Welcome Remarks
Question 37: What is the panel's experience in operating chlorided isomerization units, which were not designed specifically for benzene saturation, in a benzene saturation mode? How are operational parameters adjusted for the different operation?
MUEHLBAUER (Valero Energy Corporation – Benicia Refinery)
This is a good question; because as we blend more ethanol into the gasoline pool, a lot of refineries become octane-long and RVP (Reid Vapor Pressure)-long as well. So, the incentives to isomerize are diminishing as you blend more ethanol. Within Valero, we looked at one of our isomerization units and did a project to convert it into a benzene saturation unit but without capital improvements. We accomplished this by operating a unit designed for isomerization in benzene saturation mode. The key component is the cooling of the first reactor effluent. The benzene concentrations are normally maximum 4% to 5% in the isomerization feed. For each percent of benzene in the feed, you get about a 20°F exotherm on the reactor. So, the more you can cool the first reactor effluent, the more favorable the iso-to-normal equilibrium will be in the second reactor.

In terms of ways to cool the effluent, increasing unit recycle is one method, assuming there are not any hydraulic or space velocity limitations. Essentially, the benzene in the reactor feed, compared to the unit feed, is much more dilute. The other method would be trying to improve cooling by installing an inner reactor quench. I have heard that some facilities will bypass some of the feed around the first reactor into the second reactor to get exotherm and perhaps a little more cooling that way.
DUBIN (Axens North America)
We have seen that the quantity of benzene in the isomerization feeds has gone up due to the current regulations on benzene in the gasoline pool. For those units that do not have a dedicated benzene saturation reactor, the impact is a little more significant. The rule of thumb that Joe mentioned, 20ºF exotherm for every vol% (volume percent) of benzene, is applicable.
When processing feeds with a benzene content higher than design, we recommend reducing the inlet temperature to your first isomerization reactor to lower than typical. The goal is to have the temperature at the outlet of what we term the benzene saturation zone (that is, the top layer of the first reactor where all of the benzene is hydrogenated) at the ideal isomerization reaction inlet temperature.
Those refiners who are targeting maximum octane improvement, as opposed to just benzene destruction, may still need a higher overall temperature through that first reactor as you are bringing in more naphthene rings that will have to be opened before you can turn them into isoparaffins.
PATRICK BULLEN (UOP, A Honeywell Company)
We have had some licensees who do not have octane as a priority in their Penex unit but who still need to remove the benzene. In several cases, they have cut out their chloride injection in the Penex unit. They still typically see around a five upgrade across the unit, even without the chloride injection. They are able to handle the loss in activity by raising reactor temperatures to around the 400°F range for peak temperature. The system seems to be fairly stable. Of course,
you get the normal deactivation from oxygenates in the feed, for example, that eventual kill the catalyst. But for that type of operation, it seems to work well for them.
Our customers have seen some sulfur upsets in this type of unit operation. Reinstating their chloride injection seems to help recover some of the activity and still allows them to continue getting that minor octane upgrade they wanted. I agree with the other comments that, generally speaking, using the start-up line from the bottom of the stabilizer as a dilution source certainly dilutes the benzene that is in the feed as long as the hydraulics of the unit will allow it. We have some customers who have put in the separate benzene saturation reactor in front to try to minimize the impact on the Penex unit itself as a capital option.
DUBIN (Axens North America)
Axens has a number of units operating in benzene saturation mode even if the original design did not call for the same level of benzene reduction. With increasing benzene content in the isomerization feed, an increasing percentage of the isomerization catalyst in the first reactor is now utilized for benzene saturation. The refiner should lower the inlet temperature to the first reactor so that the temperature at the outlet of the ‘benzene saturation zone’ in the first reactor is
in line with the desired temperature. An increased overall temperature profile in the first isomerization reactor may still be required for ring opening of the additional naphthenes in the system brought about by the increased benzene content in the feed. The typical rule of thumb for liquid phase benzene saturation is a 20°F exotherm for every 1 vol% of benzene saturated.
AREIN LAZONDER (Albemarle Corporation)
Benzene saturation is possible in an isomerization unit loaded with platinum promoted chlorinated alumina catalyst. The bi-functional catalyst only needs the platinum function to saturate benzene. The acid sites on the catalyst have no additional function for the hydrogenation of benzene.
