TSA Pipeline Security Directives Implementation
Implementation of the TSA Security Directives are in full swing---hear the latest on any implementation and compliance issues and the next steps for pipeline security.
Implementation of the TSA Security Directives are in full swing---hear the latest on any implementation and compliance issues and the next steps for pipeline security.
DHS’s Cybersecurity and Infrastructure Security Agency works closely with industry to help build a sector that can defend against both cyber and physical threats. During this session, CISA will speak to the future direction of the Chemical Facility Anti-Terrorism Standards (CFATS) regulations, CFATS reauthorization priorities, and insights from the first year of CFATS inspection audits. This session will also provide brief conference attendees on the current and future work of the Global Chemical Security Congress.
MELDRUM (Phillips 66)
Carbon monoxide can form in reformer units as the hydrocarbon reacts with moisture under very low-unit pressure conditions. Typically, semi-regeneration reformer net gas would have nil CO and only a minimal amount in a CCR-type unit. I expect it to probably be on the order of 5 ppm (parts per million), though some units report routine measurements of 10 to 20 ppm CO in their net hydrogen off gas.
One of our cyclic units that was operating at 400-pound had CO as high as 20 ppm in its net hydrogen stream when the recycled moisture rose to around 300 ppm. The excessive water entered the reformer from a leaking side reboiler on a wet debutanizer that used a slipstream of the reformer reactor effluent as the heat source. The water then returned to the reformer product separator. The high CO caused deactivation in the catalyst in a downstream isomerization unit.
Accurate measurements of CO in the net gas are difficult. Reformer units are not expected to have much CO, so they seldom have an online analyzer. A colorimetric tube – Gastec or Dräger type – can be used to give an indication of the presence of CO, but accuracy for a quantified number is difficult and requires the use of a carbon pre-tube to remove the hydrocarbons.
CO is detrimental to downstream hydrogen-using units in three principal areas. CO in hydrogen being fed to a distillate hydrotreater will methanate, consuming the hydrogen that would have otherwise been used for the desulfurization reactions. This will have the effect of lower catalyst activity. CO in hydrogen fed as a makeup stream to an isomerization unit will also methanate and form moisture that will deactivate the isomerization catalyst. CO that did not methanate in the second example could act as a poison to the platinum metal function of the isomerization catalyst. UOP suggests a CO limit of 1 ppm max for isomerization hydrogen makeup gas. My Answer Book response also includes some of the common steps used to minimize CO formation in reformer units, particularly in a CCR unit.
BULLEN (UOP LLC, A Honeywell Company)
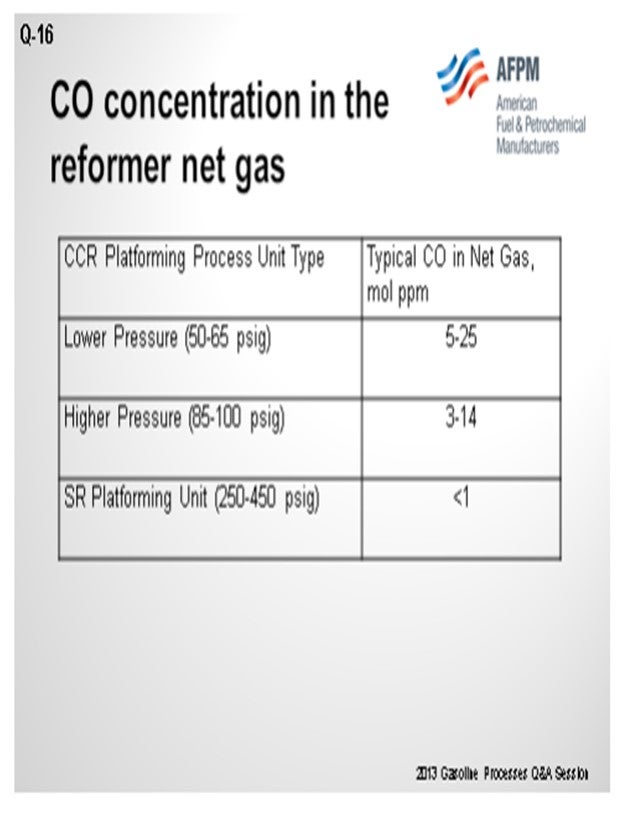
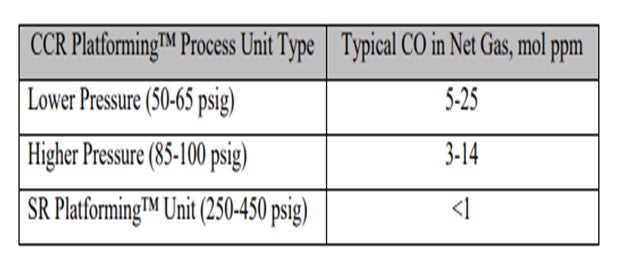
As you can see in this table, we have correlated some different types of operation and ranges of CO levels. As Craig alluded, the numbers vary quite a bit, which can be due to conditions in the unit, as well as analytical capabilities. There seems to be a trend that the lower pressure units generate more CO than higher pressure units.
The laboratory method we recommend using is UOP 603, which is a laboratory method for CO and CO2 and hydrogen. However, a lot of refiners cannot do this method. The gas detection tube route is fairly common. Our point of view is that with the gas detection tubes, if one carbon pre-tube is good, then two is better. So, we usually ask them to use two tubes instead of one to help eliminate the breakthrough of hydrocarbons that can make a false high value for CO.
