Question 34: Hydroprocessing reactor pressure drop can increase due to feed particulates, corrosion by-products and polymerization reactions. How can bed design and loading method be optimized to avoid pressure drop limiting the cycle length or throughput?
McARTHUR (Phillips 66)
There are a lot of approaches to helping out with pressure drop problems in a reactor, and I will go through them. We use all of these at Phillips 66. I will start at the top. There are particulate catching trays. These are relatively new. We have had limited use with these, although we think they have been fairly successful. We are reviewing potential applications at some of our other units that have some historical issues.
Many of our units have had, and some still do, trash baskets or scale-catching baskets. But in general, we are removing them because they are difficult to load around. Removing them tends to add a lot of time at turnarounds, and they can actually create some maldistribution issues at the top of the bed. That is not always the case. There are some cases where we choose to keep them in the reactor. It is a site-by-site decision.
We think the primary workhorse is with the graded bed at the top of the reactor for two purposes. One is for particulate removal. There are many different kinds of particulates. They can come in with the feed and have several layers of size change. Shape change will spread this catch of particulates out across a large volume of material with a lot of void space and leave a lot of room for the oil to keep going through for longer and longer periods through the cycle.
Likewise, these graded beds can have some amounts of increasing activity built into them. We start with very low activity material to initiate some of those reactions, which helps avoid polymerizing compounds onto the main catalyst bed. The primary reaction here is saturation of the diolefin bonds that would otherwise, when hitting the primary high activity catalyst, polymerize right out. Also, if these polymers are going to foul out, I would rather they foul out in the high void space up in the graded bed rather than in the main bed.
Moving down to the loading method for the main catalyst bed, if there are typically DP (differential pressure) limitations associated with the main bed, then decreasing amounts of dense loading or eliminating dense loading can leave a little room for improvement in pressure drop across the reactor. This generally is not too significant, but it can help. And then actually, the choice of catalyst pellet size can be a factor as well. Smaller catalyst pellets tend to have a little higher pressure drop, so choosing a little larger pellet can have a small impact on DP across the main catalyst bed.

The diagram is what I have walked through. The main takeaway we would like to tell our engineers is that it is very site-specific; and really, the best way to learn the location of your issue is to do sampling at the end of the run, not just dump the catalyst and plan to load it like you loaded it last time because you got a “pretty good” run last time. It is really through sampling that you learn where the problem is in the reactor, what you are catching, and the cause of the problem. And then, you can optimize the bed for future runs after that.

WRIGHT (Hunt Refining Company)
One method we have used to avoid pressure drop is, as Scott mentioned, balancing the amount of filtration material with the demet or graded bed. For example, our diesel hydrotreater was running into a particulate problem that resulted in high ∆P (delta P, pressure differential) on R1 (Reactor 1). So we stole some active catalyst volume and replaced it with filtration material to help us get the second half of the run finished. For our hydrocracker, we had the unusual experience of buying larger than required reactors relative to the design feed. [Laughter] We are on Cycle 2 now. In Cycle 1, we did not use up all of the space. We had some adventures in our feed resulting in some ∆Ps on R1, Bed 1. So during the skim, we filled up the volume that was there with the filtration material, again, to help us make the second half of the cycle. And as Scott mentioned, you can use the activity grading to help you balance pressure drop with activity, as well as employing dense versus sock loading at the appropriate times.

TEMME (Albemarle Corporation)
I agree with what Scott and Samuel just went over. Definitely, it is good to know the past to address the present. If you have something occur on the current cycle, it is hard to address quickly; but hopefully, you have had some past history to give you potential ideas for a path forward. You definitely want to make sure that a comprehensive size- and activity-grading catalyst loading is employed, looking particularly to see whether or not it is a particulates problem, a polymerization problem, or maybe a combination of the two and also whether or not that occurs at the interface of any one particular layer.
You have to check to see if dense loading is feasible. If not, do the unit hydraulics cause pressure drop limitations? In that case, sock loading has to be used. Yes, sock loading does give a lower initial reactor DP, but it can lead to less optimal flow distribution and potentially preferential coking, which then can lead to increased reactor DP if the sock loading is not done well. Another consideration is whether a larger sized catalyst may yield a lower reactor DP, but you must be certain that the loss of catalyst activity can be tolerated by the unit activity demands.

Really, you have to take a look at – I think in terms of a holistic way – the big picture and whether the upstream impacts are significant contributors. What is going on with the feed management? Is cracked feed storage problematic, in terms of nitrogen blanketing? Purchased feeds: Do they seem like a bad actor? Can the cracked feeds be hot-linked to run directly to the unit instead of going out to the storage? Are the purchased feeds properly blended and managed where you will not have purchased feed volume swings from high to low and back to high again, which can cause some issues, in terms of hydrogen consumption variability and potentially DP type of problems?
Upstream, especially at the crude unit: Are there good monitoring and inspection programs? Is there a comprehensive corrosion coupon installation in the crude unit that is monitored regularly? Have feed tanks been running for 10 years? Maybe they have not been inspected or cleaned, and you just have a high level of particulates, iron sulfide, or other solids buildup in the tank that is now working its way over into the unit. Now a lot of people have feed filters, but are they being worked on and maintained properly? And then there is their assessment of the captured materials. Have a particle-size distribution assessment been done to help give you an idea of how you might put your grading system together? So, we feel that all of this can be very helpful in avoiding pressure drop problems.

