenance expenses for exchanger cleaning. Fouling happens due to changes in feedstock quality, exchanger temperature, fluid velocity, degree of vaporization and exchanger configuration leading to formation of hard carbon deposits (coking), deposition of undesirable polymers (polymerization) and corrosion products.
For Coker Naphtha Hydrotreater:
• Feed quality issues:
Coker naphtha (CN), by the nature of thermal cracking reactions, contains free radicals, which react with diolefins and olefins to form oligomers and polymers. By itself CN presents a fouling problem in a NHT, however when combined with stored SRN there exists the potential for significant fouling. Storage of CN prior to processing can have disastrous results, as the combination of diolefins, free radicals, and oxygen (peroxides) can lead to rapid fouling on the feed side of the combined feed exchanger (CFE), the NHT charge heater, and the NHT reactor. This fouling can be serious enough to cause premature pressure drop increase along with loss of heat transfer due to fouling in a matter of days if not hours. The downtime associated with addressing this fouling costs the refiner time and money.
The highly reactive diolefins in CN are the four carbon and five carbon species, at the front end of the boiling range. Longer chain diolefins tend to be reactive, but less reactive than the short chain diolefins. Simply increasing the initial boiling point of CN (reducing the quantity of light diolefins) may reduce the tendency of CN to cause fouling. When cracked stocks with significant diolefin concentrations are present, it is UOP’s practice to include a diolefin saturation reactor as a first, low temperature reaction stage in a two-stage reactor system. In this reactor, most of the diolefins are saturated. This reactor is located in between CFE shells and its position is selected such that the inlet temperature is in the range of 320-370°F.
• Design considerations:
o Feed tank blanketing
o Design of feed tanks (Fixed/ floating roof)
o Hydrogen Injection to preheat exchangers
o “Over-Sized” exchangers for clean duty
o Exchanger velocities
o Dry Point location
For VGO HDT:
• Feed quality issues:
o Fouling is also experienced in units that run straight run feed only, so it is not just a phenomenon that requires cracked olefinic feeds.
o Fouling from asphaltene precipitation.
• Design considerations:
o Same design considerations as coker naphtha HDT except the dry point location.
o Thermal cracking of feed VGO in feed effluent exchanger can be of main issue if separate feed heating is being used as design feature over combined feed heating.
• Fouling Mitigation Strategies:
Many methods exist for managing fouling. The costs of these methods vary, as does their effectiveness. In order to choose the most effective method for managing fouling, an understanding of the source of foulant precursors should be established. Analytical methods are available that can be used to characterize a feed for gums, asphaltene or stability in the presence of oxygen. While these methods may or may not provide a complete solution to exactly where the fouling problem comes from, they may help to characterize the different feeds at a given site and help narrow down the probable root cause.
• Avoid oxygen contamination of feed.
Direct feeding – Supply feedstock to hydrotreater from upstream unit without using intermediate tankage.
Benefits:
o Eliminates residence time in intermediate tankage, thus minimizing formation of other free radicals.
o By far the cheapest solution and reduces working capital.
Risks:
o Lacks flexibility to accommodate swings in feedstock rate and unit outages.
Tank blanketing – If tanks must be used, they should be blanketed. Nitrogen is the best blanketing gas owing to its reliably low O2 content and ease of venting to atmosphere. Gas blanketed internal floating roof tanks are most effective in minimizing oil contact with O2 and evaporation losses to blanket gas.
Benefits:
o Commercially just as effective as direct feed and overcomes all the limitations.
o O2 cannot react if not in system, therefore should reduce foulant generation.
Risks:
o Cannot impact O2 brought in with import through other feeds
o Choice of correct seal for floating roof and its periodic checking and maintenance.
• Remove Oxygen from Contaminated Feeds.
Oxygen Stripper – Strips out free O2, including import O2 and removes the potential for further formation of peroxides. Common scheme is for ambient temperature hydrogen stripping of the feed to fuel gas system.
Benefits:
o Only feed streams exposed to O2 need to be stripped.
o Maximizing direct feed to the unit in combination with stripping the small O2 contaminant stream is generally more economical than stripping the complete feed stream.