As Craig said, the issue with chloride and alumina isomerization catalyst is that you will deactivate the catalyst. However, if you are using another type of catalyst, like the Par-Isom catalyst or zeolitic catalyst, the actual suppression you will get will be very dependent on what temperatures you are running. As you approach the 400°F temperature, you tend to methanize the CO in the first part of the bed. So CO tends to have less of an effect on the metal function of the isomerization catalyst and becomes more of an issue of activity suppression due to the water on the acid sites. The same would apply if you had a saturation unit with platinum catalyst. It would also behave in a similar manner to these higher temperature isomerization units.
R.K. (RICK) GRUBB (Chevron Products Company)
Another aspect needs to be mentioned for the lower pressure reforming units. You have to take into consideration your nickel carbonyl formation when you shut down a hydroprocessing unit that is using the reformer hydrogen. You may have to either swap the hydrogen source or think of another shutdown procedure that will ensure no nickel carbonyl formation.
CRAIG MELDRUM (Phillips 66)
CO is detrimental to downstream hydrogen using units for three principal reasons:
1) CO will methanate in HDS (hydrodesulfurization) units consuming hydrogen, which will take away catalyst activity.
2) Much of the CO will methanate in isomerization units, forming water that will deactivate the isomerization unit catalyst.
3) The non-methanated CO in the isomerization unit will poison the metal function of platinum on the catalyst.
Note: The UOP suggested CO limit on isomerization unit hydrogen makeup gas is 1 ppm (10 ppm for CO + CO2). UOP reports that CO levels greater than 6 ppm will not allow the isomerization unit catalyst to meet its cycle life guarantee.
The common steps to minimize CO formation in the reformer are:
• Minimize moisture in the system (feed water control and good regeneration drying),
• Minimize the last reactor temperature,
• Maximize the H2/HC ratio, and
• Minimize catalyst circulation rate in a CCR.
PATRICK BULLEN (UOP LLC, A Honeywell Company)
The CO concentration in reforming unit net gas can be impacted by a number of factors: system pressure, temperature, and moisture in the recycle gas, as well as the H2/HC (hydrogen/hydrocarbon) of the operation. Operating pressure has the most significant impact on CO production in a reforming unit. CO formation in reforming operation is produced via steam reforming of hydrocarbons:
H2O + CH4 ↔ CO +3H2
Thermodynamically, this CO formation reaction is more favorable at lower pressures. CO production is inversely proportional to the pressure squared. As such, a semi-regeneration reforming unit, being significantly higher pressure than typical continuous reforming units, will tend to produce less CO than a typical continuous reforming operation. Likewise, the lower pressure high severity reforming unit operation is more favorable for CO productions.
Commercial reforming net gas CO data from CCR Platforming™ process units can range from 1 to 40 mol ppm. The table below indicates typical ranges for various unit types. Typically, CO levels in the semi-regeneration reforming units are at trace ppm levels due to the high pressure and low-moisture range operation.
For testing of CO in reformer net gas, UOP recommends method UOP 603 for trace CO and CO2 in hydrogen. For CO in light gaseous hydrocarbons, analysis by GC is recommended. Analysis by gas detector tubes can also be considered for measuring CO at elevated levels when used with several carbon adsorbing pre-tubes.
CO in reforming net gas can have an impact on downstream users that may be sensitive to CO or H2O that may be formed due to the reverse steam reforming reaction, also known as the methanation reaction. In the case of a Butamer™ and Penex™ catalyst, water is a permanent deactivator. A typical rule of thumb is that 1 pound of H2O kills 62 pounds of Butamer™ and Penex™ catalyst.
For other types of catalysts, such as Par-isom™ Process and zeolitic isomerization catalysts that operate at higher temperature, the water generated from methanation of CO is a temporary activity suppressant. Platinum-based BenSat™ catalyst behaves similarly to these isomerization catalysts.
GARY HAWKINS (Emerson Process Management)
With respect to the second part of the question, the carbon monoxide content, as well as other components in the net gas of a naphtha reforming unit, can be measured with a variety of measurement principles depending upon the accuracy and reliability required, other species present that may interfere with a particular technology, and the expected range of concentration of carbon monoxide. These same comments apply to measuring other refinery gases, such as the net hydrogen and PSA (pressure swing adsorption) tail gas from steam reforming units for hydrogen production.
ADAMS (HollyFrontier Corporation)
HollyFrontier has two CCRs: one at Tulsa that was converted from a semi-regeneration reformer and one at Artesia. We have not done a catalyst change at the Tulsa refinery CCR, but we have done an online catalyst change at Artesia. Essentially, we just made up more catalyst and purged the catalyst out of the regeneration section. It took about four or five days to completely purge the old catalyst out and replace it with a new catalyst. The downside was that we spent more on catalyst; because as the new catalyst moved through the system, we ended up using 10 to 15% more in order to complete the purge.
BULLEN (UOP LLC, A Honeywell Company)
Based on our experience, we estimate that about 10% of the reloads have been on-the-fly-types of change-out, which equates to about three units a year. We have a procedure for how to do this. As Mark said, one typically has to use more catalyst than if doing a shutdown and change-out just to make sure all of the old catalyst is purged.
Some other issues involve catalyst handling. It can be difficult to get personnel to make sure they segregate the old catalyst from the new, resulting in accidentally putting the old catalyst back into the unit. So, a rigorous process is needed for that.
The circulation rate for the catalyst can be limited by the mechanical moving of the drums and the addition of the drums to the unit, so it may end up being necessary to change the operating conditions to accommodate the more-active catalyst to minimize the extra coke. As the new catalyst comes into the system, it generates a different quantity of coke than the old catalyst; so the operators must be aware that they have to go and adjust the regenerator conditions. One of the drawbacks of doing an “on the fly” type of change-out is that one cannot inspect the system; in particular, the reactors. If there are any issues related to the reactors, such as high ∆P (differential pressure; DP) or poor distribution of the reactor temperatures, those problem will still exist after changing the catalyst.