JAMES (TIM) CAMPBELL (Eurecat U.S. Incorporated)
I want to add a comment for those folks who use regenerated catalysts; because many times, there is a concern about pressure drop with regenerated. At Eurecat, we address that. We can test for relative pressure drop of the regenerated catalyst versus its fresh version and give you a factor that you could use in your evaluation.
SHRIKANT MADHAV VAIDYA (Indian Oil Corporation Limited)
I have one observation. The minute we have a recycle gas flow failure, after which the plant gets restarted, we have a step change in the R1, Bed 1∆P. So, could there be any reason for that?
McARTHUR (Phillips 66)
I guess there could be several reasons. If it typically trips your heater when you lose your compressor, the rapid cooling of the tube will sometimes cause coke material to slough off the tubes. And then when you start back up, you will carry that back onto the reactor bed. We have had that problem a few times. Also, any significant period of time where the oil on catalyst is a hot temperature with no hydrogen there, you are going to have some coking issues and fouling with that, too.
WRIGHT (Hunt Refining Company)
We were having recycle gas compressor issues in Cycle 1, so we were getting these nuisance trips. We saw that the fouling material in the CFEs (combined feed exchangers) would then flow to R1, Bed 1. And later on in the cycle, we were having charge heater limitation; so we intentionally pulled out the feed in order to slough the foul end off of the CFEs and onto the filtration material of R1, Bed 1. So we serendipitously observed that phenomenon and used it to our benefit later on.
PANKAJ DESAI [Shell Global Solutions (US) Inc.]
I agree with what the panel has said that having a well-designed graded bed is necessary to mitigate pressure drop issues. However in spite of that, upsets can occur. If you need extra insurance or protection against pressure drop, we offer a couple of devices in our reactor internals portfolio. If it is simply a case of inorganic iron or scale deposits, then we offer a simple scale-catching tray that helps to minimize pressure drop. For more complex feedstock issues with gum precursors or treating cracked feedstocks, we offer a more sophisticated device called a filter tray that helps to minimize pressure drop. Both of these devices sit in the dome of the reactor and do not take away any active space in the reactor from loading active catalyst.
JEFF JOHNS (Chevron USA, Inc.)
We have already invited you to come to the Principles & Practices session tomorrow morning. We have an hour set aside to continue the discussion. I appreciate the good responses from the panel. We hope to continue with more discussion and less advertisements.
SERGIO ROBLEDO (Haldor Topsoe, Inc.)
A properly designed catalyst system can go a long way in mitigating pressure drop issues. A key factor for designing a catalyst load is a properly designed graded bed. A fixed bed catalytic reactor can be graded in a number of ways, depending on the nature of the feedstock. We must differentiate between contaminants entrained in the feed as opposed to compounds, such as diolefins or metals-containing molecules in the feed that react upon entering the reactor environment and have the tendency to produce byproducts [polymer, Fe (iron), Ni (nickel), and V (vanadium) deposits]. In extreme cases, polymerization can manifest itself as a very hard crust which, in some cases, has to be jack-hammered out of the unit. Such agglomeration causes bypassing of catalyst and maldistribution in the catalyst bed.
In other cases, the material is very fine, in terms of particle size (5 to 10 microns); and although not forming a crust, it will also deposit in a small area, resulting in a reduced void fraction which causes the pressure drop across the catalyst bed to increase exponentially. Our experience, since Haldor Topsoe, Inc. invented grading in 1979, shows that the critical point is reached when the available bed void is reduced to approximately 20 to 25%. We therefore prefer to use what we term “useful bed void”. This means the fraction of the reactor void which is available for deposition or storage of contaminants before the critical limit where the pressure drop increases. Please refer to the following table:
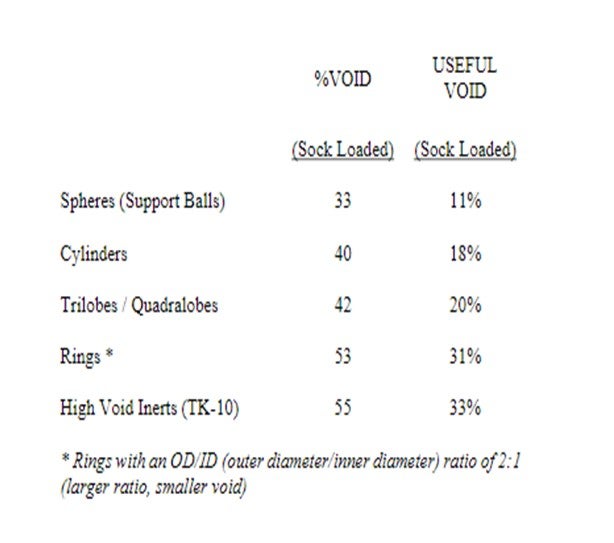
Inorganic material such as iron or polymers can quickly fill up the spaces between particles of a main bed catalyst effectively cementing them together, causing maldistribution and eventually crust formation with a resulting increase in pressure drop. The properly designed graded bed system can extend the cycle length of a main bed before pressure drop becomes an issue.
Fouling of a catalyst bed can be caused by a number of different factors or sequence of events in the refinery; however, in most cases, one of the most commonly encountered contaminants is iron. Organic iron can be present in refinery feedstocks; but more often than not, the iron originates from corrosion within the refinery itself. This is especially true for refineries using naphthenic-type crude. Processing these high TAN crude types accelerates corrosion in plant piping, tankage, etc., due to the presence of naphthenic acids. This iron can have a wide range of particle size distribution ranging from submicron up to large flakes.
When grading for iron, the graded bed system must be able to handle the iron in all its sizes Haldor Topsoe, Inc.’s shape-optimized TK-26 TopTrap™ is the successor of our very successful TK-25 TopTrap™. The optimized shape of TK-26 TopTrap™ provides an improvement in pickup capacity of 21%. TK-26 TopTrap™ is designed to load with an interstitial void of 61% and an additional 25% void in the pore system, for a total void volume greater than 85%. This gives TK-26 TopTrap™ the ability to trap not only larger quantities of deposits but also the ability to trap the smaller sized particulates. Larger-sized material deposits in the interstices between the individual particles. Fines or smaller-sized materials enter the TK-26 TopTrap™ pore system and are trapped within the structure of the particle itself. Not all grading is created equal, and it is not just a question of creating a high interstitial void fraction. Porosity and surface area are also important functionalities for graded bed material designed to trap particulate matter. Please note that the material trapped in the pore system of TK-26 TopTrap™ will not add to the pressure drop. This is the reason why TK-26 TopTrap™ is more effective in mitigating pressure drop issues than other ceramic trap materials in the marketplace.

