
Question 52: What strategies have you employed to profitably manage Tier III regulations (i.e. crude purchasing strategy, pre/post treating, FCC operational changes, sulfur reducing additives, etc.)?
TODD HOCHHEISER (Johnson Matthey)
Hydrotreating has been the major way that refineries have achieved Tier III sulfur compliance. Pre-treating FCC feed improves yields and lowers sulfur in all products but has higher capital cost than post-treating. Post-treating is less capital intensive but reduces gasoline octane and also has no impact on FCC yields or the sulfur in other FCC products. Most new hydrotreating units for Tier III compliance were post-treating. If post-treating is used for sulfur reduction, it is beneficial to have multiple gasoline cuts. Excluding light FCC naphtha from hydrotreating will reduce overall octane loss as most olefins are found in the light FCC naphtha. For refineries with existing feed hydrotreaters, some have increased severity to comply with Tier III leading to a shorter catalyst cycle length.
The most significant FCC operational change to reduce gasoline sulfur has been to reduce the gasoline cutpoint. This removes the fraction of gasoline with the highest sulfur concentration. Additionally, some FCCs have been able to reduce gasoline sulfur by increasing conversion.
The quality of the gasoline / LCO fractionation can also have significant impact on gasoline sulfur. Benzothiophene boils at 430 degrees F. For units that are under-cutting gasoline, benzothiophene should be in the LCO stream. For one refinery, over 60% of the gasoline sulfur species were benzothiophene and alkyl-benzothiophenes. A fractionation improvement project would shift a significant amount of these sulfur molecules to LCO where they belong.
Sulfur reduction additives have been used to reduce FCC gasoline sulfur by 15-30%. Many gasoline sulfur reduction additives utilize technology that saturate thiophenes to tetrahydrothiophenes which are cracked to H2S and non-sulfur containing hydrocarbons. Gasoline sulfur reduction additives are usually less effective when the base catalyst contains high rare earth and high vanadium or when the feed contains previously cracked hydrocarbons. For most refineries, gasoline sulfur reduction additives are not the sole answer for Tier III compliance but rather are used in conjunction with other strategies.
Finally, sulfur credits have also been used to meet Tier III compliance. These credits can be from other refineries in the corporation or can be purchased in the open market.
COLIN BAILLIE (W. R. Grace & Co.)
Refiners have various options to comply with Tier 3 regulations for gasoline sulfur, though post treatment of FCC gasoline lies at the heart of many refiners’ strategies. As there are limited capital projects for new installations relating to Tier 3 that have been announced, it would appear that many refiners have opted to revamp, expand capacity, or simply increase the severity of existing hydrotreaters. Post-treatment of gasoline can involve various process configurations, including Selective Hydrogenation Units, Splitters, and Hydrodesulfurization units (HDS), to best fit the gasoline pool requirement and maximize refinery profitability. The downside of post-treating gasoline is the resulting loss of octane. This can be alleviated when pretreating feed upstream of the FCCU through FCC feed hydrotreaters and/or hydrocrackers, which allows the HDS unit to run at lower severity.
In terms of FCC operational changes, if the unit is equipped with a splitter the LCN cut point can be decreased to reduce the LCN sulfur, which is typically blended directly into the refinery gasoline pool. However, this results in a higher volume of HCN to the HDS section, which increases hydrogen consumption. Another option is to reduce the overall FCC gasoline cut point. This can significantly reduce the sulfur content of the FCC gasoline, and therefore allow a lower severity of post-treatment with benefits for octane preservation. However, gasoline yield is reduced, and the increased yield of LCO requires available distillate hydrotreating capacity and increases hydrogen consumption.
Switching to lower sulfur feeds can allow refiners to more easily achieve gasoline sulfur targets, though many refiners may not have the flexibility to do this. However, the ramping up of IMO 2020 legislation, combined with a lightening global crude slate, could be additional drivers for refiners to move to a lower sulfur crude diet, which would also have benefits for Tier 3 compliance.