Finally, the density of the two catalysts – the old versus the new – can impact the circulation system: so be aware of those differences. Also, while oxidized catalyst can be used, this generally puts more stress on the regeneration system to dry out the extra water from the oxidized catalyst. Oxidized catalyst tends to have more water on it.
Finally, you tend to generate more fines when doing a “change-out on the fly”, so keep close attention on the dust collection system.
MELDRUM (Phillips 66)
One of our Cyclemax™ CCR units changed catalyst “on the fly” with such success that the next catalyst changes on another of our Cyclemax™ units will plan to use the “on the fly” method. Benefits include decoupling the catalyst change from a unit outage. The catalyst change mentioned used 106% catalyst to complete the changeover. I want to emphasize that you lose an opportunity to do some vessel inspections with the “on the fly” change. I also emphasize that dust formation is to be watched. In our experience, the reduction zone filter DP increased significantly, resulting in much more frequent changes of that filter. In my Answer Book response, I have provided some parameters used for the example I have referenced.
MARK ADAMS (HollyFrontier Corporation)
HollyFrontier has two CCRs in its system: Artesia and Tulsa. In Artesia, the CCR catalyst was changed while the unit continued to operate. The upside is that the unit continues to run. The downside is that 10 to 15% more catalyst is used as some fresh catalyst is purged along with the old catalyst due to mixing. At Tulsa, the catalyst in the CCR has not been changed since the unit started operation.
PATRICK BULLEN (UOP LLC, A Honeywell Company)
The basic principle of the “Change-out on the fly” procedure is that while the reforming unit continues more or less normal operation, used catalyst is continuously withdrawn from the unit and replaced with fresh catalyst. The used catalyst is removed from below the regeneration tower, allowing the coke to be combusted before unloading. The fresh catalyst is added into the catalyst circuit just below the withdrawal point using the normal catalyst addition system.
In recent years, UOP estimates that about 10% of all continuous reforming unit reloads have been done while the reactors continue to process feed and produce valuable products. The procedure can be implemented on all types of UOP CCR Platforming™ Process units (atmospheric, pressurized, and Cyclemax™ regeneration sections) and also many of the units designed by others. Using this procedure allows operators to capture the benefits of improvements in catalyst technology or to replace catalyst that is underperforming as a result of poisoning or operational problems without a complete shutdown of the reforming unit.
While the “Change-out on the fly” procedure is a simpler compared to a full unit shutdown, there are a number of issues that need to be considered in the planning stages.
• Catalyst handling, either of the fresh or used catalyst, is frequently the rate limiting step in the change-out procedure. The team responsible for catalyst handling will need to carefully plan how to safely and efficiently move and keep separate the drums of old catalyst and the new catalyst.
• While continuing to process feed and producing reformate and hydrogen, it may not be possible for the reactors to operate at design conditions. It is necessary to keep the coke level on the catalyst in a range that the regenerator can sufficiently burn off the used catalyst before it is removed from the unit. This may limit throughput and severity of reforming unit operations given the rate of catalyst circulation that can be supported by the catalyst handling arrangements. This is more likely to be an issue with larger units.
• The goal should be to execute the replacement of the catalyst as quickly as is safely possible so that unit operations with fresh catalyst can be optimized (with regard to feed rate, octane and product flow) and the system tuned to optimize performance with the new catalyst.
One of the significant drawbacks to replacing the catalyst in this way is that there is no opportunity for inspection of the reactor internals and the execution of maintenance on the reactor side of the unit. In particular, the use of the “Change-out on the fly” procedure is not recommended if there are any indications of catalyst flow problems or reactor internals’ damage (pressure drop or unusual distribution of reactor delta temperature). If there are unresolved issues with the internals, they will impact the performance of the unit with the fresh catalyst in the same manner as with the used catalyst.
There are several other issues related to “Change-out on the fly”:
1. Difficulties can occur when the density of the fresh and used catalysts are different. The unit circulation is calibrated on the used catalyst and care must be taken as the fresh catalyst goes to the regenerator and the mass through the regenerator changes. This may require changes to the regeneration section operation.
2. Changing out catalyst using oxidized fresh catalyst is generally not recommended, as there can be issues with increased water native to the catalyst along with the water generated by reduction. This increased load on the drying section of the regenerator can result in getting the Platforming™ unit wet.
3. There can be a tendency to generate more fines while doing a “Change-out on the fly”. The dust collector system will need to be closely monitored.
CRAIG MELDRUM (Phillips 66)
An example of parameters used for a change-out is as follow for a 35 kbpd (thousand barrel-per-day) Cyclemax™ unit with 200,000 lbs catalyst inventory and 3,000 pph (pounds per hour) catalyst circulation:
• The old catalyst is removed below the regenerator to minimize coke on the catalyst.
• Catalyst circulation is reduced to 50% of design.
• 106% of the catalyst inventory is changed-out at an average load rate of five drums per hour.
• Total personnel resources used are eight operators, 14 catalyst handlers, two crane operators, and two engineers.