This specialty trap would be loaded right under the high-void, high-density topping layer, such as TK-10 or TK-15. TK-10 or TK-15 act as a hold-down, as well as storage space, for larger size inorganic material.
For units processing a high number of cracked stocks, a high quantity of olefins – which can cause large temperature rises in the reactor – need to be considered when designing a graded bed. In the presence of small amounts of oxygen, or at elevated temperatures above 450°F, these molecules will radically polymerize to form gum that can foul exchangers or reactors causing poor heat transfer, as well as high reactor pressure drop. If the feed blend contains coker naphtha, then we also need to consider the highly reactive diolefins compounds, which are severe coke precursors.
Cracked stocks should preferentially be sent directly from the upstream unit to the hydrotreater to prevent contamination with oxygen. Even straight-run stock that may be part of the feed component must be prevented from being contacted by oxygen by using nitrogen-blanketed storage tanks.
Even without oxygen ingress, the diolefins in the coker naphtha can polymerize at elevated temperatures. With high olefinic and/or cracked stocks, saturation will occur regardless of catalyst activity. However, an option does exist whereby the reaction front is spread out over a large volume, rather than at the top of the bed, or at the reactive interface between the inert topping and active catalyst.
This option is referred to as an “activity gradient”. We use a number of low- to medium-activity Raschig ring-shaped catalysts in 1/8” and 3/16” sizes on top of the main bed catalyst. Furthermore, high void inerts and rings have the advantage of improving the radial distribution and are also recommended to enhance distribution.
Haldor Topsoe, Inc. produces our 1/8” ring catalysts with a very large ID of the hole to allow for maximum feed contaminant capacity without sacrificing the crush strength. The ratio of the OD/ID for our 1/8” rings is 2:1. Furthermore, we manufacture all of our grading material to strict ISO (International Organization for Standardization) specifications from fresh raw material and not from scrap catalyst or regenerated catalyst, which may contain impurities from its prior use.

The particle size will definitely impact the SOR (start-of-run) pressure drop of the catalyst bed because each particle has an inherent interstitial void. For comparison, a 1/16” TL (trilobe) will have, on average, about an 80% lower SOR pressure drop than a 1/20” TL. This will have minimal to no impact on the SOR activity of the load but will allow for a larger window between SOR and EOR (end-of-run) pressure drop. This strategy can be further refined in reactors with multiple beds where a larger sized catalyst can be installed in the top bed where pressure drop is a concern, and a smaller sized (i.e., 1/20”) catalyst installed in the lower beds where pressure drop is less of an issue.
The same strategy is also true for loading method. On average, the SOR pressure drop of a dense loaded bed will be twice that of the same bed sock loaded. Therefore, sock loading can be considered when pressure drop is severely limiting cycle length and not activity. However, sock loading will mean that the activity will be about 15% lower than for a dense loaded bed. Again, the same method of titration can be employed in sock versus dense loaded portions of the catalyst bed. In multiple bed reactors, it is sometimes advisable to sock load the top bed and continue dense loading the lower beds. This will minimize the impact on the overall activity of the system but will improve the window for pressure drop build on the top bed where pressure drop is typically an issue. One point to keep in mind is that radial flow profiles in a bed may be impacted when changing to sock loading which could limit the cycle length due to peak operating temperature.
In units with severe pressure drop issues and where activity is not an issue, both a size change and loading method may be a viable solution. These two approaches – size and/or loading method – can also be employed in units with pressure drop issues in lower beds.
Scale Catchers
In addition to an effective graded-bed system, Haldor Topsoe, Inc. also recommends the use of our patented scale-catching trays, which are placed in the top of the reactor. The tray is installed above the tangent line to avoid reducing the catalyst volume. Haldor Topsoe, Inc. has designed scale-catching trays for both mixed-phase and 100% vapor-phase operation.
Clients have seen as much as a doubling of the cycle length after installing our scale catching trays, and we often find two to three inches of material deposited on the tray. This material would have fouled the catalyst bed without the presence of the scale catcher tray. This gas oil hydrotreating unit increased the cycle by 100% after installation of a Topsoe scale catcher.
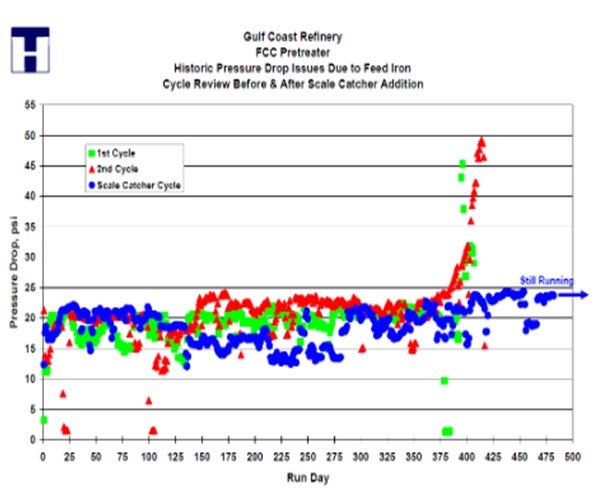
CHRIS STEVES (Norton Engineering)
All catalyst vendors provide hydroprocessing reactor grading design that can help to manage pressure drop during the catalyst cycle in order to keep reactor throughput as desired. It is important to understand the historical sources of pressure drop in the unit, though, so that the proper grading system can be chosen: a system that is designed to handle particulate contaminants may not work as well in a situation where the pressure drop is due to polymerization reactions. Samples from the top of the bed after every run should be used to confirm sources of pressure drop and then taken into account when designing the grading system.
DORIAN RAUSCHNING (Criterion Catalysts & Technologies)
Catalyst bed pressure drop growth mitigation can be achieved by integrating the results of empirical bed unloading observations, spent catalyst fines analysis, and subsequent design of a deep bed filter. While a multi-cycle iterative approach using trial and error can often result in the final development of a deep bed filter system, a more rapid resolution can be obtained by taking the time to gradually remove (vacuum) the top bed catalyst, visually observe the location and quantity of dust/fines/crust and collect intermittent spent catalyst/crust/dust/fines samples for chemical and particle size distribution analysis.
Based on the combined observations and analytical results, a deep-bed filter can be developed which utilizes void fraction grading of specialized grading materials and catalysts that facilitate selective trapping of different sized feed particulates, gums, and/or soluble metal naphthenates. A large diameter and intersticial void catalyst layer is first used to remove large-sized feed particulates while simultaneously still providing sufficient void space to permit the medium/small-sized particulate to penetrate deeper into the reactor and not form a high-density crust. Subsequent ever-smaller diameter and intersticial void fraction grading or catalyst layers are then used to promote further selective feed particulate removal until the primary conversion catalyst is reached. These layers are all sock loaded. The net effect of such deep-bed filters is that the feed particulate is distributed over a greater depth of reactor volume and do not form immediate, high density, narrow layers that restrict feed or treat gas flows.
The deep bed filter should also provide an activity gradient to mitigate the formation of adhesive gums that can form from olefinic polymerization reactions. Feed unsaturates can react to combine longer chain polymers which can act as gums and form coke that bridges catalyst pellets. In addition, these gums interact with feed particulates causing rapid pressure drop growth. To mitigate these reactions, the deep bed filter should be graded for activity as well. The bed’s upper layers should have lower activity catalysts to slow down the rate of polymerization and promote hydrogenation. The deep bed filter’s subsequent catalyst layers’ activity begins to increase as the concentration of polymer-forming compounds decreases.
Criterion’s OptiTrap™ product line is designed to provide a wide range of top-bed grading catalyst products from which a deep-bed filter can be constructed. OptiTrap™ products offer selective particulate removal in combination with an activity gradient. In addition, Criterion has extensive troubleshooting experience in resolving reactor pressure drop growth issues in naphtha, distillate, FCC PT, and hydrocracking applications. Criterion also offers a full range of research and analytical support.
Question 41: Have the panel members considered 15% ethanol (E15) gasoline blending?
KOONTZ (HollyFrontier)
My first slide shows a little background. The EPA administers the Renewable Fuel Standard program that has volume requirements for renewable fuels. They established these volume requirements under the Energy Independence and Security Act of 2007. The EPA tracks compliance with the Renewable Identification Number (RIN) system, and they assigned a RIN to each gallon of renewable fuel.

