Question 1: What are the benefits of alumina treating in sulfuric acid alkylation and HF alkylation? Has this technology been proven commercially?
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
-
Typically, all alkylation units treat the effluent to remove free acid and/or contaminants
-
Fluorides and Sulfates are the typical contaminates removed from HF and Sulfuric acid units, respectively
-
Activated alumina is commonly used in HF service
-
Caustic treating is common in sulfuric acid units but dry treating using alumina is also used
LIZA PACHECO (STRATCO)
In the sulfuric acid alkylation process, droplets of acid are found in the effluent stream from the reactor. These droplets, which are formed primarily from the alkylation chemistry itself and the formation of intermediates, are small and stable and cannot be removed by gravity settling and / or coalescing alone. Traditionally, a caustic/alkaline water system has been used to remove the acidic droplets from the effluent stream through neutralization. Consequently, the water that is introduced in this section travels to the reaction section where it will dilute the acid and increase corrosion. When activated alumina is used, the acid droplets are removed by the chemical reaction of the acid with the activated alumina. No water is introduced into the system which will be beneficial due to the decrease of acid consumption and corrosion in the treating and reaction sections. Refiners have reported longer Contactor™ tube bundle life when operating a dry system using alumina compared to the caustic/ alkaline water effluent treating system where water is introduced into the process and recycles back to the reaction zone. While several STRATCO® alkylation units have operated with dry alumina treating for many years, only in the last few years has this been part of the standard STRATCO® alkylation offering. As part of this transition, DuPont has made a commitment to research for continuous improvements to this process.
KURT DETRICK and TROY SMALL (UOP)
Alumina and promoted alumina products are commonly used in HF alkylation to remove organic fluorides from the propane and butane product streams. The benefits of using alumina to remove organic fluorides from the product streams include prevention of corrosion in downstream fractionation towers, prevention of catalyst deactivation in downstream catalytic units (such as Butamer) and making the propane safe for in-home use (such as in stoves and heaters). While both alumina and promoted alumina, products have been commercially proven, the industry trend is towards promoted alumina products, which generally have a higher fluoride capacity than non-promoted alumina products.
Alumina treating in sulfuric alkylation units is not common.
Question 2: What metallurgy works well and does not work well for use in alkylation units? In what applications does the alternate metallurgy perform better/worse?
LANCE TALLMAN (CITGO Petroleum)
HF
The standard metallurgy for use throughout HF alkylation units is carbon steel. Showing a high degree of corrosion resistance in both low and very high acid concentrations, carbon steel is also useful for its relatively low cost compared to more exotic materials. In most applications throughout the unit, carbon steel will display a general corrosion loss rather than the more difficult to detect pitting or localized corrosion. However, it must be noted that when used in a stream that will contain HF acid, there are several restrictions that are placed on the steel composition (particularly Cr, Ni, and Cu) in order to try and reduce the unique circumstances that can set up localized areas of accelerated HF corrosion. Despite the possibility of accelerated corrosion, carbon steel has a long history of suitable use throughout Alkylation Units throughout the country and performs well in most streams present in an Alkylation Unit.
There are some streams that, through use and time, have been found to require an alternate metallurgy to the standard approach of carbon steel. These areas typically involve a higher temperature than are encountered in most of the unit (most refiners using carbon steel for HF containing streams at operating temperatures below 150 – 200°F). At temperatures higher than this, or in areas that previous experience with carbon steel has provided unsuitable corrosion resistance, consideration is made for employing the use of a high nickel alloy. The common choices for use vary between Monel 400 and various Hastelloy’s (C-276, B grades, etc.), with Monel getting the preponderance of use. These high nickel materials provide suitable corrosion resistance at the higher temperature operation than are possible with carbon steel. Monel 400 brings about its own set of challenges with the possibilities of cracking due to high residual stresses, particularly in services that contain oxygen or mercury contamination. A stress relieving heat treatment is usually performed at initial fabrication to reduce the possibility of this catastrophic failure mechanism.
Most notable of the common materials used in refining that should not be employed in HF Alkylation units are stainless steels. While they can sufficiently resist corrosion in anhydrous environments containing HF acid, they perform worse than carbon steel under aqueous conditions. All grades of stainless steel can undergo rapid corrosion losses (sometimes on the order of 100 mpy) at certain conditions and should therefore be avoided. Other materials to avoid are those that contain silicon, as this will react with HF acid and form a highly corrosive fluorosilicic acid.