• Logistics: 950 catalyst drums and 80 flow bins
SONI OYEKAN (Prafis Energy Solutions)
At my last oil refining company, we accomplished that catalyst replacement process successfully with the assistance of the catalyst/technology supplier. It is most important to get the catalyst and technology supplier technical experts and support on board early while the project is being considered as refiners do not frequently conduct “on the fly” catalyst replacements. Prior to initiating the “on the fly” catalyst replacement project, adequate planning is required with the assistance of the catalyst supplier, catalyst handling company, refinery technical service, and Operations, Maintenance and Environmental staff. The following are required:
• adequate catalyst supply,
• revised and updated catalyst on-stream replacement procedures,
• plans for reforming at moderate reforming severity process conditions to manage coke make for complete black burn operations,
• safe procedures for handling hot catalyst exiting the regenerator, and, of great importance,
• safe use of nitrogen.
The hot catalyst issue can be of greater importance depending on the type of CCR technology that the oil refiner is using. The oil refiner should also plan to potentially use 110 to 120% of system catalyst inventory as fresh catalyst since there will be some mixing of fresh and spent catalyst and since it will take a little more fresh catalyst to ensure complete spent catalyst replacement. The money invested in the CCR catalyst is not a significant issue as the unused catalyst would be used as makeup catalyst over the course of reforming operations.
Having written that, I must confess that I am not a strong advocate of “on the fly” catalyst replacement for a number of factors: I fully understand when refining business conditions call for and drive the need to use “on the fly” catalyst replacements. However, I am strongly in favor of the standard complete catalyst replacement.
The “on the fly” catalyst replacement option does not provide the opportunity to conduct an appropriate reformer turnaround to permit full inspections and necessary repairs in the reformer reactors, catalyst transfer equipment, and regenerator sections. In addition, it does not permit inspecting and conducting necessary repairs to the chloride reduction equipment and system for meeting RMACT 2 regulations for regenerator vent gas. Sometimes the application of the “on the fly” catalyst replacement could encourage the refiner to operate without a turnaround for years longer than that recommended with consequent post-“on the fly” catalyst replacement reformer and regenerator performance and reliability problems that, over time, could negate any economic benefits realized via application of the “on the fly” catalyst replacement.
BULLEN (UOP LLC, A Honeywell Company)
The obvious answer to the high-pressure drop would seem to be that you have too many fines on the bags. But realistically, why do you get too many fines? The causes could be that you waited too long to do the reverse jet blowing or, a less common cause, that you are getting hydrocarbon carryover which is causing the fines to clump together on the bags. We recommend doing the blowback procedure at five inches of water ∆P. You also need to confirm that you have the right velocity, so you do not get too large of a blowback and then cause even more fines to occur by fluffing the material in the filter.
Some other causes for high fines could be too high elutriation velocity. There has been experience with the wrong filter elements being used, and that will cause problems.
We do have some experience with continuous blowback systems. However, one caution is that you can actually overpressure the system with continuous blowbacks. You have to watch for that. Also, you tend to blow off the permanent fines layer that accumulates on the bags which helps trap some of the bigger particles and improves the efficiency of the bags. So, watch for that as well. Some of these fine particles that can be blown off will also plug some of the holes in the bags. So, if you are going to do continuous blowback, you should actually check with your vendor for recommendations when using that kind of system.
PATRICK BULLEN (UOP LLC, A Honeywell Company)
In UOP’s experience, the most common cause for high differential pressure (DP) across CCR Platforming™ process unit dust collectors are too much fines accumulation on the filter elements. There are a few possible causes of extra fines accumulation on the filter elements; however, the most probable causes are waiting too long to reverse jet the filter elements or hydrocarbon carryover from the catalyst collector. As fines are collected on the filter elements, the DP across the dust collector will start to increase. Once the DP reaches about 5 inches of H2O, the reverse jet blowback system should be commissioned. Prolonging the period between blowbacks will allow more fines to accumulate and create a stronger “fines barrier”. The barrier can eventually get too large and prevent the blowback system from effectively removing the bulk of accumulated fines. UOP recommends that the reverse jet system be initiated whenever the DP across the dust collector approaches 5 inches of H2O. Furthermore, if hydrocarbons are carried over to the dust collector system they can condense on the bags causing fines to stick and cake up on the bags leading to high DP. Other causes for high dust collector DP include inadequate reverse jet pulse time, low N2 blowback pressure, too high of an elutriation velocity (which causes more dust make and an overload of the dust removal system), and even using the wrong type of filter elements. The dust collector vendor should specify the type of filter elements that should be used based on UOP current felt material type bags specifications. UOP no longer specifies sintered metal filters.
There have been some refiners using continuous blowback. Typically, it can help with reducing the DP across the filter bags. However, it has also been known to cause lifting problems when the lift system is overpressured with nitrogen. Operating with a continuous purge could also remove the “permanent” fines layer that is present on the filter bags. The fines layer increases the filter efficiency by plugging off some of the bigger holes of the filtering elements. Removing the fines layer could free up some of the bigger holes that could allow dust to pass through to the lift system. Furthermore, continuous blowback could result in creating microscopic holes (due to the constant back pressuring) in the dust collector bags that could result in dust passing through to the lift gas blower and lift system. UOP recommends checking with the dust collector vendor before moving to a continuous blowback operation.
BULLEN (UOP LLC, A Honeywell Company)
There are two ways to address the issue: 1) try to increase the coke that you are making and 2) adjust the regenerator. It would be great if you could just adjust how much coke you are making. However, most of the time you are restricted in what you can do with the unit, given your octane targets and the feed flows and qualities are fixed. The people who do have some adjustment ability do so by lowering the hydrogen-to-hydrocarbon ratio. You can do that to some extent, but you must make sure you have sufficient hydrogen to lift the liquid up through the CFE (combined feed exchanger) to be evenly dispersed within the exchanger. Again, depending on whether you are using Texas towers or a Packinox™ will determine how much you can drop the ratio. It varies depending on the design. We have noted that most CCRs are able to get down to about 30% of the normal coke-burning capacity. However, each CCR is individual, in terms of type, regarding what you can do to get down to that level.