HollyFrontier satisfies much of its requirement for conventional biofuel, which is essentially corn ethanol, by blending E10 gasoline at many of its terminals. Most of HollyFrontier’s gasoline is sold via pipeline to terminals owned by others; therefore, we are not able to supply our full mandated volume. HollyFrontier does purchase RINs from others. The decision to purchase ethanol to blend or the RINs is based on the economics of the cost of the RINs.

Ethanol blending for the refiner does have a significant impact on two critical gasoline properties: namely, octane and RVP. The hydrocarbon blend stock used for 90% of the E10product, which HollyFrontier calls sub-grade, has an octane rating of about 84. After blending with the 10% ethanol, the resulting octane is the regular 87. So being a refinery that adjusts total octane with its reformer severity, this allows us to run a lower severity, which is especially beneficial to those refineries with semi-regen reformers that operate at relatively high pressures and relatively low liquid volume product yield.

RVP is the other critical property affected by blending. When ethanol is blended with naphtha at a low concentration, the RVP of the gasoline is increased. Pure ethanol does have a low RVP; but when it is blended with hydrocarbon, it behaves more like a light hydrocarbon and actually raises the RVP. For example, with E10 for naphtha having an RVP of 9, the resultantE10 product has an RVP of about 10. So, to encourage ethanol blending, in 1990, the U.S. Congress passed a waiver known as the “One-Pound Waiver” which allows E10 gasoline to be sold at one psi (pound per square inch) higher than that normally required.
For the refinery, E15 would allow lower octane severity reformer operation, which would be beneficial. However, the EPA regulation implementing the “One-Pound Waiver” specifically references gasoline containing between 9% and 10% ethanol. The EPA has refused to extend this One Pounder Waiver to E15. Therefore, marketing E15 requires a sub-grade blendstock that has an RVP approximately 1 psi lower than normal gasoline sub-grade blendstock used for E10.

In addition, since January 2011, E15 has been permitted for use in light-duty motor vehicles manufactured after 2001. It was not approved to be used in small gasoline engines or other vehicles built before that due to concerns of material incompatibilities and corrosion. Furthermore, I have seen several places where current automobile manufacturers will not honor their warranties if the person used E15, even if the vehicle was manufactured after 2001. Also, the EPA requires that in order to sell E15 gasoline, a Misfuelling Mitigation Plan must be in place to prevent consumers from using the product in an unapproved engine. Today, there are very few retailers who have chosen to go through the additional trouble in order to sell the E15.
In conclusion, due to the absence of the “One-Pound Waiver” and the legal risk of corrosion or voiding the warranties of customers’ cars, HollyFrontier has chosen not to produce or blend E15.