Operating in a benzene only saturation mode with no additional isomerization of the paraffins can be done in one of two ways: (1) by the injection of a chloriding agent to conserve the acid sites to be able to isomerize the paraffins once the benzene saturation mode is over or (2) without the injection of a chloriding agent if the unit will continue operating in a hydrogenation mode. With the latter operation the acid sites will be lost in the course of time and the isomerization activity cannot be fully recovered. Chloride inhibits the hydrogenation activity of platinum partly. For optimum hydrogenation activity, ceasing chloride injection can be considered. In both cases the operating conditions are moderate, at a RIT (reactor inlet temperature) = 120°C/250°F benzene is already hydrogenated. An excess of hydrogen is mandatory to prevent the catalyst from coking. At the reactor outlet the H2/HC (hydrocarbon)ratio should still be 0.1 molar ratio. Benzene saturation is exothermic so, if large amounts of benzene are processed, the heat release can be significant. To prevent temperature excursions, it is recommended to keep the maximum benzene concentration to be processed at less than 7 wt%.
Question 38: Does the panel have experience with mercury contamination of feedstocks to gasoline processing units? What are the typical crude sources that can potentially contain mercury? What units are likely to be most impacted, and how?
DUBIN (Axens North America)
Mercury is an ever-increasing problem that we see. It is coming in higher quantities and in more various crudes. Crudes from the East Asia region have historically been known to contain quite a bit of mercury. We have experience with a Thai crude that contained 3,000 [weight parts per billion (wppb) of mercury. However, a neighboring country has crude with only 10 wppb of mercury. The crudes of the Americas and Venezuela have been seen with mercury contents of up to 4,000 wppb. This variation tells us that the mercury impact on your refinery is going to be very site-specific and will depend on your particular blend and the source of your crude. At the same time, gas condensate streams, which are becoming a larger picture of the refinery feed, have been sampled with up to 2,000 wppb of mercury.
Where does the mercury go in the refinery? It can be found everywhere downstream of crude fractionation. Predominantly, it concentrates in the straight-run naphtha portion of the refinery; however, the most volatile species end up in the LPG (liquefied petroleum gas) and light ends. This leads to contamination in the light ends units, such as steam crackers, gas plants, and gasoline processes. For the gasoline processes, the major units in question are reforming,
isomerization, etherification, and hydrogenation.
The impacts from mercury contamination could be consolidated into three main groups: process impacts, finish stock impacts, and effluent impacts. Process-wise, we have seen aluminum heat exchangers in LPG plants that have been severely corroded due to mercury. For
the gasoline units, poisoning of catalyst is the major concern. Anywhere from 10 wppb to 20 wppb of mercury has been known to kill various precious metal catalysts. Therefore, it may not take much mercury in your crude to bring down a unit and short cycle it.
Finished products: If any mercury makes it into your finished products, say, a feedstock for a downstream user that is using your product as a steam cracker feed, then contamination by mercury may devalue that feedstock or make it unsellable, based on that user’s requirements, thereby creating a massive financial impact.
On the effluents, emission of mercury to the air or to the ground through purges is a concern, as is refinery personnel. Maintenance on any equipment that handles mercury then becomes an issue during turnarounds.
It must be kept in mind that hydrotreating mercury-containing feedstocks do not remove mercury; it only changes the mercury from its organic state to its elemental state. You will still need a guard bed downstream of the hydrotreater to remove the mercury and protect your downstream processes. Axens can provide sampling systems to determine exactly what species of mercury you have present. And through our RAM II process, we can design a guard bed
system to remove mercury down to the detectable limit of 1 wppb.
KOONTZ (HollyFrontier)
I do not have too much to add. At HollyFrontier’s refineries, primarily due to where we get our crude – which is from Canada and the Mid-Continent, we have not experienced mercury problems in any of our catalyst waste. We have not noticed any problems that we would attributeto mercury.
DAVID SMITH (UOP, A Honeywell Company)
I agree with all of Geoff’s comments, which were quite thorough. I have found that it is quite common for refineries in Southeast Asia to have mercury contamination in many of their hydrocarbon streams. Where they typically remove the elemental mercury is out of the naphtha stream downstream of the hydrotreater, as Geoff indicated. That appears to be the optimum location and the easiest way to remove the mercury with a non-regenerative mercury adsorbent, such as GB-346S.
PATRICK BULLEN (UOP, A Honeywell Company)
In units that do not have the guard bed technology, we have noticed that the typical stripper receiver and stabilizer receiver in the isomerization unit are places where mercury will accumulate. It becomes HSE issues when you do inspections at turnarounds. There is a little pool of mercury in the receiver, and there are typically no boots on the stabilizer receivers in the isomerization units to drain out the mercury; so that makes it more difficult, from an HS&E
perspective.