In general, UOP recommends trying to keep the regenerator in continuous white burn mode. That will help both save utilities and keep the yield structure and conditioning of the catalyst proper. In general, the regenerator is limited by low coke operation, but you can reduce catalyst circulation. You can change the burn zone targets, but you must monitor the temperature profile. Finally, depending on the design, you can vent more excess air to maintain the drying of the catalyst in the bottom of the regenerator. For individual units, you can contact your UOP representative who can talk to you in more detail. We also can provide engineered solutions for you to modify your unit, so your regenerator burns less coke.
There are levels of coke make where you just have to shut down the regenerator. When you do that, we recommend that you continuously circulate the catalyst while the regenerator is shut down to ensure that you are able to start back up without any plugging (e.g., catalyst circulation issues) and so that you will evenly lay down the coke across all the catalyst.
ADAMS (HollyFrontier Corporation)
Our experience in Artesia is that when we have gotten into those modes, we reduced the hydrogen recycle and lowered the pressure. So it is that hydrogen-to-hydrocarbon ratio management.
PATRICK BULLEN (UOP LLC, A Honeywell Company)
There are two ways refiners can approach low coke operations: increase unit coke make or adjust the regenerator operation to burn less coke. Unfortunately, in most cases units have little control over trying to increase their coke make. The “typical” ways to try to increase coke on catalyst are as follows:
• Increase feed rate,
• Increase octane,
• Use leaner feed PONA (paraffins, olefins, naphthenes, and aromatics) (more paraffinic),
• Use higher feed endpoint,
• Lower average reactor pressure, or
• Lower reactor H2/HC.
This list may look like there are many options; but in reality, a refiner may not have control over many of these items. Typically feed rate, target octane, and PONA are dictated by the Planning department and cannot be easily changed. The engineer can request a higher feed endpoint, but that is subject to refinery economics and is only adjustable up to a certain point because it can effect reformate endpoint and color. Lowering the average reactor pressure significantly would likely require a unit revamp. The best tool an engineer has to try to increase coke make is to reduce the recycle gas H2/HC ratio. This will also have an added benefit of utilities savings (lower compressor load). However, the lowest compressor flow rate is often associated with the flow rate required to properly distribute the liquid feed in the CFE.
The area where the refiner has more options to manage low coke operations is on the regenerator side. CCR Platforming™ process unit regenerators are designed to burn catalyst with 5.0 wt% (weight percent) coke at the design circulation rate (e.g., 1000 pph CCR section is designed to burn 52 pounds of coke per hour). The low side of the design range is approximately 60% of design coke load; however, most regenerators can operate well below that 60% point (some as low as 30%). UOP recommends keeping the regenerator in continuous white burn operation for both utilities savings and proper reconditioning of the catalyst. White burn is the normal operating condition for the UOP CCR Platforming™ process unit regenerator. Continuous regeneration in white burn allows the air and chloride injection to be sent to the normal locations rather than the startup or black burn locations.
Each style of CCR Platforming™ process unit regenerator has its own limitations in low coke operation, but the typical responses to low coke are as follows:
• Reduce catalyst circulation. – Provides additional residence time in the burn zone to reduce residual coke,
– Provides additional residence time in the chlorination zone to maintain optimal platinum dispersion,
– Raises drying air to catalyst ratio to 5 scf (standard cubic feet) gas/lb cat (0.3 Nm3 /kg-cat) (Normal meter cubed per kilogram of cat), and
– Helps keep recycle gas moisture low. Lower circulation means less catalyst is being reduced.
• Reduce burn zone oxygen setpoint and monitor temperature profile closely.
• Vent more excess air, if possible.
– Maintain minimum air heater flow rate and
– Maintain drying air to catalyst ratio to 5 scf/lb cat (0.3 Nm3 /kg-cat).
Please contact your UOP representative for specific information related to the potential limiting factors for each style of regenerator and responses to low coke operation.
Regarding extremely low coke make, in some instances, Platforming™ unit coke make is so low that it is not practical to keep the regenerator running in white burn. In this case, it is recommended that the catalyst be continuously circulated until sufficient coke is laid down to restart the regenerator.
MARK ADAMS (HollyFrontier Corporation)
Artesia: The recycle hydrogen is turned down and pressure reduced to minimum during low severity operation to enhance coking and avoid shutting down the regeneration system.
Tulsa: The CCR at Tulsa has not been in a low-coke operating mode since startup. The alternatives are to reduce pressure and hydrogen recycle to the extent possible and try to maintain catalyst circulation. Otherwise, the regeneration system can be shut down for three to four days and then restarted.
SONI OYEKAN (Prafis Energy Solutions)
If I believe that this question was addressed in a previous AFPM or NPRA Q&A conference. A number of changes in process conditions and naphtha characteristics type suggestions were provided for improving low coke naphtha reforming operations in CCR units. More recently, improved design enhancements of the regenerators have also been offered by a process technology licensor. Those who have limited naphtha reforming productivity, due to low coke naphtha reforming in their CCR units, and wish to assess some of the mechanical designs for improved CCR operations should consult their technology provider.
As a review, typical past suggestions for managing low coke naphtha reforming included the following:
(a) Manipulation of reforming process variables via changes in the octane severity, recycle gas H2/HC molar ratio, LHSV (liquid hourly space velocity), and reactor pressure;
(b) Changes in naphtha feed characteristics: paraffinic naphtha, naphtha N+A quality, naphtha feed EP, and light ends in naphtha feed; and,
(c) Processing schemes to manage intermittent regenerator operations and exercising special care to avoid catalyst and regenerator screens damages.