SUBHASH SINGHAL (Kuwait National Petroleum Company)
Does the 15% have to do with the oxygen in the ethanol and other oxygenate like MTBE, or it is just because of the RVP limitations and other issues that you explained? From safety point of view, is there oxygen contained in the old oxygenate like ethanol? Is that one of the criteria for limiting the blending from 15% or 10%? Does this have to do with the oxygen
attached even though it is oxygenate?
KOONTZ (HollyFrontier)
My understanding, from reading, is that the E15 decision is not really based on logic. I think it was more of a U.S. Congress action. I do not really understand why they have not extended the “One-Pound Waiver” to E15. I do not think it is based on science.
KOONTZ (HollyFrontier Corporation)
The Environmental Protection Agency (EPA) administers the Renewable Fuel Standard (RFS) program with volume requirements for several categories of renewable fuels. EPA establishes the volume requirements for each category based on EISA (Energy Independence and Security Act of 2007) legislated volumes and fuel availability. EPA tracks compliance through the Renewable Identification Number (RIN) system, which assigns a RIN to each gallon of renewable fuel.
HollyFrontier satisfies much of its requirement for Conventional Biofuel (essentially corn ethanol) usage within RFS by selling E10 (10% ethanol) at many of its terminals. Most of HFs’ gasoline is sold via pipeline to terminals owned by others; therefore, to fully satisfy its mandated volume, HF purchases RINs from others. The decision to purchase ethanol from others and blend to E10 or to purchase RINs from others is based on economics.
Ethanol blending has a significant impact on two critical gasoline properties controlled by the refiner: octane and RVP. The hydrocarbon blendstock used for 90% of the E10 product (termed sub-grade by HF) has an octane rating of ~84. After blending with 10% ethanol (octane ~114) the resultant E10 octane is “regular” 87. For a refinery that normally adjusts reformer severity to satisfy the total gasoline pool octane, producing sub-grade allows for lower reformer severity and higher liquid yield. This improved yield is more pronounced for a semi-regeneration reformer that operates at relatively high pressure.
RVP is the other critical gasoline property affected by ethanol blending. When ethanol is blended with naphtha at low concentration, the RVP of the gasoline is increased. Even though pure ethanol has a low RVP [about 2 psia (pounds per square inch absolute)] due to O-H bonding, it behaves more like a hydrocarbon with a molecular weight of 46 when mixed with naphtha at low concentration. If ethanol is blended to 10% with 84 octane naphtha having an RVP of 9, the resultant E10 gasoline has an RVP of ~10. In order to encourage ethanol blending, the U.S. Congress passed the One-Pound Waiver in 1990 allowing E10 gasoline RVP to be 1 psi higher than that normally required by the EPA (One-Pound Waiver).
E15 would allow a refiner to produce an even lower octane sub-grade to blend with the ethanol and the RVP effect would be similar. However, the EPA regulation implementing the One-Pound Waiver specifically references gasoline containing between 9% and 10% ethanol. The EPA has refused to extend the one-pound waiver to E15. Therefore, to market E15 requires a sub-grade blendstock having an RVP over 1 psi lower than that required for E10.
Since January 2011, E15 has been permitted for use in light-duty motor vehicles manufactured after 2001. However, it is not approved for use in small engines and older vehicles due to concerns with material incompatibilities and corrosion. Furthermore, several automobile manufacturers will not honor their warranties if E15 gasoline was used in the vehicle (even for those manufactured after 2001). The EPA requires that in order to sell E15 gasoline, a Misfueling Mitigation Plan must be in place to prevent consumers from using the product in an unapproved engine. There are very few retailers who have chosen to get approval to sell E15.
Due to the absence of the One-Pound Waiver for RVP, the significant legal risk in selling a controversial product, and the minimal market demand HF has decided that it would be unwise to enter the E15 market at this time.
Question 24: Given the potential consequences of back flow in high pressure hydroprocessing services, such as furnace tube rupture and pump shutdown, what layers of protection are being employed to reduce risk?
ESTEBAN (Suncor Energy, Inc.)
We are going to skip the first slide.