Risks:
o Residence time, particularly in imports, may result in some polymer reaction occurring.
o Expensive option in terms of equipment, and is not so effective if the peroxides/ polymer has already been formed upstream of the stripper.
Injection of anti-oxidant chemical – Antioxidant chemicals have been used with a degree of success in some locations.
Benefits:
o Act as chain stoppers that react preferentially with O2 and peroxides, making them unavailable to take part in free radical polymerization reactions.
Risks:
o Although chemical treatment can help, it is not always successful and it tends to be most effective when the antioxidant is dosed into the upstream unit rundown ahead of the storage tank.
• Remove foulant/prevent laydown.
Hydrogen treat gas injection – Inject hydrogen treat gas upstream instead of downstream of preheat exchangers.
Benefits:
o Hydrogen gas increases turbulence and can also help to reduce polymer formation reactions.
o For VGO HDT hydrogen injection especially for units with separate heating of VGO will prevent thermal cracking of VGO.
o Avoid dry point in exchanger areas where the feedstock is completely evaporated towards dryness as severe fouling may happen. Polymer and gum tends to build up on the shell-side behind baffles, because of relatively stagnant zone. Evaporation of feed leaves less liquid solvent for the gums and gums get deposited. Most severe at the liquid dry point.
• Modify exchanger design – Modify exchanger internals, maintain high velocities in exchangers, appropriately oversize exchangers to lower high tube wall temperatures below the critical temperature required for coking or polymerization.
Parallel exchanger – Flexibility for bypassing and cleaning.
Benefits:
o Clean all exchangers on-the-run, extra exchangers mean no loss of throughput to clean.
Risks:
o No reduction in rate of fouling.
o Additional design features required (such as PRV’s) to safely by-pass/isolate exchangers.
• Anti-foulant chemical injection.
Benefits:
o A reduction in the rate of fouling.
Risks:
o Fouling mechanisms will still occur, probably downstream.
• Prevent corrosion
Corrosion resistant tube metallurgy – select appropriate tube metallurgy to prevent formation of corrosion products that aid the process of foulant formation such as naphthenic acids or high TAN feeds.
Benefit:
o Easy to implement for new unit and revamp of existing unit.
Risks:
o May not be the best solution as metallurgy upgrade is expensive and components other than tubes can still provide corrosion products to aid fouling.
IHSAN RAAD (Shell Catalysts & Technologies)
There are several types of fouling in Hydrotreating feed/effluent exchanger units, the three most common types in the industry are:
1. Inorganic particulates.
2. Organic deposits.
3. Ammonium salts.
Each type of fouling has its own characteristics and deposition locations. Knowledge of the type of fouling and the underlying deposition mechanism is essential to tackle the fouling problem. This can either be done by eliminating the root-cause, or by selecting a fit-for-purpose and cost-effective abatement approach.
1. Inorganic particulates: Inorganic fouling is mainly caused as a result of iron sulfide, sodium or coke fines that can either be carried from upstream units or generated in-situ in the preheat exchanger network. These foulants are:
• Iron Sulphide (FeS) and Iron Oxide (FeO, Fe2O3): Scales of iron oxide (FeO, Fe2O3) and iron sulphide (FeS) are generated as corrosion products within the unit itself but can also come from upstream units, intermediate storage and transport from well to refinery. Important corrosion sources are furnace tubes (hot sulphur corrosion), the CDU overhead condenser and the reactor effluent air cooler. Iron corrosion products in VGO’s are also associated with processing of naphthenic crudes.
• Sodium (Na): Na can come from brackish or salty cooling water (i.e. leaking heat exchangers) or from processing water-containing slops or imported feeds. Sodium in combination with iron has been known to promote coke formation under conditions of high temperature and low pp H2.
• Coke (C): Coke fines can be entrained from VBU’s and cokers, which cause mainly fouling of the feed side of the feed/effluent heat exchangers, furnace tubes and the top beds of reactors. VGO hydrotreaters might also experience coke formation due to a poor separation in the upstream HVU.