Process and catalytic developmental studies continue on innovative approaches for improving refiners’ low coke naphtha reforming operations. Stay tuned as I expect some useable innovative solutions soon.
CHRIS STEVES (Norton Engineering)
With changing specifications for gasoline (lower benzene, lower octane demand) and cheaper sources of hydrogen (natural gas reforming), many refiners now find that their reforming units are operating well below nameplate capacity. With low feed rates (and sometimes lower endpoint naphtha as well), the unit will produce lower coke and the regenerator may not be able to continuously operate at the low coke levels (control of oxygen in the regenerator can run up against turndown limits). If this is the case, the regenerator circulation can be stopped and coke levels allowed to build so that a good burn can be obtained when the circulation is restarted. Calculation tools to predict the coke on catalyst are recommended so that the increase in coke on catalyst can be tracked and regenerator circulation restarted at the appropriate time (within regenerator coke burning capacity).
STREIT (KBC Advanced Technologies, Inc.)
First, there are not a lot of innovative solutions here on this list. For the most part, everyone is doing many of these things already. They are fairly straightforward. First, just maximize the amount of gasoline you are dropping in the distillate. The amount of gasoline that you can drop into distillate is usually constrained by the 10% point of the diesel, cetane, or flash point. It can also be constrained by the drivability index of the gasoline.
The next typical way of trying to upgrade gasoline is to reformulate the FCC catalyst to get more distillate selective yield off of the FCC and “unmake” the FCC gasoline in favor of distillate. Another path is actually to raise severity on the FCC and crack the gasoline into olefins. Obviously, depending on whether or not you have an outlet for those olefins will determine if that makes sense.
You can also do aromatics extraction. Generally, that is done on reformate, but it can be done on other streams. If you have an outlet for the aromatics, you can reduce your gasoline. Obviously, that will impact the octane on your gasoline, so you will have to make sure that it is in balance.
Finally, you can reform FCC gasoline by sending it to a reformer. There are a number of refiners who do that, at times. This step will convert some of that FCC gasoline into aromatics that you can recover as product.

ERIC STREIT (KBC Advanced Technologies, Inc.)
The first processing option, and one that most refiners are trying, is to make sure as much of the gasoline product as possible is shifted into distillate products through cutpoint adjustments. The maximum amount of naphtha that can be dropped into distillate is usually set by the jet or diesel product flashpoint or 10% point. Gasoline drivability or other specifications may also limit the amount of heavy gasoline material that can be shifted into distillate.
Because most of the gasoline in a U.S. refinery is usually made from the FCC, trying to reduce the FCC gasoline make is the next logical step. The first place to look for reducing FCC gasoline is usually catalyst formulation. Replacing catalyst with distillate selective catalyst, which is available from most catalyst suppliers, will help shift yields away from gasoline and toward distillate. Of course, this creates a new problem: what to do with the increased LCO production. Because LCO is a low-cetane blendstock, the amount that can be blended into diesel or diesel hydrotreater feed may be limited by the final product’s cetane specification. LCO is also a huge hydrogen consumer in a diesel hydrotreater, so it may be difficult to process in units that are not designed for high hydrogen makeup rates.
Raising FCC severity will shift gasoline yield toward light ends, increasing C3 and C4 olefins that can be sold as product or alkylated. This change in operations will also increase ethylene yield, which may be another product that can be recovered. While the olefin yield is increasing, so is the production of low-value saturated gases; so, any changes in FCC operation should take into account the downgrading of product into fuel gas, as well as the upgrading of product into olefins.
Aromatics extraction will reduce gasoline production. Most refiners who make aromatics extract the aromatics from reformate. However, heart-cut FCC naphtha can potentially be sent to an aromatic's extraction unit as well.
FCC gasoline can be reformed, as well. Refineries with an aromatic's outlet can hydrotreat the FCC gasoline to make reformer feed and then convert that material to aromatics. The lightest portion of the FCC gasoline does not make very good reformer feed and it is very high in olefin content so any refiner considering reforming FCC gasoline should look at splitting the material beforehand and only reforming the heavier portion.
Another process that is currently available is purported to convert the olefins in the FCC gasoline to aromatics. The byproducts of this process are LPG and fuel gas.
STREIT (KBC Advanced Technologies, Inc.)
The typical olefin limit in a C5/C6 isomerization unit is 2 to 3%. The olefin content of the feed is constrained by the high heat release that the olefins cause across the reactor. Olefins can polymerize and cause coking on the catalyst in an isomerization unit. This is typically more noticeable in isomerization systems that are once-through hydrogen because there is less hydrogen in the system.
With regard to the molecular sieves on the frontend of the unit, during normal operation, the olefins are not that much of an issue. Problems arise at the high temperatures experienced in regeneration of the molecular sieves. Those olefins that are on the sieve can polymerize at the high temperature and shorten the life of the filters or of the molecular sieves.

MELDRUM (Phillips 66)
My comments are similar to Eric’s. The limit is usually based on hydrogen consumption, heat release, and the feed dryers. Olefin saturation consumes hydrogen that would be needed for the isomerization reactions. So as the level of olefin increases in the isomerization unit feed, the isomerization unit reactions in the catalyst stability can be hindered. The exothermic temperature rise as olefins are saturated can compromise equipment design and increase the reaction temperatures to a suboptimal equilibrium range. Recommended reactor temperature limit is 400°F for both the C4 and the C5/C6 isomerization units. I agree with Eric on the molecular sieve deposits that can form primarily due to the higher temperatures used in the regeneration.