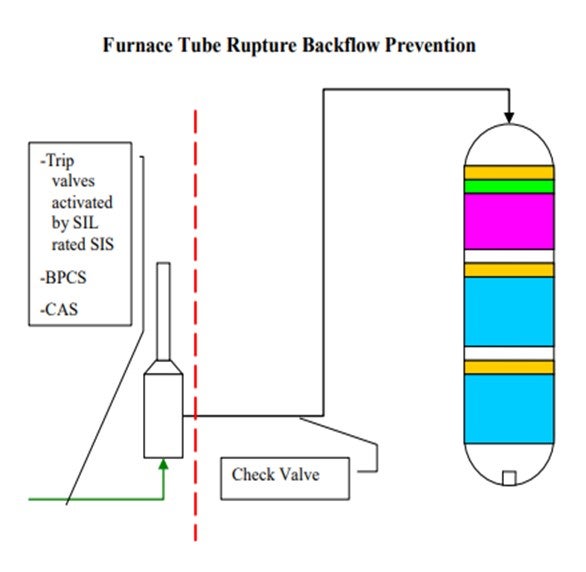
The second slide shows a simple depiction of the layers of protection that we use at our different sites. In some cases, we have relief protection, basic process controls, and critical alarm systems on our feed drums to prevent a backflow scenario or the consequences of a backflow scenario. That being said, though, relief valves do not always provide an adequate level of protection for high pressure units. So obviously, take that with a grain of salt. Our primary layer of protection is provided by our trip valves which are activated by SIL-rated instruments. We do not have an SIL rating in all cases; but in some cases, it is required to get the level of protection we need. And then, of course, we also employ dual check valves of differing types downstream of our pumps. Those check valves will typically wind up on our critical check valve system as well.
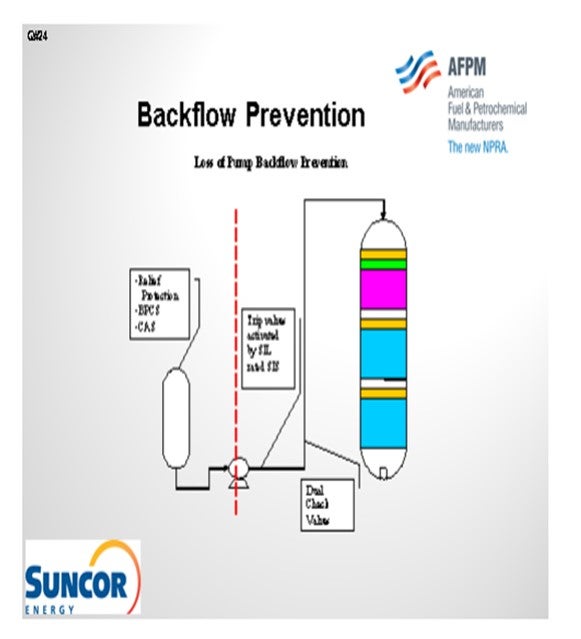
The scenario is similar where you have backflow. It is not so much the concern of backflow of reactor contents through the furnace, but more just a loss of containment in the furnace itself. We do not treat these furnaces in our hydroprocessing units any differently than we do any of our other furnaces in the refinery. They all have an integrity operating window that we would like to stay in. That window defines at what burner pressures we need to operate and, of course, at what skin and overall box temperatures we can operate.
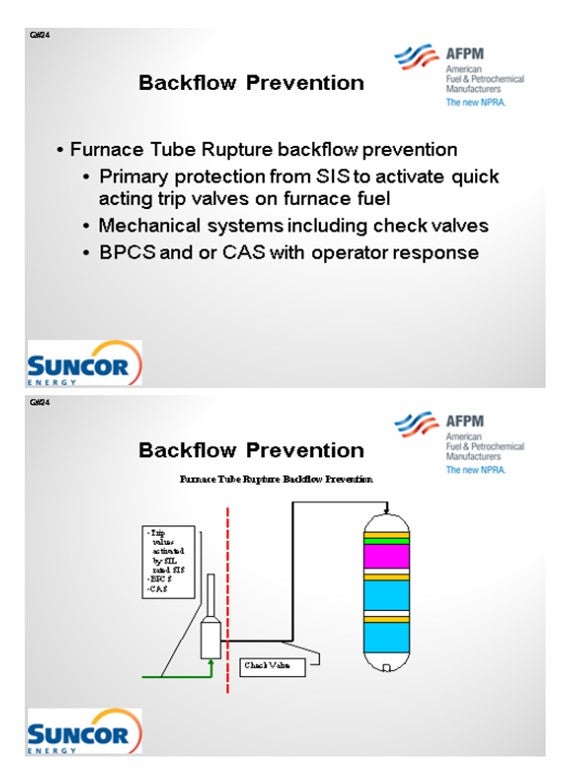
Our layers of protection are very similar here in that we have trip valves activated by SIL-rated instruments and which are only SIL-rated as required. And of course, we have basic process controls and critical alarm systems. In some, but not all, cases, we do have check valves downstream of our furnaces. That is not a standard at all our sites. However, on some sites, we are consistent about having check valves downstream of our furnaces.

KEVIN PROOPS (Solomon Associates)
I would like to comment on the heater part of the question. Reactor charge furnaces potentially have substantially higher consequences of failure than do most of the other furnaces in your refinery, so you need to be a lot more scared of them than you do of the other ones.
First, this is generally an exothermic process; so, the best case is probably that the furnace is not firing or is only minimally firing. Adequate feed-effluent heat exchange reduces firing and thus the risk of failure from flame impingement. Second is the inherent safety design. If you can go to a single-phase furnace instead of a two-phase furnace, then if it does rupture, your consequence will probably be a lot less. That also gets into the control system issues. Some refiners use hot oil utility instead of a fired heater in the hydrotreater. This is inherently safer.
Then you get into how to avoid a tube failure in the first place. There are a lot of ways to do that, but consider dry point in naphtha units, burner ring pressure controls and interlocks, and maintaining the cleanliness of the burners (fuel gas filtering). Adequate burner-to-tube spacing, feed filtration, tube monitoring (thermography), operator rounds frequency, and upgraded tube metallurgy can all add layers of protection.
Finally, it comes down to culture as well. You do not want to get into a situation of risk of a failure competing with profit to keep the unit maximizing at full throughput. Unfortunately, I have seen a case where that did happen: A furnace was experiencing flame impingement, and the operators did not reduce charge to the unit. After one shift, a tube failed, which led to the entire unit being consumed in a flash fire within a few seconds. We were very fortunate that there was no one outside at the time it happened; if there had been, we would have killed anyone in the unit.
ESTEBAN (Suncor Energy, Inc.)
We do treat them differently depending on their operating pressures and/or requirements. Certainly, from a design standpoint, the operating envelope for each individual piece of equipment changes how the furnace is designed overall. That being said, we evaluate all equipment using the same standard with a process hazard analysis to determine the appropriate layers of protection. Given the required layers of protection, we identify additional safeguards as required by LOPA. SIL-rated instruments, for example, are not required for all burner management systems. However, in some cases, they may be required because the consequences of failure are higher.
So yes, the consequences are significantly greater on a high-pressure furnace. The assessed risk ranking would define how those layers of protection will appear. In some cases, you will see a simpler system on a furnace; and in others, much more complex layers of protection will be applied because of the potential consequences of equipment failure for that furnace. So, to re-phrase my response, I will say that we evaluate every piece of equipment using the same processes.
ESTEBAN (Suncor Energy, Inc.)
In order to reduce the risk of potentially catastrophic consequences related to backflow in high pressure hydroprocessing services Suncor Energy, Inc. uses several independent layers of protection at operating pressure boundaries. One common boundary is for hydroprocessing units, is between the unit feed drum and the reactor charge pump. A typical hydroprocessing unit will have relatively low design pressure equipment upstream of the reactor charge pump which boosts the operating pressure of the feed stream to the much higher reactor operating pressure. As such preventing back flow in the event of the loss of a feed charge pump is critical to prevent equipment failure in upstream equipment with catastrophic consequences. In this application Suncor Energy, Inc. applies the use the following layers of protection:
1. Primary protection is typically a Safety Instrumented System (SIS) that monitors the run status of the feed charge pump via multiple direct and indirect instrumented signals and activates quick acting trip valves and in some cases closes the feed charge control valves in the event of a shutdown. In some cases, depending on the unit specific hazard analysis these systems may be SIL-rated to ensure reliable operation when activated. In addition, these systems are often designed to be activated by any one of several different instruments used to sense a potential backflow scenario, i.e., low-low flow shutdowns and low-low feed controller differential pressure shutdowns.
2. In some cases, pressure relief valves are used as layers of protection for overpressure due to backflow, but caution must be applied when relying on a relief valve as protection for vessels, such as feed drums, since these valves are not always sized for backflow scenarios.
3. Mechanical safety systems are also employed depending on unit design. While these systems are often not credited in a process hazard analysis of a unit they can provide additional layers of protection. Typical installations include dual check valves of different design which are often deemed critical check valves that require routine maintenance.
4. Provided the design of the system and equipment in some cases basic process controls and/or critical alarms with operator response are employed as additional layers of protection.