DUBIN (Axens North America)
Axens has significant experience in assisting refiners who have found mercury in their feedstocks. The quantity of mercury found in crudes can quite significantly depending on the region, and even the source of crude within a region with levels from as little as 1 wppb up to4,000 wppb of mercury being observed. Crudes from the East Asia region have been known to contain mercury. Axens has experience with Thai crude with 3,000 wppb of mercury. Yet,
crudes from nearby countries may have as little as 10 wppb of mercury. Closer to the U.S., Venezuelan crudes have been observed with up to 4,000 wppb of mercury. But mercury contamination is not limited to just crudes. Gas condensate streams with levels of mercury in the 2,000 wppb range have also been found.
A wide range of mercury species can be found downstream of the crude units, but significant amounts mercury is often found in the straight-run gasoline portion of the crude. At the same time, the most volatile mercury species are usually found in the light ends/LPG portion of the crude. This leads to mercury contamination issues predominately in light ends/LPG units (gas plants, steam crackers, etc.) and gasoline processing sections (isomerization, reforming, and etherification). The impacts on these units can be essentially grouped into three main categories, process impacts, effluent impacts, and finished product impacts.
Process impact wise, catalyst poisoning is the most common. Impurities in the wppb level have been known to cause cycle life degradation in gasoline hydrogenation units. But catalyst poisoning is not the only concern with mercury contamination. Severe corrosion of aluminum plate heat exchangers, in both LNG (liquefied natural gas) facilities and ethylene plants, has been observed and traced back to mercury.
The effluent impacts are harder to both identify and quantify, but they are still quite serious. The potential exists for the emission of mercury through effluent streams to both the air and to the ground. Protecting refinery employees from mercury contamination is also a key consideration. During their daily rounds, refinery personnel can be exposed to mercury in both normal operation and during maintenance of any equipment that handles mercury-contaminated
feedstocks.
The other major concern with mercury contamination is in the pollution of finished materials. Should mercury find its way into a finished stock, if that finished stock has to be sold as a contaminated one, the value of the material could drop drastically. And depending on the service for which the finished stock is being sold, it may not take much mercury at all to be deemed contaminated.
KOONTZ (HollyFrontier Corporation)
HollyFrontier processes crude primarily from Texas, Canada, and the Mid-Continent. HF also processes some natural gasoline from the Mid-Continent. I was unable to find any data regarding mercury in the crudes or natural gasoline that we process. We routinely do metals analyses on catalyst and other process samples, but mercury is not in the normal list, and we do not ask for a special analysis looking for it. We also send catalyst and other wastes out for metal
reclamation and/or disposal and we have never experienced environmental concerns due to mercury contamination.
ILYA ARANOVICH (GTC Technology)
Mercury is a poison for all of the isomerization catalysts as it has properties to be able to replace the metals that form the framework of the catalyst and also substitute the protons in the acid sites. Both of these phenomena lead to a decrease in the amount of active catalytic sites (acid and metal) and lead to faster catalyst deactivation. For the SI-2TM catalyst the recommended level of mercury is 20 wppb for normal operation to meet the guaranteed values on performance. A light naphtha pre-hydrotreatment block significantly decreases the amount of metal impurities that hit the downstream isomerization reactor. We have not seen problems in any of our 10 operating units with maintaining this level of mercury with a pre-HDT (pre-hydrotreatment) block. We highly recommend using this approach and properly preparing the isomerization feed.
ANDREA FOSTER and JAMIE CHISAMORE (Johnson Matthey Catalysts)
Mercury is found in an ever-increasing number of hydrocarbons worldwide. The level of mercury can vary significantly depending on location. Data published by the Environmental Protection Agency (EPA) show reported levels of mercury in various crudes. Some of these are listed in Table 1. Published levels of mercury found in refinery product streams are shown in Table 2.