BULLEN (UOP LLC, A Honeywell Company)
I concur with Eric and Craig. The only additional comment I have is that the olefin is a double-edged sword on the dryer sieve. Not only does it reduce the life; we expect about 50% reduction in life for a number of regeneration cycles compared to non-olefinic feeds. That is probably down in the 200- to 250-regenerations range. In addition, when the sieve sees olefins, it will preferentially absorb them, resulting in pushing off some of the other contaminants; so you tend to have to regenerate the drier more frequently due to a reduction in delta loading. Not only do you get less life, but you also have to regenerate more often. It all becomes an economic decision, trading sieve life for catalyst life.
CRAIG MELDRUM (Phillips 66)
Isom feed olefin limits are defined by hydrogen consumption, heat release, and impact on feed dryers. Olefin saturation consumes the hydrogen needed for the isomerization unit reactions; so as the level of olefin in the feed increases, the isomerization unit reactions and catalyst stability can be hindered. Exothermic olefin saturation can compromise equipment design and increase reaction temperature to suboptimal equilibrium ranges. The recommended reactor temperature limit is 400°F for both C4 and C5/C6 isomerization units. Olefins can form coke in molecular sieve dryer material, limit the ability of the mol sieve to remove sulfur from the feed, and shorten dryer life by 50 to 75%.
UOP suggests limiting Butamer™ feed total olefins to 0.1 wt%, isobutylene to 100 ppm, and diolefins to 10 ppm. Penex™ olefin limits are suggested to be 2 wt% and Bromine number limited to 4. Typical NHT feed has a Bromine number of 0.1. The limit of 4.0 is to allow for sulfolane raffinate or reformer splitter stream to be routed to the unit. The volume percent olefin is approximately half the Bromine number.
PATRICK BULLEN (UOP LLC, A Honeywell Company)
In general, the olefin content to an isomerization unit is limited. The saturation of olefins is an extremely exothermic reaction; and in general, the content is limited to that fraction of total temperature rise that will not impede or interfere with other exothermic reactions that are also concurrently occurring in the isomerization reactor, such as benzene saturation, isomerization, hydrocracking, and ring-opening.
For isomerization units with a drier on the fresh feed stream (Penex™ or Butamer™ process units), the olefins in the fresh feed streams can damage the molecular sieve in the feed driers. The molecular sieve will capture a limited amount of the olefins; and upon the higher temperature during sieve regeneration, the olefins will polymerize to form carbon on the molecular sieve. This will result in reduced molecular sieve life and require more frequent change-outs of the molecular sieve beds and more frequent regeneration as the bed ages.
The presence of olefins can also compete with other contaminants, such as sulfur and oxygenates, for the adsorption sites on the molecular sieve leading to potential breakthrough and contamination of the Penex™ or Butamer™ catalyst.
If olefins do enter the isomerization reactors, in the absence of other contaminants, the olefins should fully saturate to their respective saturated hydrocarbon counterpart.
ERIC STREIT (KBC Advanced Technologies, Inc.)
Olefin content in isomerization unit feed can be as high as 2 to 3% for C5/C6 units that process light reformate or raffinate streams. However, the typical C5/C6 feed olefin content is less than 0.5%. Olefin contents of C4-derived feeds are typically lower than 0.5%.
Olefins in the feed to isomerization units typically saturate to their corresponding paraffin. In doing so, they consume hydrogen and generate heat due to the exothermic reaction. The maximum temperature rise across the lead reactor can become limiting if the feed olefin content is too high. Increased temperatures can lead to excessive cracking. Olefins that do not saturate polymerize and form coke on the catalyst. The polymerization and increased cracking shorten the catalyst life. This is more evident in hydrogen once-through units where the amount of excess hydrogen is limited.
Coker and FCC naphthas are fewer desirable feedstocks unless the diolefins in these streams have been removed. The highly reactive diolefins polymerize more readily, leading to shorter catalyst life.
At normal operating temperatures, olefins have little impact on the molecular sieve driers. A small amount of olefins may absorb on the sieve; and when heated to regeneration, temperatures, these olefins polymerize. This polymerization reduces the effectiveness of the driers to remove moisture to protect the catalyst. Shorter drier life can be expected as a result. As with any stream containing olefins, if oxygen is present in the feed along with the olefins, gums can be formed that can contribute to fouling on any of the equipment upstream of the reactors. This is normally not an issue in isomerization units because the oxygen is a poison for halided isomerization unit catalyst; so usually, there is no oxygen in the feed.
This session will discuss the strange, unpredictable, unregulated security issues that sites must contend with and how partnerships can help create innovative solutions to these odd and unexpected wicked security problems!
MELDRUM (Phillips 66)
Let’s quickly review some concepts for good caustic scrubbing, caustic strength (not just pH, but also the weight percent sodium hydroxide), contacting of the treat stream with the caustic using pre-mixing or submerged inlets, packing within the scrubber, and caustic circulation to keep the caustic uniform in concentration. Poor circulation can cause local pockets of neutralized materials and limit removal of the heat of reaction.

Carryover from the caustic can lead to fouling and salt deposits in the downstream areas and some caustic cracking, and in severe cases of high temperature downstream, some caustic corrosion. Beware of non-post-weld heat-treated equipment that can exceed temperature limits when steamed out. Mitigation of carryover includes minimizing of the vapor rate. It is not uncommon, as a unit increases capacity over time, that sufficient attention is not given to the additional rates occurring through the scrubber. So, review that system to make sure you have proper cross-section in the scrubber. Control foaming by keeping the caustic slightly warmer than the inlet stream to avoid hydrocarbon condensation that could contribute to the foaming. Ensure that there is a sufficient disengaging section at the top of the scrubber, maybe even considering the use of some coalescing material on the outlet of the scrubber. Finally, using a sand filter or water-washing downstream of the scrubber will help minimize caustic carryover. In the Answer Book, I have a few more details on specific applications pertaining to the reformer, the isomerization unit, and Merox™ treatment.