2. Organic deposits: The organic foulants are primarily gums formed as a result of processing cracked material and accelerated if the material is exposed to oxygen at any time. The types of foulants are:
• High di-olefin Content: Di-olefins (molecules containing (multiple) double bonds) are mainly found in product streams from (thermal) cracker units but can be present in other streams as well. At the right temperature level (~400-500°F) they polymerize readily to form gum-like substances often showing up as greyish flakes on the FEHXers. The rate of this reaction increases at higher temperatures, making the FEHX especially vulnerable. Elimination of feed streams with high di-olefin content is an easy way to reduce fouling but may not be preferred economically. If long periods of operation with high di-olefins content are expected, an option to avoid high fouling rates could be the installation of a low temperature di-olefin saturation reactor.
• Oxygen in Feed: Oxygen can form a range of different molecules when it is dissolved in a hydrocarbon stream (e.g. peroxides, carboxylic acids, aldehydes and other oxygenated compounds). Amongst other problems, these molecules can initiate polymerization reactions to form gums. Oxygen can enter a feed stream in several ways, including but not limited to air-breathing storage tanks, marine or surface transport vessels, leaks in equipment operating in sub-atmospheric pressure or faulty pump seals. One common point for oxygen to enter feedstock is during storage. Feed streams can be routed directly from unit to unit to avoid intermediate tank storage. Another option is to put in a bypass jump-over on the tank such that only the extra feed goes to tankage and the rest will bypass and go directly to the hydrotreater. All tanks used for storing hydrotreater feed should be nitrogen blanketed, also for straight-run feed. If this is not done, gum formation and other side reaction might happen in the tank itself. Other mitigation is to run it through a stripper, fractionator or distillation unit before introducing it to the unit in order to strip away both dissolved oxygen and the oxygenated compounds, like peroxides. A last option that is used to avoid oxygen related fouling is by injecting anti-oxidants into the feedstock. These compounds are only effective when they are injected prior to the feed stream coming into first contact with the oxygen. So, it should be ideally be injected at the source prior to transportation to site or send to storage. Also, good mixing of the anti-oxidant with the hydrocarbon streams is essential. Only then can they prevent gum formation during storage.
3. Ammonium salts: There are several types of salts that can formed in the effluent exchanger.
• If ammonia and HCl are present, ammonium chloride may deposit directly from the gas phase. The sublimation point in the process depends on the operating conditions (pressure and temperature profile) but also on the concentrations of ammonia (generated from hydrogenation of nitrogen compounds) and chlorides. Main locations of deposition are the feed/effluent heat exchangers (effluent side), the air cooler and recycle gas compressor valves. In the dry state this salt is not corrosive, but in areas where the water dew point is approached, the deposited NH4Cl salt will become moist and can be very corrosive (NH4Cl salts are hygroscopic, therefore the stream temperature must be maintained 15-20 C above the water dew point to assure dryness). Furthermore, apart from the danger of excessive corrosion, NH4Cl deposition can drastically increase the pressure drop and to decrease the effective duty of feed-effluent heat exchangers.
• Like Chloride, organic bromide will convert to hydrogen bromide after hydrotreating and then react with ammonia to form ammonia bromide salt in the exchanger.
• Ammonium bisulphide (NH4HS) hydroprocessing reactors convert sulphur and nitrogen compounds in the feed to H2S and NH3. On cooling, these two compounds react to form NH4HS. In the absence of water, NH4HS deposits to form a crystalline solid that can cause plugging of the reactor effluent air cooler. This will occur at relatively low temperatures, 10-30°C. If the dew point of water is reached in the effluent air cooler (or water cooler) or if insufficient wash water is injected.
In summary, foulants are typically found on the feed side of the preheat exchangers include various gums or polymers, iron sulfide, and salts. The organic fouling due to gums and polymers results from the polymerization of unstable species in the unit feed. Therefore, in order to determine the risk of organic fouling for a particular feed stream, detailed analysis of the feed is required to determine the problematic species in order to evaluate the fouling propensity and mitigation strategies. Another key factor to consider is the oxygen content of the feed stream as this can promote the polymerization of various unstable compounds, particularly olefins. Therefore, it is a good practice to exclude oxygen from feed storage tanks using a nitrogen blanket. However, this method is ineffective with streams already exposed to oxygen. The inorganic fouling is mainly caused as a result of iron sulfide that can either be carried from upstream units or generated in-situ in the preheat exchanger network. Identifications of the contaminants source and mitigations are key to eliminate the inorganic foulants.