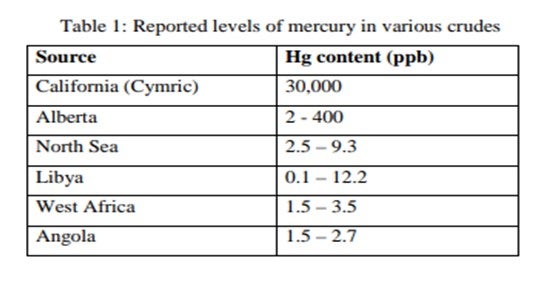
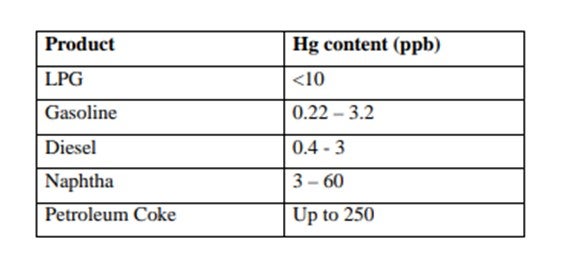
Although the levels of mercury recorded are low, the tonnages of liquid hydrocarbons handled are enormous, so downstream processing equipment is exposed to a substantial amount of mercury. The main worries are corrosion, poisoning of catalysts and health and safety. These can involve serious financial losses.
Precious metal catalysts such as platinum and palladium are particularly prone to poisoning by mercury. They react to form a 1:1 amalgam, i.e., platinum-mercury (PtHg) or palladium-mercury (PdHg) that is stable at low temperatures. With low metal loadings very low levels of mercury can have a significant impact on performance. The mercury can be driven off by heating above 300°F to 400°F, but this accelerates sintering and hence loss of active surface
area.
Johnson Matthey has many Refinery and Petrochemical customers that have installed mercury guard beds upstream of units using precious metal catalysts. The use of PURASPECJMTM* fixed-bed mercury removal absorbents has resulted in significant increases in catalyst life and also provides environmental protection, removing mercury from the lighter product streams.
*PURASPEC is a trademark of the Johnson Matthey Group of Companies.
AREIN LAZONDER – Albemarle Corporation
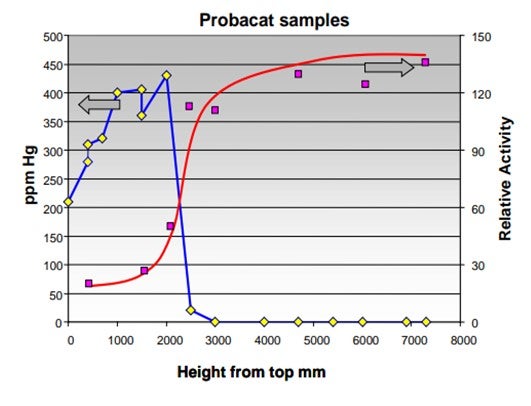
Mercury is a very effective poison for platinum promoted, chlorinated alumina catalyst. As can be seen in the graph below, the isomerization activity decreases very fast with mercury being deposited on the catalyst.
Question 39: What options are available for managing blend stream Reid Vapor Pressure (RVP) and pentane content during the summer blend season? Discuss operational changes in existing equipment, simple modifications, and large capital projects.
STEVES (Norton Engineering Consultants, Inc.)
Each refinery configuration will obviously be very different, so there is never going to be a one-size-fits-all solution for managing gasoline component RVP or dealing with pentane streams. However, there are certainly many common elements that can be explored. First of all, it is important to understand the light-end C4s and C5s present in each of the gasoline components in your refinery. Next, try adjusting fractionation to reduce the C4 content of each gasoline component stream. Fully debutanizing these streams will make it much easier to optimize blending to ensure that the finished product will meet the required vapor pressure specifications.
Refiners should also consider whether all butane streams that can be refined to produce gasoline are being effectively handled. For instance, are all of your butylene molecules being alkylated to produce gasoline, or are some of those butylene molecules ending up in your gasoline pool and increasing the RVP? In addition to making sure that all available butylene streams are being refined through alkylation or polymerization, for instance, a refiner may also consider alkylating some of the amylenes that exist in the gasoline pool.

After evaluating each of the operational changes possible to reduce the gasoline stream RVP, it may become apparent that capital investment is required. If a particular gas stream cannot be fully debutanized, the refiner should consider re-traying that tower to improve its performance for removing all C4s. Similarly, if extra capacity is not available in the alkylation unit to alkylate all of the butylenes, or if amylene alkylation is desired, then expansion of the alkylation unit may need to be explored; or you may consider looking at other processing options for the propylene to make room for those streams in the alkylation unit.

In addition to segregating and separating the butane streams from each gasoline component, it can also help to segregate the pentanes from the butanes and put those pentanes in dedicated storage. Segregation of the pentanes from the butanes will allow high quality butanes to be exported for storage or sold as feedstocks to other refiners and the segregated pentane to be blended back into the gasoline pool. A mixed butane and pentane stream is going to be much harder to get rid of.