GROPP (GE Water & Process Technologies)
In addition to what Craig said, caustic towers are common in the ethylene industry and typically more problematic than those found in the refinery due to the reactive nature of the ethylene-formed polymers and foulants. GE has significant experience in helping to prevent foaming, fouling, and corrosion in those units. While we do not have a lot of experience with caustic scrubbers specifically associated with isomerization units, the Best Practices we have learned certainly apply to all caustic scrubbers. Craig mentioned some of these including controlling the vapor velocity to avoid liquid entrainment and foaming tendencies. Fouling in the tower can cause issues as well. Localized high velocities associated with fouling can cause foaming problems. Caustic management must be practiced religiously: strength, cleanliness, and recirculation rates. Corrosion can be an issue due to wet acid gas in various components of the system; so as Craig mentioned, corrosion needs to be controlled. Over the years, the application of defoamer additives, antifoulants, and caustic-resistant corrosion inhibitors have been helpful in mitigating some of these problems.
BULLEN (UOP LLC, A Honeywell Company)
UOP experience with the common cause of corrosion in caustic scrubbers in isomerization service is due to not changing out the caustic on time. Logistical issues, sampling liquids properly, issues with doing the analysis in the lab properly, and looking at free caustic versus percent spent all contribute to not changing the caustic in time. We also see corrosion in some of the units that have heaters for winter to maintain the caustic temperature warm. In those areas, sometimes the steam used is too hot or has some super-heat to it which causes corrosion due to overheating the caustic. Steam tracing can cause corrosion externally because it overheated the caustic at the tube wall.
Oversized gas inlet distributors tend to corrode due to back-filling with caustic that is trapped in the distributor pipe and then becomes highly acidic.
Some designs have a two-stage system where a water-scrubbing packed bed is arranged above the top of the caustic packing area to wash down any caustic entrainment. But generally, the idea of having downstream traps is a good for the off-gas system.
MUKESH PATEL (Reliance Industries Limited)
When you say ‘sand filter’ for this particular application, what is the design criteria that are followed?
MELDRUM (Phillips 66)
Sand filters typically are used in the Merox™ system but not as much in the isomerization scrubbing or the reforming system.
MUKESH PATEL (Reliance Industries Limited)
You said that sand filtering is considered for capturing caustic carryover. What are the design criteria that are considered?
MELDRUM (Phillips 66)
I do not have design basis available to me right now.
LUCAS BERGER (Design Criteria)
I can probably help you with that later. I do not have it off the top of my head, but I am sure I can find design basis for that kind of system.
ALLEN KAISER (Delek Refining, Ltd.)
The design basis would easily be inserted into the transcript that comes out next year.
BULLEN (UOP LLC, A Honeywell Company)
In my experience, the most important aspect of a sand filter is getting the right sand.
CRAIG MELDRUM (Phillips 66)
Caustic scrubbing is achieved with:
1. Proper caustic strength, not just pH but wt% NaOH (sodium hydroxide) greater than 2%,
2. Good contacting of the treat stream with the caustic (premixing, submersed inlet, packing and sprays), and
3. Good caustic circulation to keep the caustic uniform with no pockets of neutralized caustic and remove the heat of reaction.
Carryover of the caustic can lead to fouling from salt deposits, caustic cracking, and caustic corrosion in downstream equipment. Be aware of non-PWHT vessels exposed to caustic and later to heat from process conditions and/or steam out operation. General mitigation of carryover includes:
• Minimize the flux through the scrubber by verifying that the scrubber has sufficient cross-section area for the gas flow and asking if the unit capacity been expanded without expanding the scrubber.
• Minimize the scrubber level.
• Control foaming to ensure that the caustic is slightly warmer than the inlet stream to avoid hydrocarbon condensation and liquid buildup that can contribute to foaming.
• Use disengaging or coalescing material on the outlet of the scrubber.
• Use sand filtering and/or water-washing downstream of the scrubber. Some additional mitigation considerations for specific process units are:
1. SR Reformer Regeneration
• Minimize separator level.
• Control foaming (keep solids in control with caustic change-out).
2. CCR Reformer Vent Gas Scrubber and Isom Scrubbers
• Use and maintain a waterwash section.
• Control level with water addition to minimize caustic dehydration and salt precipitation as the dry off gas becomes saturated through the scrubber.
4. Merox™ Treatment • Use a water-wash sand filter.
RONALD GROPP (GE Water & Process Technologies)
Caustic towers are common in ethylene units and are typically more problematic than those in refineries due to the more active nature of the hydrocarbon to form polymers. GE has significant experience helping ethylene producers reduce foaming, fouling, and corrosion in these systems. While GE has limited experience treating caustic towers associated with isomerization units, we have learned the following general Best Practice rules in the petrochemical industry which should apply:
- Control vapor velocity to avoid liquid entrainment and foaming tendencies.
- Fouling in the tower can cause issues with scrubbing and lead to high local velocities that can increase foaming tendencies.
- Caustic management must be practiced (strength, hygiene, contaminants, etc.) to keep the system running well.
- Corrosion can be an issue due to wet acidic gas in various components of the process.
- Defoamer additives, antifoulants, and caustic resistant corrosion inhibitors, such as those offered by GE, can be applied and may help minimize foaming, fouling, and corrosion issues.