Sulfuric Acid
Carbon steel is used for most of the equipment and piping in sulfuric acid alkylation units including the reaction zone (contactors & acid settlers). However, CS doesn’t work well in the presence of sulfuric acid with high temperatures and/or high velocities. The fresh and spent acid piping is typically constructed of 300 series SS, and/or Alloy 20; however, some units have successfully operated with CS piping in this service. Additionally, in the effluent treating section & depropanizer feed treatment system, the static mixers and upstream/downstream piping are typically upgraded to Alloy 20 and/or Hastelloy C. Although the contactors (shell & tube bundle) are constructed of CS, due to the highly turbulent service, the internals such as the reactor impeller & distributors are upgraded to Hastelloy C which will significantly extend the life of this equipment.
Monel is not recommended for material of construction in sulfuric acid alkylation units as it is not compatible with equipment and piping containing large amounts of sulfuric acid.
KURT DETRICK (UOP)
API RP 751 has a good description of the experience with different materials in the HF Alky unit in Section 3 and Annex D.
Carbon steel is the most common material used in HF Alky units in areas of relatively low temperature and low water content in the acid. The surface of fresh steel reacts with HF to form iron fluoride, but under the proper conditions (primarily low water concentration in the acid), this layer of iron fluoride protects the underlying steel from further attack.
Monel is used in areas of the HF Alky unit where temperatures are higher, or water content of the acid is higher (such as in the Acid Regenerator or Rerun tower). Hastelloy (primarily C276) has also been used successfully in these areas of elevated temperature and/or water concentration.
316 Stainless steel vessels are used for transporting fresh anhydrous HF to the Alky unit, but stainless steel should only be used for high purity fresh acid. In commercial experience, stainless steels (both Austenitic and Martensitic) are aggressively attacked by the plant acid in an HF Alky unit and these stainless steels should never be used in the HF Alky unit itself.
ROMAIN LEMOINE (McDermott Lummus Global)
In less than 7 years, McDermott’s Lummus Technology has successfully commercialized and licensed an advanced version of sulfuric acid alkylation technology, called CDAlky®, to clients worldwide. CDAlky technology focuses on effectively eliminating the root cause of drawbacks inherent to conventional alkylation units. A leading area of concern is excessive corrosion and the need for higher-grade metallurgy.
Lummus’ CDAlky Technology not only operates at lower temperature than conventional sulfuric acid alkylation processes, but it also eliminates the need for a reactor effluent post-treatment section. The key to these technical breakthroughs resides in CDAlky reactor proprietary internals: AlkyPak™ & Distributor Plates. By eliminating caustic and water introduction in the alkylation section, and by maintaining a low temperature operation, CDAlky technology can eliminate the need for higher-grade metallurgy. Recent turnaround inspections conducted on multiple CDAlky units have confirmed these technical steps out benefits:
-
Low Temperature operation reduces significantly the corrosion rates.
-
Neither reactor effluent wash nor post-treatment steps are required to remove sulfuric acid or sulfates from the alkylate.
-
No fouling material is observed in any fractionation reboilers.
-
Stainless steels and carbon steels are suitable material of construction.
GREG TROUTMAN (DuPont)
Although careful consideration is required when selecting appropriate metallurgy for any sulfuric acid alkylation unit, a sound process design relegates the recommended use of exotic material to only a few areas. Carbon steel is by far the most common material found throughout the unit as it is both economical and capable of handling the range of temperatures and velocities observed in normal conditions as well as most alkylation unit excursions. The typical hierarchy of metals found in a sulfuric acid alkylation unit is as follows:
Carbon Steel < 316L Stainless Steel < Alloy 20 (in certain situations) < Alloy C276
With respect to sulfuric acid, carbon steel can be used if the velocity/turbulence through the piping or equipment is not high enough to disturb the protective passive layer of iron sulfate that forms on carbon steel when contacted with sulfuric acid. Many refiners have utilized carbon steel with a corrosion allowance of either 1/8” or 1/4” for normally flowing and normally not flowing lines, respectively. In piping where, high velocity/turbulence cannot be avoided such as through valves, Alloy 20 or PTFE-lined valving should be used to avoid excessive corrosion/erosion. When velocities high enough to avoid hydrogen grooving and low enough to avoid eroding the passive layer are not obtainable with sulfuric acid piping, 316L stainless steel is a good substitute.
Areas where neutralization takes place will require at least Alloy 20, especially since most of the neutralization takes place in a static mixer where there is high turbulence. It should be noted that while Alloy 20 is an excellent material in many applications, situations such as high temperature and certain contaminants or environments can cause it to become active and fail quickly.
For the reactor itself, carbon steel is the preferred choice for the bulk components (shell, heads, tube bundle, etc.). Carbon steel is economical where the process conditions (velocity, turbulence and temperature) are typically mild. Since the carbon steel within the reactor forms a protective passive layer, a long life is typical. The reactor impeller, wear ring, feed nozzles and other high velocity/turbulent areas can be constructed of Alloy C276 to dramatically improve the life span.