Another way refiners in North America have approached Tier 3 is through the use of sulfur credits. EPA’s sulfur credit program allows refiners to accumulate credits by achieving lower sulfur levels than required. The credits can then either be “cashed in” at a later date to offset higher sulfur levels or be sold to other refiners who are struggling to meet the 10-ppm sulfur target. For example, Tier 2 credits generated prior to 2017 (by achieving less than the required 50 ppm gasoline sulfur) are being used by some refiners to bring them within Tier 3 compliance, though these Tier 2 credits are only valid until the end of 2019. Refiners can continue to generate credits by achieving less than 10 ppm sulfur, which are valid beyond 2019. However, due to the difficulty of achieving <10 ppm sulfur, significantly less Tier 3 credits will be generated compared to Tier 2 credits. With a low supply of credits, and an expected high demand, this is likely to be a very costly strategy for refiners to rely on.
Finally, FCC gasoline sulfur reduction catalyst and additive technologies can be used by refiners to complement the strategies described above. Grace ‘s GSR® technologies include D-PriSM® and GSR® 5 additives, as well as SuRCA® catalysts, and have been used by more than 70 refineries worldwide to successfully reduce gasoline sulfur. Many of these applications have included heavily hydrotreated feedstocks where the FCC gasoline sulfur is already low reducing or eliminating the need for post treatment. The lower gasoline sulfur obtained with GSR® technologies can allow a reduction in FCC gasoline post-treatment severity, thus minimizing octane loss and reducing hydrogen consumption. They also allow a reduction in the undercutting of FCC gasoline, resulting in increased volume while maintaining overall sulfur level. They can also be used to help generate additional sulfur credits, for example by allowing the LCN cut point to be extended without increasing sulfur. Finally, GSR® technologies allow higher sulfur feeds to be processed, which improves feedstock flexibility.
GEORGE HOEKSTRA (Hoekstra Trading LLC)
Tier 3 puts high value on reducing octane loss in FCC gasoline desulfurizers. It is possible to reduce octane loss by making unit changes that reduce olefin saturation at constant product sulfur. An effective way to do this is to add a feed splitter, which can be done with low capital investment. This also provides opportunity to adjust the light/heavy naphtha cut point to optimize desulfurization/saturation selectivity.
Our pilot plant, laboratory, and field testing has shown that adjusting the heavy naphtha end point can also improve selectivity by driving heavy sulfur and/or octane-enhancing aromatics into or out of the gasoline.
These cut point optimization steps are strongly dependent on feed composition and are facilitated by use of detailed hydrocarbon type analysis and sulfur speciation data.
Other operating variables also affect sulfur/octane selectivity in ways that can be measured with modern analytical methods and adjusted beneficially. We describe unit performance with a performance curve showing RON octane loss versus product sulfur. Here is the performance curve for one commercial unit that doesn’t have a feed splitter:
This next chart shows our estimate of how this curve can be shifted up by the combined effects of
1. adding a feed splitter
2. optimizing feed end point
3. employing operating conditions that improve sulfur/octane selectivity.
In this case, the combined effect saves 4 RON octane at 10 ppm product sulfur, which is more than enough to pay for a feed splitter and LOTS of laboratory analyses.
These optimization tools, and the data on which they are based, are available to anyone at negligible cost through Hoekstra Trading.
Question 1: What are the benefits of alumina treating in sulfuric acid alkylation and HF alkylation? Has this technology been proven commercially?
CHRISTIAN ARNOUX (Valero)
The opinions expressed are those of the author and do not necessarily represent the views of Valero Energy Corp.
-
Typically, all alkylation units treat the effluent to remove free acid and/or contaminants
-
Fluorides and Sulfates are the typical contaminates removed from HF and Sulfuric acid units, respectively
-
Activated alumina is commonly used in HF service
-
Caustic treating is common in sulfuric acid units but dry treating using alumina is also used
LIZA PACHECO (STRATCO)
In the sulfuric acid alkylation process, droplets of acid are found in the effluent stream from the reactor. These droplets, which are formed primarily from the alkylation chemistry itself and the formation of intermediates, are small and stable and cannot be removed by gravity settling and / or coalescing alone. Traditionally, a caustic/alkaline water system has been used to remove the acidic droplets from the effluent stream through neutralization. Consequently, the water that is introduced in this section travels to the reaction section where it will dilute the acid and increase corrosion. When activated alumina is used, the acid droplets are removed by the chemical reaction of the acid with the activated alumina. No water is introduced into the system which will be beneficial due to the decrease of acid consumption and corrosion in the treating and reaction sections. Refiners have reported longer Contactor™ tube bundle life when operating a dry system using alumina compared to the caustic/ alkaline water effluent treating system where water is introduced into the process and recycles back to the reaction zone. While several STRATCO® alkylation units have operated with dry alumina treating for many years, only in the last few years has this been part of the standard STRATCO® alkylation offering. As part of this transition, DuPont has made a commitment to research for continuous improvements to this process.