SERGIO ROBLEDO (Haldor Topsoe, Inc.)
To answer this question, we need to differentiate between feed-side fouling and effluent-side fouling. Potential causes and mitigations will depend on which side is experiencing the fouling.
Feed-side fouling in your F/E exchangers can be the result of:
• Olefins
• Oxygen
• Particulates
Olefins/Oxygen
Olefins are normally introduced with cracked stocks in the feed. Typically, olefin gumming happens at lower temperatures (300 – 350 °F). Gums formed from peroxides, as a results of oxygen contamination of straight run feed, usually occurs at >400 °F.
In the case of coker naphtha, conjugated diolefins are present which are highly reactive species. In the presence of very small amounts of oxygen, or at elevated temperatures above 450 °F, these molecules will radically polymerize to form gum that can foul exchangers causing poor heat transfer as well as high pressure drop. If the feed contains significant quantity of coker naphtha then these Diolefins must be removed to prevent gum formation.
The coker naphtha should preferentially be sent to directly from the coking unit to the hydrotreater to prevent contamination with oxygen. Even straight run stock, which may be part of the feed component, must be prevented from contacting oxygen by storing the feed in a nitrogen blanketed storage tank.
Even with strict adherence to avoid feed contact with oxygen, the diolefins in the coker naphtha can polymerize at elevated temperatures. A dedicated saturation reactor operating in the range of 300 °F to 450 F will ensure that these highly reactive species are removed from the feed before polymerization can take place. Once the diolefins are removed from the feed then the feed can be heated to the required temperature for the required operating scenario.
Keep in mind that even though cracked stocks are not fed directly to a unit, there is potential of introducing cracked stock in sites that process slop in their Crude unit.
Particulates
Particulates, at high enough concentrations, in conjunction with low tube velocities, can result in these particulates settling out and plating on the surface. If no filter is present, then plans should be made to engineer and install a filter system to reduce the amount of particulates present in the feed. There are also companies that offer tube inserts to reduce the likelihood of particulates settling out and plating on the tube surface, preventing a loss of heat transfer.
Even with filters and tube inserts, if any gumming is taking place in the exchangers, then any small amount of particulates present will be picked up by the gums formed.
Examples in industry where fouling occurred on the feed side are:
• Cracked stocks blended with Canadian crude coming down the pipeline. These formed gums with oxygen and fouled the exchangers.
• Crudes from Venezuela blended with cracked stocks.
• Virgin jet was exposed to oxygen and fouled in exchangers operating >400 °F (this happened in multiple units).
o Similar example with natural gasoline.
As mentioned before, preventing oxygen ingress via direct, hot feed of cracked stocks to the unit, along with floating roof and/or nitrogen blanket on tanks is imperative. Tube inserts are also a viable option to prevent fouling where tube velocities are low enough allowing particulates to drop out in the exchanger. Most importantly, quality control is paramount in preventing this, or reducing further loss in performance. Examples of actions are:
• Notify the shipper and have the diluent stream changed.
− What is the crude source? Are there potential cracked stocks coming down the same line? How about Canadian crudes?
• Notify crude supplier about the poor quality.
• Install an oxygen stripper.
− Done for the virgin jet example.
• Install nitrogen blanket on feed tank or change to floating roof tank.
• Inject antioxidants into the tanks (mixed results).
• Bypass tank and go hot (direct) to the unit.
As for effluent side fouling, this is typically the result of NH3Cl (salts), which are the result of high levels of Cl in the feed. A water wash should be installed to remove these salts, and continuous is recommended versus intermittent.
• A licensor can help calculate where the water should be injected.
– Need to inject enough water and at the right spot to keep it as a liquid and not vaporize
– Liquid water will wash out the salts while steam will not
– Licensor can calculate where the dry point will occur and how much water needs to be injected
• Many examples of where water wash helps
– If done at the right spot with the correct amount of water and at the right temperature
• Boiler fouling
– Water treatment company can help with this
There is also a very good P&P this year covering reactor effluent diseases jointly presented by Flint Hills Resources and Marathon. Please plan to attend to learn more.