DuPont never recommends the use of Alloy 20 within a sulfuric acid alkylation reactor due to process conditions that can cause the metal to become active and fail. In addition, areas where neutralization takes place with elevated temperatures such as the mixing of sulfuric acid and hot alkaline water are better suited for Alloy C276 rather than Alloy 20.
A few additional considerations:
For areas with high amounts of propane such as the depropanizer overhead, depressurization could result in temperatures cold enough to require low temperature carbon steel.
Carbon steel is suitable for caustic (typically in the 10-12% range) but post weld heat treating should be used in areas where the temperatures are expected to be higher than 180 °F.
Both stainless steel and Alloy 20 should not be used when significant amounts of halide ions such as Cl- are present as these may cause pitting corrosion.
Water is not corrosive by itself, but it can cause major corrosion issues if there is an area with a high probability of contacting trace SO2 or sulfuric acid.
As for metallurgies that are not compatible in sulfuric acid alkylation units, Monel is not compatible with equipment and piping containing large amounts of sulfuric acid. However, Monel has been shown to be effective in hydrocarbon streams with weak acid. This is a benefit when considering HF to sulfuric acid alkylation conversion as Monel is prevalent in HF alkylation units.
Non-metallics such as PTFE, polypropylene, and glass have excellent corrosion resistance and can be considered for various portions of the plant. However, fire resistance and physical strength must be also considered.
Corrosion is a complicated phenomenon and is not always predictable or even explainable by current metallurgical knowledge. DuPont continues to experiment and gain knowhow on the performance of specific materials in sulfuric acid alkylation units. For the latest information, DuPont and/or a metallurgist should be involved in corrosion evaluations and upgrade decisions.
Question 3: What type of release mitigation safety systems do you use for sulfuric acid and HF alkylation units?
LANCE TALLMAN (CITGO Petroleum)
HF
-
Rapid Acid De-Inventory systems to move HF acid from unit to remote acid storage drums
-
Acid Settler, reactor risers and coolers (includes unit shutdown)
-
Acid Storage Drum
-
Water Curtain surrounding high acid area of unit (with auto-activation)
-
Water deluges on Acid Settler, Remote Acid Storage Drums and acid-service pumps
-
Elevated, remotely operable fire monitors with independent supplemental water supply
-
Camera systems to improve response time
-
Point source hydrocarbon detectors
-
Point source HF detectors
-
Bi-level perimeter laser HF detectors
-
Remote capabilities to start, stop, isolate, vent to flare and deluge acid-service pumps
-
Remote control panel for acid truck unloading (isolation valves)
-
Closed circuit monitoring of unit
-
Sulfuric Acid
Sulfuric acid alkylation units do not require the robust safety systems as the HF Alky units since sulfuric acid is a liquid at atmospheric conditions. The release mitigation safety systems typically used are curbed and/or diked areas to contain the liquid sulfuric acid in event of a leak. This exists in the reaction section, blowdown section, and fresh/spent acid tanks. The same mitigation systems are in place in the effluent treating section and the depropanizer feed treating system where caustic is present. When Operators respond to any release, they’re equipped with the appropriate PPE, which include slicker suit, face shield, and proper gloves. In addition, hydrocarbon detectors are present in and around the unit due to the presence of LPGs.
SHANE PRESLEY (DuPont)
Sulfuric acid is a liquid at atmospheric conditions and will pool if released rather than form a vapor cloud. During the summer of 1991 an independent consultant performed a series of large-scale sulfuric acid leak tests. In all, thirty-six release tests were conducted, three tests utilizing alkylation equilibrium acid from an operating commercial alkylation unit and thirty-three tests using an acid/hydrocarbon emulsion. The tests involving equilibrium acid resulted in essentially 100% recovery of the acid released; the remaining tests resulted in an average 97.6% recovery of the acid released (Figure 1) [1].
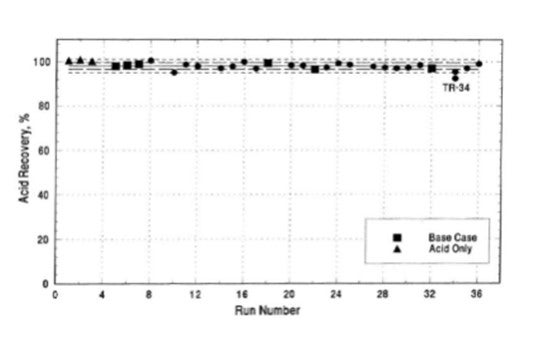
Figure 1: Sulfuric Acid Test Recovery
Because sulfuric acid will pool when released rather than forming a toxic vapor cloud, only basic mitigations are required. These mitigations typically include curbed and/or diked areas around sections of the unit containing sulfuric acid to contain the acid in the event of a leak. These areas typically include the reaction section, acid blowdown section, and fresh/spent acid storage areas. In the event of small or minor leaks, sulfuric acid can be neutralized with soda ash before removal. Larger spills may require physical removal of the acid. Other release mitigation systems in place are not specific for sulfuric acid and are typical for other processing units containing LPG hydrocarbons.