KURT DETRICK and TROY SMALL (UOP)
Alumina and promoted alumina products are commonly used in HF alkylation to remove organic fluorides from the propane and butane product streams. The benefits of using alumina to remove organic fluorides from the product streams include prevention of corrosion in downstream fractionation towers, prevention of catalyst deactivation in downstream catalytic units (such as Butamer) and making the propane safe for in-home use (such as in stoves and heaters). While both alumina and promoted alumina, products have been commercially proven, the industry trend is towards promoted alumina products, which generally have a higher fluoride capacity than non-promoted alumina products.
Alumina treating in sulfuric alkylation units is not common.
Question 2: What metallurgy works well and does not work well for use in alkylation units? In what applications does the alternate metallurgy perform better/worse?
LANCE TALLMAN (CITGO Petroleum)
HF
The standard metallurgy for use throughout HF alkylation units is carbon steel. Showing a high degree of corrosion resistance in both low and very high acid concentrations, carbon steel is also useful for its relatively low cost compared to more exotic materials. In most applications throughout the unit, carbon steel will display a general corrosion loss rather than the more difficult to detect pitting or localized corrosion. However, it must be noted that when used in a stream that will contain HF acid, there are several restrictions that are placed on the steel composition (particularly Cr, Ni, and Cu) in order to try and reduce the unique circumstances that can set up localized areas of accelerated HF corrosion. Despite the possibility of accelerated corrosion, carbon steel has a long history of suitable use throughout Alkylation Units throughout the country and performs well in most streams present in an Alkylation Unit.
There are some streams that, through use and time, have been found to require an alternate metallurgy to the standard approach of carbon steel. These areas typically involve a higher temperature than are encountered in most of the unit (most refiners using carbon steel for HF containing streams at operating temperatures below 150 – 200°F). At temperatures higher than this, or in areas that previous experience with carbon steel has provided unsuitable corrosion resistance, consideration is made for employing the use of a high nickel alloy. The common choices for use vary between Monel 400 and various Hastelloy’s (C-276, B grades, etc.), with Monel getting the preponderance of use. These high nickel materials provide suitable corrosion resistance at the higher temperature operation than are possible with carbon steel. Monel 400 brings about its own set of challenges with the possibilities of cracking due to high residual stresses, particularly in services that contain oxygen or mercury contamination. A stress relieving heat treatment is usually performed at initial fabrication to reduce the possibility of this catastrophic failure mechanism.
Most notable of the common materials used in refining that should not be employed in HF Alkylation units are stainless steels. While they can sufficiently resist corrosion in anhydrous environments containing HF acid, they perform worse than carbon steel under aqueous conditions. All grades of stainless steel can undergo rapid corrosion losses (sometimes on the order of 100 mpy) at certain conditions and should therefore be avoided. Other materials to avoid are those that contain silicon, as this will react with HF acid and form a highly corrosive fluorosilicic acid.
Sulfuric Acid
Carbon steel is used for most of the equipment and piping in sulfuric acid alkylation units including the reaction zone (contactors & acid settlers). However, CS doesn’t work well in the presence of sulfuric acid with high temperatures and/or high velocities. The fresh and spent acid piping is typically constructed of 300 series SS, and/or Alloy 20; however, some units have successfully operated with CS piping in this service. Additionally, in the effluent treating section & depropanizer feed treatment system, the static mixers and upstream/downstream piping are typically upgraded to Alloy 20 and/or Hastelloy C. Although the contactors (shell & tube bundle) are constructed of CS, due to the highly turbulent service, the internals such as the reactor impeller & distributors are upgraded to Hastelloy C which will significantly extend the life of this equipment.
Monel is not recommended for material of construction in sulfuric acid alkylation units as it is not compatible with equipment and piping containing large amounts of sulfuric acid.
KURT DETRICK (UOP)
API RP 751 has a good description of the experience with different materials in the HF Alky unit in Section 3 and Annex D.
Carbon steel is the most common material used in HF Alky units in areas of relatively low temperature and low water content in the acid. The surface of fresh steel reacts with HF to form iron fluoride, but under the proper conditions (primarily low water concentration in the acid), this layer of iron fluoride protects the underlying steel from further attack.