Finally, construction of a new unit can also help reduce the quantity of butanes that have to either be exported or blended into the gasoline pool. It will obviously cost a lot of money. Construction of a butane isomerization unit, for instance, will reduce the quantity of butane that needs to be exported or blended and create a nice isobutane stream which can be used to reduce or eliminate isobutane imports needed for an alkylation unit.

Question 40: What process options are available for U.S. refiners to economically make 10 ppm sulfur gasoline? Discuss impacts to other product qualities (such as octane and vapor pressure) and how these secondary effects impact the gasoline pool.
DUBIN (Axens North America)
There are several routes to Tier 3 gasoline: post-treating, pre-treating, or a combination of the two. For Tier 2, we saw that about 80% of refineries invested in post-treaters. At the time, octane barrels were valuable, gasoline was in demand, and the capital cost was justified for installing a post-treater. As we move to Tier 3, we see a more stagnant gasoline market. Ethanol, as well as increased jet kerosene production, is limiting the amount of FCC naphtha available. There is not a strong demand for gasoline, and it does not appear to be changing.
For Tier 3, we will likely need to meet 10 wppm sulfur in the gasoline pool. This may entail 10 wppm to 20 or 25 wppm sulfur out of the FCC naphtha to meet the pool specs. Co-processing of orphaned gasoline streams will also be critical, like straight-run, unprocessed light cat (catalytic) naphtha, and any other raffinates. Those orphaned streams will need to be treated to help meet the 10 wppm in the pool.
Some of the Tier 2 units do have additional capacity available. Due to undercutting the FCC naphtha, there are additional barrels available to co-process in those units; however, not every unit is in that position. What may be required for these Tier 2 units is to increase the severity to get to Tier 3. This severity increase may be accomplished through elevated reactor temperatures or the addition of catalyst volume. Other revamp options to overcome hydraulic bottlenecks or product octane limitations include:
• adding a second HDS (hydrodesulfurization) reactor,
• choosing a more selective catalyst,
• adding a splitter to fractionate the light cat naphtha from the heavy cat naphtha if you are processing full-range naphtha,
• polishing catalyst to help control recombinant mercaptans, or
• adding of a complete second stage to your existing post-treater.
For those refiners who did not invest in a post-treater, there are fewer knobs to turn. Getting a cat feed unit to operate for a four- or five-year cycle is unlikely. Deeper desulfurization out of the cat feed hydrotreater may be a challenge. It could be entirely possible that the existing pressure level of the cat feed hydrotreater does not allow for the significantly deeper desulfurization needed to meet a Tier 3 direct gasoline off of the FCC main fractionator.
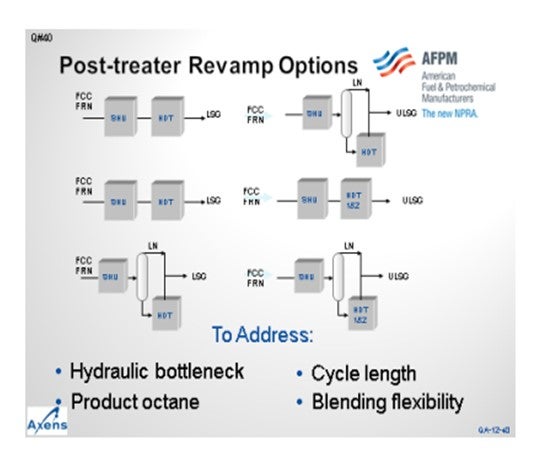
There are also some revamp ideas for cat feed hydrotreater units. A conversion of the cat feed to a mild hydrocracker has the benefit of increasing distillate production while meeting direct Tier 3 gasoline. Axens offers a permutable guard bed reactor system called PRS, which is a lead-lag guard bed for the cat feed hydrotreater. The PRS system allows for a continuous operation of the cat feed hydrotreater to help match your FCC cycle. The PRS system is an
interesting option for a capital-intensive unit like a cat feed hydrotreater.
However, there are still concerns for producing Tier 3 gasoline directly from the FCC main fractionator. Without a post-treater, you do not have any final control over the product sulfur meaning that any upsets in either the cat feed hydrotreater or the FCC main fractionator can have an impact on whether you can meet your gasoline pool sulfur requirement. For the cat feed hydrotreater, of concern would be the desulfurization level. On the FCC main fractionator, you need to maintain distillation control on the tail end of the heavy cat naphtha to ensure that you are pulling more sulfur into the naphtha tail during a main fractionator upset. Having a post-treater allows you to play catch-up with sulfur if there is an upset, as well as to operate with your cat feed hydrotreater down.