Question 4: What are the requirements for a unit depressurization valve in isom unit using a chlorided alumina catalyst based? Do you require the depressurization valve to be operable from the console and/or emergency shutdown device (ESD) panel?
GAYL MERCADO (Axens North America)
Axens recommends that in cases of emergency depressurization of the reactor circuit, the circuit fluid is depressurized into the product stabilizer with the light gases still being treated in the off-gas caustic scrubber before being sent to flare.
An Isom unit is typically designed such that there are several automatic shutdown process interlocks that will place the unit in a non-hazardous status should a major failure occur. An emergency shutdown hand switch that is not an automatic shutdown, but a board mounted hand switch that can be used at the discretion of the operator, is also a standard design philosophy. Should an emergency occur (e.g., temperature runaway in the reactor, fire), the operator has the capability to activate this switch which will initiate an emergency shutdown. It should be noted that care should be taken to protect pressure safety and emergency depressurization valves in the reactor circuit to prevent corrosion of the valve and subsequent loss of relief capabilities.
The depressurization valve can either be operable from the console or an ESD panel based on site preference or company standards. At minimum, the valve should be operable from an ESD Panel. If the valve can be operated from the console, safeguards should be in place to prevent unintended depressurizations.
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
-
One licensor is adding Rx depressurizing to new Isom units
-
The first step in depressuring an Isom Rx should be through the net gas caustic scrubber to fuel
-
Keep adequate positive pressure to prevent back flow from the fuel gas system
JOCELYN DAGUIO (UOP)
In UOP’s current design of a Hydrogen Once Through (HOT) Penex™ or Butamer™ unit, dual (2) depressurization valves are located at the outlet of the Lag (or Last) Reactor into the flare header. Two valves are specified to prevent HCl leakage into the flare header. The valves and line sizes are sized appropriately to meet a target velocity during a depressurization event.
The valves are opened when the reactor depressure Emergency Shutdown (ESD) is initiated from the board-mounted hand switch. UOP recommends flushing the flare header with dry nitrogen after the depressuring ESD to purge the HCl containing hydrocarbons that would be in the flare. This is a preventative action to minimize flare corrosion.
For revamps of existing units, UOP recommends, but does not require a retrofit using the current reactor de-pressure ESD.
Question 5: What is your experience with CCR catalyst on-the-fly replacement compared to changing at turnaround?
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
Valero’s best practice is to evaluate changing CCR catalyst during turnarounds as it saves about 25% of the cost vs. on-the-fly change out as follows:
-
-10% vs. On the Fly replacement which typically adds 110% catalyst to account for back mixing
-
-10% vs. the next T/A Dump Screen and Reload following the on-the-fly change out
-
-5% for miscellaneous costs such as extra platinum reclaim/ losses and extra catalyst handling
Don’t necessarily change catalyst out during every turnaround but evaluate at turnaround intervals. Only change catalyst when it is economic.
GAYL MERCADO (Axens North America)
On-the-fly catalyst replacements are becoming more common, but still an economic decision for the refiner based on trade-offs between the continuous replacement approach and the historical changing at turnaround approach. On-the-fly catalyst replacement allows for full catalyst replacement while catalyst circulation is maintained, with both reaction and regeneration sections still in operation to allow for reduced unit downtime. Keeping the unit in full operation gives refiners the ability to replace catalyst outside of a planned turnaround while the unit is still in operation to take advantage of improved yields by replacing with fresh catalyst.
On-the-fly catalyst replacement requires a 24-hour loading operation for a 6 to 7-day period, but with minimal production loss. During the change-out, catalyst circulation is redirected so that the unit catalyst is unloaded from a Lock Hopper drum at the bottom of the regenerator and fresh catalyst is loaded in its place upstream of the reduction chamber. Backmixing of spent catalyst is minimized to less than 3 to 4% with this procedure. Safety risks are also minimized with on-the-fly change-outs as reactor entry and drying is not required.
With on-the-fly catalyst change-out, refiners do not have the ability to inspect CCR reactors and regenerator since the unit is still on stream. Axens recommends that refiners plan to clean and inspect these vessels during a regularly scheduled turnaround.