Monel is used in areas of the HF Alky unit where temperatures are higher, or water content of the acid is higher (such as in the Acid Regenerator or Rerun tower). Hastelloy (primarily C276) has also been used successfully in these areas of elevated temperature and/or water concentration.
316 Stainless steel vessels are used for transporting fresh anhydrous HF to the Alky unit, but stainless steel should only be used for high purity fresh acid. In commercial experience, stainless steels (both Austenitic and Martensitic) are aggressively attacked by the plant acid in an HF Alky unit and these stainless steels should never be used in the HF Alky unit itself.
ROMAIN LEMOINE (McDermott Lummus Global)
In less than 7 years, McDermott’s Lummus Technology has successfully commercialized and licensed an advanced version of sulfuric acid alkylation technology, called CDAlky®, to clients worldwide. CDAlky technology focuses on effectively eliminating the root cause of drawbacks inherent to conventional alkylation units. A leading area of concern is excessive corrosion and the need for higher-grade metallurgy.
Lummus’ CDAlky Technology not only operates at lower temperature than conventional sulfuric acid alkylation processes, but it also eliminates the need for a reactor effluent post-treatment section. The key to these technical breakthroughs resides in CDAlky reactor proprietary internals: AlkyPak™ & Distributor Plates. By eliminating caustic and water introduction in the alkylation section, and by maintaining a low temperature operation, CDAlky technology can eliminate the need for higher-grade metallurgy. Recent turnaround inspections conducted on multiple CDAlky units have confirmed these technical steps out benefits:
-
Low Temperature operation reduces significantly the corrosion rates.
-
Neither reactor effluent wash nor post-treatment steps are required to remove sulfuric acid or sulfates from the alkylate.
-
No fouling material is observed in any fractionation reboilers.
-
Stainless steels and carbon steels are suitable material of construction.
GREG TROUTMAN (DuPont)
Although careful consideration is required when selecting appropriate metallurgy for any sulfuric acid alkylation unit, a sound process design relegates the recommended use of exotic material to only a few areas. Carbon steel is by far the most common material found throughout the unit as it is both economical and capable of handling the range of temperatures and velocities observed in normal conditions as well as most alkylation unit excursions. The typical hierarchy of metals found in a sulfuric acid alkylation unit is as follows:
Carbon Steel < 316L Stainless Steel < Alloy 20 (in certain situations) < Alloy C276
With respect to sulfuric acid, carbon steel can be used if the velocity/turbulence through the piping or equipment is not high enough to disturb the protective passive layer of iron sulfate that forms on carbon steel when contacted with sulfuric acid. Many refiners have utilized carbon steel with a corrosion allowance of either 1/8” or 1/4” for normally flowing and normally not flowing lines, respectively. In piping where, high velocity/turbulence cannot be avoided such as through valves, Alloy 20 or PTFE-lined valving should be used to avoid excessive corrosion/erosion. When velocities high enough to avoid hydrogen grooving and low enough to avoid eroding the passive layer are not obtainable with sulfuric acid piping, 316L stainless steel is a good substitute.
Areas where neutralization takes place will require at least Alloy 20, especially since most of the neutralization takes place in a static mixer where there is high turbulence. It should be noted that while Alloy 20 is an excellent material in many applications, situations such as high temperature and certain contaminants or environments can cause it to become active and fail quickly.
For the reactor itself, carbon steel is the preferred choice for the bulk components (shell, heads, tube bundle, etc.). Carbon steel is economical where the process conditions (velocity, turbulence and temperature) are typically mild. Since the carbon steel within the reactor forms a protective passive layer, a long life is typical. The reactor impeller, wear ring, feed nozzles and other high velocity/turbulent areas can be constructed of Alloy C276 to dramatically improve the life span.
DuPont never recommends the use of Alloy 20 within a sulfuric acid alkylation reactor due to process conditions that can cause the metal to become active and fail. In addition, areas where neutralization takes place with elevated temperatures such as the mixing of sulfuric acid and hot alkaline water are better suited for Alloy C276 rather than Alloy 20.
A few additional considerations:
For areas with high amounts of propane such as the depropanizer overhead, depressurization could result in temperatures cold enough to require low temperature carbon steel.
Carbon steel is suitable for caustic (typically in the 10-12% range) but post weld heat treating should be used in areas where the temperatures are expected to be higher than 180 °F.
Both stainless steel and Alloy 20 should not be used when significant amounts of halide ions such as Cl- are present as these may cause pitting corrosion.