In addition to backflow prevention and protection as it relates to pressure boundaries, furnace tube ruptures can result in backflow from multiple large high-pressure vessels to atmosphere with catastrophic consequences. In order to address the release of reactor and high-pressure circuit equipment, layers of protection must be applied to the feed furnaces that prevent operating windows that have the potential to create damage resulting in tube rupture. The layers of protection employed for this scenario do not differ from those on other furnaces in Suncor’s refineries, as all furnaces are evaluated for tube rupture scenarios. However, in this application Suncor Energy, Inc. applies the use of the following layers of protection:
1. Primary protection is typically a SIS that monitors furnace flows, temperatures, and fuel and box pressures via multiple direct and indirect instrumented signals and activates quick acting trip valves on fuel supply and in some cases closes the fuel supply control valves in the event of operation outside a preset operating window. These SISs often activate related SISs to stop process flows. In some cases, depending on the unit specific hazard analysis these systems may be SIL-rated to ensure reliable operation when activated. In addition, these systems are often designed to be activated by any one of several different instruments used to sense operation outside of the specified window, i.e., low-low flow shutdowns and high-high burner pressure shutdowns.
2. Mechanical safety systems are also employed depending on unit design. While these systems are often not credited in a process hazard analysis of a unit, they can provide additional layers of protection. Typical installations include check valves downstream of furnaces to prevent the backflow of reactor contents. In general, these check valves are not relied upon as fail-safe devices and are not considered critical check valves.
3. Provided the design of the system and equipment in some cases basic process controls and/or critical alarms with operator response are employed as additional layers of protection.