KA LOK (UOP)
UOP’s Catalyst Changeout On-The-Fly is a design feature that allows for replacement of the total catalyst inventory in the CCR Platforming™ unit and the CCR section while maintaining the Platforming unit operations. Refineries find this design useful when there is a desire to replace old CCR Platforming catalyst with a new load of catalyst between scheduled unit turnarounds. The old catalyst is unloaded from the CCR section while fresh makeup catalyst is continuously added to the CCR section until the inventory of the old catalyst is replaced. During the Changeout On-The-Fly operation, coke is removed from the old catalyst before it is removed from the unit for easy catalyst handling and transportation. UOP incorporates this Catalyst On-The-Fly design in all three types of UOP’s CCR units which include the Atmospheric CCR, Pressurized CCR and CycleMax™ CCR. UOP has experience with this reload method for small and large size CCR units including a 6,000 lb/hr (2,727 kg/hr) CCR unit. In addition, UOP has experience with changing from regular density catalyst to high density catalyst using this reload method. Due to the economic benefits of this reload method, refineries have completed more than 60 catalyst reloads using this approach.
Alternatively, catalyst replacement can be executed during scheduled unit turnarounds. The old catalyst from the Platforming reactors and CCR section will be unloaded. The unit will be replenished with fresh catalyst during the turnaround. The turnaround schedule would have to consider of the time required for catalyst unloading and loading. Using this conventional catalyst change method, the catalyst unloaded from the reactors is not coke free. Transportation and catalyst handling of the unloaded coked catalyst will need to be arranged accordingly.
Question 6: What are the catalyst or design considerations that you employ to repurpose isom units for other services, or to repurpose another type of unit into an isom unit?
GAYL MERCADO (Axens North America)
With increasing quantities of lower octane naphthas reaching the gasoline pool and potential for higher octane demand in the finished stocks, the octane boost from isomerization units has grown in importance. While the installation of new units is ideal, many refineries have been looking at utilizing idled units or converting existing units to isomerization type operation. Repurposing of older or idled units is ideal given isomerizations generally low severity operation and clean operating environment.
Axens has experience with converting benzene saturation units, fixed bed reformers, as well as less conventional idled units and equipment into isomerization units. More often, benzene saturation units are a first look to minimize capital investment for these revamps given the overlap in feed type and quality. More importantly, provided the below key concerns are managed when repurposing existing units into an isomerization unit, many existing equipment and units are potential candidates:
- Feed and hydrogen quality for catalyst deactivation
- Reactor liquid distribution to maximize isomerization reactions
- Heat integration to maximize isomerization equilibrium
- Product fractionation for RVP control
HEIDI FRASER (UOP)
In repurposing any type of unit into another type of unit certain considerations should be made. Conversion to or from a naphtha isomerization unit is no different.
- First, you should consider the general layout of the equipment available and the requirements of the desired unit. Is this a recycle gas unit with a reactor product separator and recycle gas compressor? Some processes require recycle gas and others can be operated hydrogen once through. In UOP’s naphtha isomerization world, Par-Isom™ units are nearly always have recycle gas while Penex™ and Butamer™ units can be designed as either with recycle gas or hydrogen once through.
- Second, what is the operating pressure of the equipment available? Operating pressure is often set by the available pressure of the feed or makeup gas to the unit, or by the pressure of the destination to which the products will be sent. But operating pressure will often determine the type of metallurgy used in the construction of the equipment. And some catalyst systems will have better performance in certain pressure ranges. Also, the system pressure, along with hydrogen availability, will determine the hydrogen partial pressure achievable in the reactors which will also affect the catalyst performance. UOP’s Penex and Par-Isom units are generally designed for a reactor system pressure of 450 psig but revamped units are in operation with reactor pressures as low as 250 psig. There is some debit in yields at pressures less than 450 psig.
- Also important are the size and type of the reactor(s) available. Are the reactors large enough to hold the required quantity of catalyst for good conversion? Are the reactors downflow or radial flow? Are modifications required to the reactor internals? Will the modifications affect the amount of catalyst the reactors can hold? All UOP Isom units have downflow reactors. If converting from a radial flow reactor the center pipe and scallops would have to be removed. Hydrogen-Once-Through Penex units and some Par-Isom units will require the addition of a vapor-liquid tray to the top of the reactor. This will reduce the amount of catalyst that a reactor can hold. The reactor vessel should be large enough to hold the required amount of catalyst after internals modifications. Penex units are generally designed for a LHSV of 1-1.5 hr-1. Par-Isom units normally have a WHSV between 0.9 and 3.0 hr-1. Butamer units are designed for LHSV of 4-6 hr-1. If less catalyst can fit in the reactor, the space velocities will be higher resulting in lower conversion.