Water is not corrosive by itself, but it can cause major corrosion issues if there is an area with a high probability of contacting trace SO2 or sulfuric acid.
As for metallurgies that are not compatible in sulfuric acid alkylation units, Monel is not compatible with equipment and piping containing large amounts of sulfuric acid. However, Monel has been shown to be effective in hydrocarbon streams with weak acid. This is a benefit when considering HF to sulfuric acid alkylation conversion as Monel is prevalent in HF alkylation units.
Non-metallics such as PTFE, polypropylene, and glass have excellent corrosion resistance and can be considered for various portions of the plant. However, fire resistance and physical strength must be also considered.
Corrosion is a complicated phenomenon and is not always predictable or even explainable by current metallurgical knowledge. DuPont continues to experiment and gain knowhow on the performance of specific materials in sulfuric acid alkylation units. For the latest information, DuPont and/or a metallurgist should be involved in corrosion evaluations and upgrade decisions.
Question 3: What type of release mitigation safety systems do you use for sulfuric acid and HF alkylation units?
LANCE TALLMAN (CITGO Petroleum)
HF
-
Rapid Acid De-Inventory systems to move HF acid from unit to remote acid storage drums
-
Acid Settler, reactor risers and coolers (includes unit shutdown)
-
Acid Storage Drum
-
Water Curtain surrounding high acid area of unit (with auto-activation)
-
Water deluges on Acid Settler, Remote Acid Storage Drums and acid-service pumps
-
Elevated, remotely operable fire monitors with independent supplemental water supply
-
Camera systems to improve response time
-
Point source hydrocarbon detectors
-
Point source HF detectors
-
Bi-level perimeter laser HF detectors
-
Remote capabilities to start, stop, isolate, vent to flare and deluge acid-service pumps
-
Remote control panel for acid truck unloading (isolation valves)
-
Closed circuit monitoring of unit
-
Sulfuric Acid
Sulfuric acid alkylation units do not require the robust safety systems as the HF Alky units since sulfuric acid is a liquid at atmospheric conditions. The release mitigation safety systems typically used are curbed and/or diked areas to contain the liquid sulfuric acid in event of a leak. This exists in the reaction section, blowdown section, and fresh/spent acid tanks. The same mitigation systems are in place in the effluent treating section and the depropanizer feed treating system where caustic is present. When Operators respond to any release, they’re equipped with the appropriate PPE, which include slicker suit, face shield, and proper gloves. In addition, hydrocarbon detectors are present in and around the unit due to the presence of LPGs.
SHANE PRESLEY (DuPont)
Sulfuric acid is a liquid at atmospheric conditions and will pool if released rather than form a vapor cloud. During the summer of 1991 an independent consultant performed a series of large-scale sulfuric acid leak tests. In all, thirty-six release tests were conducted, three tests utilizing alkylation equilibrium acid from an operating commercial alkylation unit and thirty-three tests using an acid/hydrocarbon emulsion. The tests involving equilibrium acid resulted in essentially 100% recovery of the acid released; the remaining tests resulted in an average 97.6% recovery of the acid released (Figure 1) [1].
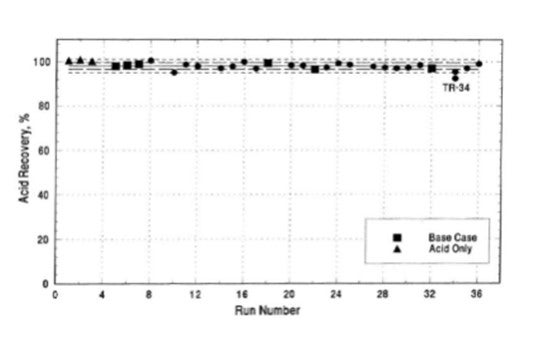
Figure 1: Sulfuric Acid Test Recovery
Because sulfuric acid will pool when released rather than forming a toxic vapor cloud, only basic mitigations are required. These mitigations typically include curbed and/or diked areas around sections of the unit containing sulfuric acid to contain the acid in the event of a leak. These areas typically include the reaction section, acid blowdown section, and fresh/spent acid storage areas. In the event of small or minor leaks, sulfuric acid can be neutralized with soda ash before removal. Larger spills may require physical removal of the acid. Other release mitigation systems in place are not specific for sulfuric acid and are typical for other processing units containing LPG hydrocarbons.