On the overall pool octane, we have seen that many refiners are octane-long right now due to ethanol. Reformers are operating at lower severity than they have historically operated, so there appears to be some room for octane loss in FCC gasoline desulfurization while still meeting the overall pool requirements.

PIZZINI (Phillips 66)
Phillips 66 has experience with almost all of the major sulfur removal technologies that can be used to meet the 30-ppm spec, including conventional hydrotreatings, scan finding, S-Sorb, ISAL, catalytic distillation, Prime-G+, and caustic treating. Now the government wants us to ratchet that down to 10 ppm. So, for 10 ppm, we will have to treat additional streams. The slide lists several: butanes, pentanes, light cat naphtha, and light straight-run gasoline. So, our strategy, at least right now, is to look at treating those streams I mentioned using regenerative caustic treating and also revamping some existing hydrotreaters or conventional hydrotreaters to the newer technology.

This example might be unique to this one location, but we do have a benzene extraction unit that removes the benzene from light reformate. That extraction is done with sulfolane, a solvent which contains some sulfur. There is a certain amount of solvent lost with the raffinate from that process, averaging about 10 ppm to 15 ppm. So, with a 10 ppm gasoline target, even that stream needs to be addressed. Right now, our plan is to put in a better solvent waterwash column. We currently have that in our scope for the Tier 3 project.

DUBIN (Axens North America)
There are several paths to meeting 10 wppm (Tier 3) ultra-low sulfur gasoline. Depending on site specifics, FCC pre-treatment, FCC gasoline post-treatment, or a combination of the two options can help refiners meet the Tier 3 regulations.
In the early to mid-2000s, approximately 80% of U.S. refiners addressed the Tier 2 regulations through the installation of FCC gasoline post-treatment. At the time of Tier 2, gasoline demand was strong, and the retention of octane barrels was key, which justified the cost of installing a new unit. Ahead of Tier 3, we see that the overall gasoline market is stagnant and octane barrels are not as valuable due to a number of reasons: ethanol, increased jet/kerosene production, etc.
For Tier 3, refiners will likely need to meet 10 wppm to 30 wppm in the FCC gasoline to meet the overall blending requirements. However, the processing of just the FCC gasoline streams may not be enough. Processing of orphaned gasoline streams will also be critical to meeting the Tier 3 pool levels. Those who invested for Tier 2 may have additional capacity in their existing units to co-process orphaned streams as many refiners are undercutting the FCC gasoline to maximize distillate production. Due to available capacity, as well as other factors, these Tier 2 units can likely meet Tier 3 through operating severity increases. Other units will need to be revamped. The primary drivers for the revamp of the post-treater all aim at addressing cycle length, hydraulic bottlenecks, product octane, hydrogen consumption, and increased blending flexibility. The main options for post-treater revamp are to add HDS catalyst volume, use the most selective catalysts, add a splitter, add polishing catalyst volume, or add an independent second HDS stage.
For those refiners who handled the Tier 2 requirements through adjustments to their FCC pre-treatment, they have fewer knobs to turn for Tier 3. Revamping the FCC pre-treater to desulfurize deeper can be difficult as a four- to five-year cycle life at a high desulfurization level is challenging. The existing CFHT pressure level also may not be compatible with desulfurizing the FCC feed to a level that will allow for direct Tier 3 gasoline. One option for those units with
a CFHT is to revamp the existing CFHT unit into a mild hydrocracker. This revamp scenario offers improved middle distillate production while helping to meet the Tier 3 ULSG (ultra-low sulfur gasoline) requirements. While this revamp scenario has a number of improvements that look attractive, a key concern when making ULSG direct from the FCC is that there is little margin for error on unit operation and FCC fractionation. This is due the lack of post-treatment to stabilize the sulfur levels before the gasoline material goes to the pool. The addition of an FCC gasoline post-treater allows for catch up on sulfur levels in the gasoline should the pre-treater suffer an upset. As well, the use of an FCC gasoline post-treater offers the ability to bring down the CFHT and replace the pre-treater catalyst while still maintaining gasoline production.
As to overall octane for the pool, further treatment of the FCC gasoline should not impact the pool significantly. While deeper desulfurization will result in lower FCC gasoline octane, that is certain, catalytic reformers are generally operating at very low severity and there is often room to increase octane barrels from the reformer.