- Another consideration is the operating temperature of the existing equipment as compared to the desired process. What type of heater is present or needed? For instance, hydrotreaters, reformers, and older zeolytic isom units operate at high temperatures and require a fired heater for the reactor charge. UOP’s current generation of isomerization catalysts operate at lower temperatures, typically 200-400°F, and require only a steam heater or hot oil heater. Fired heaters would produce too high a temperature for these catalysts and could lead to temperature excursions.
- Metallurgy requirements should also be examined. UOP Isom units are designed with all killed carbon steel construction. Recent changes to the Nelson curve may indicate PWHT requirements for reactor sections. If an Isom unit were being repurposed for other service, the evaluation of the metallurgy could be important.
- When looking at repurposing units we tend to concentrate on the reactor section. However, consideration should also be made to the other equipment in the process, some of which is more easily replaced or revamped than others. Are compressors of appropriate size to supply the needed amount of makeup gas and/or recycle gas? Are distillation columns large enough to handle the expected flow rates and achieve the necessary separation? Look at column diameter, number of trays, reboiler and condenser capacity, and reflux pump sizing. Consider if extra feed treatment is necessary for the new process and whether vessels are available that could be put to that use. These would include chloride treaters or sulfur guard beds, driers for feed, makeup gas, or recycle gas streams
Repurposing of equipment to another process is possible. UOP has experience revamping hydrotreaters and semi-regen reforming units into UOP isomerization units. We have also converted older generation zeolytic isomerization units into newer generation isomerization units. We have less experience converting isomerization units into other processes but would expect it is possible.
Question 7: How do you monitor and protect the heater tubes from overheating in high temperature services such as catalytic reformer heaters? How is the tube wall temperature monitored?
GAYL MERCADO (Axens North America)
As a first step and low investment solution, refiners will perform a visual check on the color of tubes to check for color differences or perform a thermal scan of the tubes. These solutions are not very accurate due to iron scale formation and ceramic coating on the tubes. However, a simple visual check of tube color or thermal scan can offer refiners a baseline or trend for monitoring heater tube temps.
Tube skin thermocouples can also be installed either during design or a turnaround. On reformer heaters, tube skin thermocouples are not standard during design due to the large number of thermocouples that would be required for proper measurement. Additionally, these tube skin temperatures may not be very reliable due to the thermocouples being burned out and/or the formation of coke on the probe.
More advanced tools with everyday capabilities are becoming more prevalent in the industry and will move the safety and reliability of the fired heaters to improved levels. Using data transfer and gathering improvements, software systems are beginning to replace the historical approaches that the industry has been using for some time. Axens’ Connect’InTM digital tool, which allows users to remotely monitor and analyze catalyst and unit performance, also allow engineers and operators to track key performance indicators on the heaters and detect possible issues concerning the equipment. The Connect’InTM software architecture allows users to automatically schedule data gathering, validate the data and calculate the heater yield and efficiency. Within a single tool, users can analyze data trends, calculate tube remaining life, track instrumentation problems and help diagnose issues.
Axens recommends the below maintenance approaches to minimize tubes from overheating:
-
Proper maintenance of heater burners to ensure normal flame patterns will help to protect heater tubes from overheating. If overheating is suspected, some burners can be turned off or on depending on the tube-skin temperature patterns
-
Reduction of the heat load on the limited heater which will then shift the required heat load on the other heaters that are not limited in heater tube temperature. However, this will result in non-flat temperature profile
-
Reducing excess oxygen can help to lower and protect the heater tubes from overheating
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
-
IR scans are commonly used to monitor tube skin temperature
-
Rust and scale on the tubes elevates the tube skin temperature using an IR scan
-
Ceramic coating tubes during T/A may reduce the rust and scale on tubes
-
Be aware of concentrated corrosion/erosion at tube weld attachments
-
Either licensor can calculate the tube skin temperature from process data
STEVE PHILOON (UOP)
UOP recommends the use of a calculation to estimate the temperature of the tube metal in the heaters of a naphtha reforming unit. The method described in the API Standard 530 “Calculation of Heater-tube Thickness in Petroleum Refineries” or similar can be used.
UOP’s experience is that knife edge skin thermocouples can be useful as indicators of trends and step changes but are not reliably accurate indicators of the actual tube metal temperature. Radiant heat from the flames can cause readings that are higher than the actual tube temperatures. Shielded type skin thermocouples can provide a good indication of the tube metal temperature but the installation of the shield may create a hot spot on the tube that result in metal loss due to carburization.
Infrared pyrometers provide an indication that is higher than the actual temperature of the metal of the tube because they are reading the temperature of the scale on the outside of the tube. When the tubes are clean the indication will be accurate. This will be the case immediately after the start-up of a new unit or the restart after tube cleaning or tube replacement or if the tubes are ceramic coated. These clean-tube readings provide field verification of the temperatures estimated by the calculation method. The increasing indication from the infrared readings, over-time in an operating unit when compared to the tube metal temperature estimated using a calculation method provides direct indication of the fouling on the tubes.
RICHARD TODD (Norton Engineering)
While tube metal temperature thermocouples can provide indication of TMT trends over time their long
term reliability is always problematic. Therefore, it is imperative that owners/operators of reforming heaters obtain routine IR pyrometer readings of tube temperatures as a back-up to the installed TMT instruments. Even at the start of a run, IR scan data is necessary to establish a baseline for future comparisons.
An IR scan of tubes in any heater does not provide highly accurate readings for a myriad of reasons, some controllable and some not. It is therefore important that the owner understand and control those variables which can be controlled. Chief among the controllable variables is the equipment being used to conduct the IR scan, the operator of that equipment and the target points for the scan. The ideal situation for collection of TMT data by IR scan would be a single individual conducting all scans with the same equipment shooting the same points on the heater tubes. While this is never totally practical, many
operators have been successful in monitoring operating reformer heaters by controlling these parameters
as closely as possible.
Several methods have been used to check the absolute accuracy of IR TMT measurements, all with varying degrees of success. The first method uses a target tube (partial tube without cooling) fitted with an internal shielded thermocouple. This method provides a similar tube surface to an operating tube and a highly accurate tube temperature measurement. The drawback is that this tube will operate at significantly higher temperatures than the operating tube, so the check temperature is typically 200 to 300°F higher than the actual operating tube temperatures. Another method is to check the tube surface temperature on the front face using a preformed hoop contact thermocouple which can be inserted into the firebox through a peep door and “hooked” around a tube to measure the front surface temperature at or very near an IR target point.
As an alternative to routine IR scanning, there are several vendors who can provide firebox cameras capable of continuous TMT monitoring capabilities. These cameras are not in widespread use, but they can provide invaluable operating trends for units which are being pushed to their limits.
Question 8: Fixed bed reforming and CCR reforming recycle gas compressor washing to remove salt deposits. What is the frequency? What is the typical deposit composition? What is used to wash the compressor?
LANCE TALLMAN (CITGO Petroleum)
Fixed Bed Reforming
The recycle compressor is water washed at every catalyst regeneration (typically once per year) after the chlorination step. In addition, if there is any extended outage for mechanical work, the compressor will be water washed anytime there is an opportunity to do so as precaution. Steam condensate is used as the source of water. The deposits are mostly ammonium chlorides and are readily removed by a hot condensate wash. On a rare occasion, if the compressor develops a high vibration, there could be a need to perform a compressor wash.
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
-
Nitrogen from the NHT causes ammonium chloride salt build up in cold areas of Reforming units
-
Intermittent water washing can be done, but water is a Reforming catalyst poison and must be minimized
-
Compressor washing during T/A is a good practice
-
Bar-ing (or rotating) the recycle compressor during T/A is a good practice
RUSS WILTSE (UOP)
During normal operation of a Fixed Bed Reforming Unit the Recycle Gas Compressor internals may become coated with a fine powder like deposit. This material is expected to mostly be ammonium chloride salts which are a by-product of having some organic nitrogen compounds in the Reforming Unit feed. These organic nitrogen compounds will breakdown across the catalyst bed and form ammonia. This ammonia can combine with the trace HCl in the recycle gas to from ammonium chloride salts which can deposit in low temperature sections of the unit. These areas may include the Combined Feed Exchanger, Product Condenser, Stabilizer Column, and the Recycle Gas Compressor. These ammonium chloride salts are water soluble and as such can be removed with a water wash.
UOP recommends washing the recycle compressor during each regeneration before starting the carbon burn. The wash is recommended before the carbon burn to reduce the risk of compressor vibrations and corrosion. During carbon burn the regeneration gas will be wet and it is possible to have some of these deposits spall off the rotor. This could cause balance problems that would result in high compressor vibrations. There is also a concern for under-deposit corrosion if these salts adsorb moisture from the wet regeneration gas.
UOP has observed refiners using a range of different solutions to water wash Recycle Gas Compressors. These mixtures are typically comprised of a basic solution with non-sudsing detergent. UOP typically recommends ~2% soda ash solution with 0.1-0.3% non-sudsing detergent such as Calgon, Cascade, tri-potassium phosphate, etc. It is important to discuss these chemicals and the wash procedure with the compressor vendor to ensure the wash will not cause unexpected harm to the compressor. For example, high concentrations of carbonate may damage aluminum components in the compressor. After the salts have been removed the compressor should be flushed with clean water and treated with a corrosion inhibitor.
Question 9: Are you adding unusual feeds to catalytic condensation or oligomerization units?
GAYL MERCADO (Axens North America)
Refiners have been pursuing the use of more atypical feeds to their olefin oligomerization units for various reasons. Units have historically operated on FCC LPG cuts, but an interest to process Coker LPGs or even heavier FCC olefins have begun.
The use of these alternative feeds has historically been limited by several factors, but newer catalyst generations and types have allowed a wider envelope of feeds while maintaining stable operation with long cycle lengths.
Refiners have been looking to a propane/propylene (PP) cut to units processing heavier materials. As well, options to remove C5s from the gasoline pool through oligomerization to heavier material can make sense where refiners find themselves long on light material in the pool, ultimately reducing the RVP of final blend.
Within Axens, the oligomerization suite of technologies was developed around a single robust catalyst, that can operate in wide range of environments and with various feed qualities.
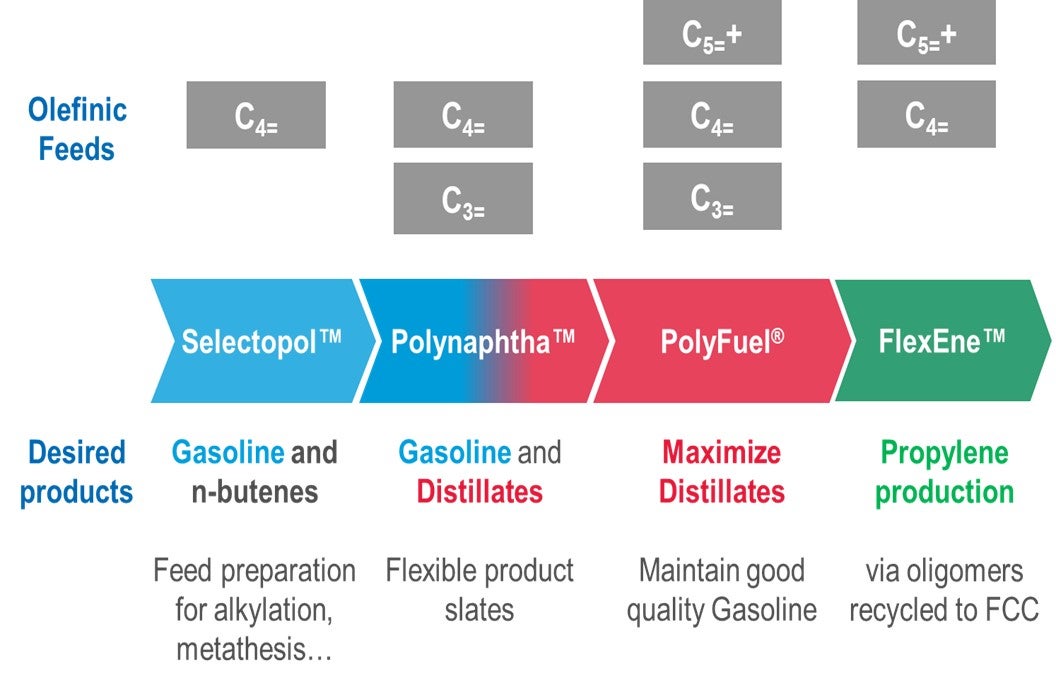
When operating with traditional FCC and Coker LPGs, the PolynaphthaTM focuses on converting into gasoline and middle distillates, providing an economical solution for refiners to tackle the unbalanced gasoline/middle distillates slates. When looking to maximize octane, the SelectopolTM technology targets conversion of only the isobutene portion of an olefinic C4 fraction to either produce a high octane, low RVP gasoline blending stock or to produce a trimethyl pentene stream for petrochemical applications. The enriched n-butene product is an ideal feed to an alkylation unit without requiring any pretreatment. Lastly, the PolyFuelTM technology expands on the operation of lighter cuts by the co-processing, or even direct processing of heavier olefins to produce a heavy gasoline and middle distillates at lower cost.
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
-
An FCC Cat-Poly reactor will convert propylene and butylene into gasoline blending components
PAT BULLEN and JOCELYN DAGUIO (UOP)
UOP has not designed any units with feeds outside of C3/C4 range. We have heard from customers discussing adding C5 olefins and benzene to their existing Cat Poly units but have not received any data from this type of operation to evaluate its efficacy.