Aimee Ford
Question 13: Severe fouling of diesel and gas oil hydrotreating preheat exchangers has been a growing problem. In your experience, what are the causes and how can these be prevented? Have you tried antifoulant injection in this service?
Dan Webb (Western Refining)
Fouling of the heat exchanger train is sometimes a problem particularly when processing cracked feed stocks. The fouling is often caused by polymer like compounds (gums) that form when petroleum distillates come in contact with air. When heated olefinic compounds react with absorbed oxygen to form gums that deposit in the preheat train.Iron scale and other particulates in the feed often adhere to these gums to produce severe fouling that restricts unit capacity and accelerates heat exchanger corrosion rates. Typically, every effort is made to avoid air ingress into any of the unit feed stocks. Fouling precursors may also be present in straight run feed stocks in the form of certain chemical contaminants that may be present in the crude or inadvertently introduced in an upstream process unit. Some precursors such as amines, carboxylic acids, and carbonyls form gums without air ingress into the feed. Antifoulants have been used successfully to mitigate fouling caused by these compounds in addition to mitigated fouling caused by oxygen contaminate cracked feed stocks.
Michael Chuba (Sunoco)
Typically distillate hydrotreaters exchanger fouling has been associated with cracked stocks that contain olefinic material and trace amounts of O2 coming in with the feed from tankage. In addition to oxygen-initiated polymerization, other impurities can lead to free radical formations that can promote polymerization reactions. These impurities include certain nitrogen and sulfur compounds well as some metal ions including iron, calcium, and magnesium.
In addition to free radial polymerization, condensation polymerization reactions can also result in fouling. In this route, two radicals can react to form a larger molecule. The new compound can continue to react and grow until it precipitates out of solution forming deposits.
What I would like to present here is an example of fouling we had on one of our units and how we have significantly reduce fouling via a simple jump over line.
Prior to conversion of this unit to ULSD the unit processed a mix of virgin and cracked distillate stocks. Historically this unit had exchanger fouling that was attributed to the presence of the cracked stocks. When the unit was converted to ULSD the cracked stocks were removed. The resulting feed was a 50:50 mix of direct rundown material from the crude unit and tankage. As a result of this change in operation it was anticipated that the fouling rate would decrease, however, during actual operation the fouling rate actually increased.
An initial program to address the problem included detailed analysis of the various feed stream followed by a targeted antifoulant chemical injection program. Results were somewhat effective but still left significant room for improvement. Continued investigation into the problem targeted O2 contamination coming from the material coming from tankage. The intermediate distillate tanks are cone roof design which would be relatively costly to convert to blanketed tanks. As a first step it was decided to install a jump over from the tank inlet line directly to the suction of the tanks’ transfer pumps. With this simple connection the average volume of material actually drawn from the tanks dropped dramatically.
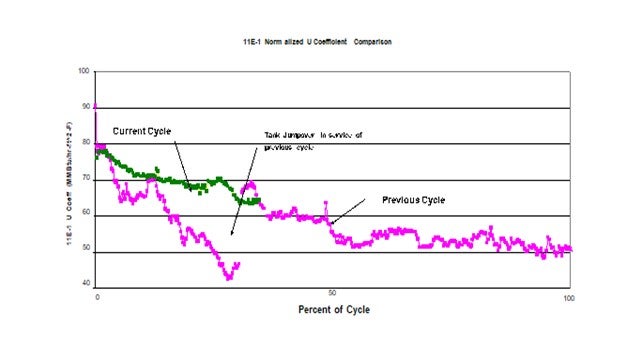
This plot shows the impact on the heat transfer coefficient of the feed effluent exchanger as a result of this simple jump-over. The pink plot represents the previous cycle. At about ¼ of the cycle the jumpover line was installed. At this point significant fouling had already occurred. The discontinuity in heat transfer coefficient a week or two later was the result of a power failure. It is suspected that the rapid depressurization dislodges some of the fouling material thereby improving the heat transfer when the unit is re-streamed. This same response has been seen in previous emergency shutdowns. The green plot represents the current cycle which started with a clean set of exchangers and operation of the jumpover in service from day 1 of the cycle. As can be seen this simple jumpover has significantly reduced the rate of fouling compared to previous cycles. Since the only change was the potential ingress of O2 from the tank, this project confirmed the impact O2 had fouling.
Gregg McAteer (Nalco Company)
Fouling can be a serious problem in hydro-desulfurization (HDS) units because of their importance in producing fuels that should meet environmental specifications. Fouling can limit a unit's ability to maintain a specific feed rate or meet an extended turnaround date. It can greatly influence product quality as well as energy consumption, and catalyst or equipment life. Stricter limits on sulfur and aromatic content of finished fuels make fouling control even more important today. To achieve today’s limits of 0.05 wt.% for diesel, refiners must increase severity of refining operations, which often worsen fouling. Fouling ultimately necessitates shutdown and extensive maintenance, a costly process, both in terms of maintenance expenditures and lost production. Causes of fouling in diesel and gas oil hydrotreaters are both organic and inorganic in nature. The organic foulants are primarily gums formed as a result of processing cracked material and accelerated if the material is exposed to oxygen at any time. Antioxidants and/or antipolymerants are used to reduce the formation of gums and dispersants are used to keep any gums already formed from growing in size.
In one case an antifoulant program utilizing both an antioxidant and a dispersant was applied to a gas oil hydrotreater that normally fouled enough to require a shut down after an average of 440 days. The antifoulant program started on a fouled system and showed
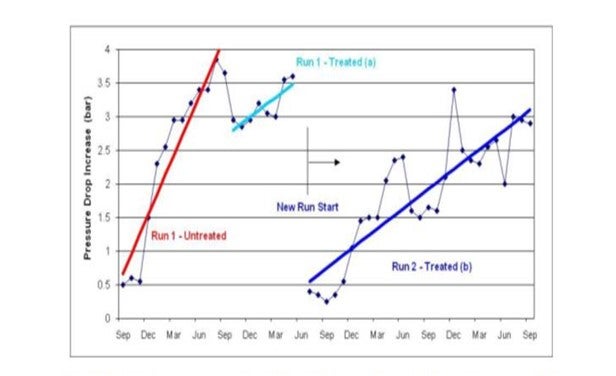
a slight recovery of pressure drop. After a shutdown they started again and achieved a 1300 day run (see graphic below).
“Run 1” is shown in red and light blue. The red trend shows the steep increase in pressure drop during normal operation (without antifoulant program). The light blue trend shows the antifoulant program started, saw a small decrease in pressure drop, and then the unit was brought down for a regeneration. “Run 2” is shown as the dark blue trend and shows a lower fouling rate and longer run length with the antifoulant program. Customer estimated the ROI to be between 400-500%.
Phil Thornthwaite (Nalco Company)
Foulants typically found on the feed side of the preheat exchangers include various gums or polymers, iron sulphide and salts.
The organic fouling due to gums & polymers results from the polymerization of unstable species in the unit feed. The problematic species include olefins (generated in cracking processes), organic acids, mercaptans, ketones, aldehydes, phenols, organo-nitrogen and organo-sulphur compounds. Therefore, in order to determine the risk of organic fouling for a particular feed stream, detailed analysis for the problematic species can be useful guide in evaluating fouling propensity and mitigation strategies.
A typical level for concern for each problematic specie is outlined below:
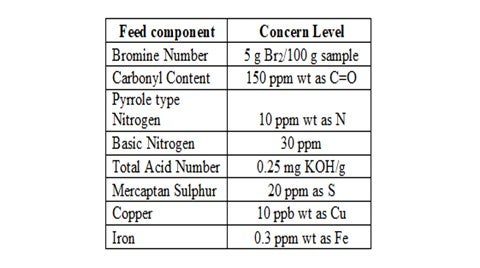
Another key factor to consider is the oxygen content of the feed stream as this can promote the polymerization of various unstable compounds, particularly olefins. Therefore, it is good practice to exclude oxygen from feed storage tanks by ensuring tank seals and vents are in good condition and through the use of a nitrogen blanket. However, this method is ineffective with streams already exposed to oxygen since the nitrogen blanket will have no effect on oxygen reaction products such as aldehydes, peroxides and hydroperoxides.
Inorganic fouling is mainly caused as a result of iron sulphide that can either be carried from upstream units or generated in-situ in the preheat exchanger network. However, the latter is not so common since refiners choose the metallurgy to mitigate against sulphidic corrosion in most cases.
In order to mitigate and control fouling in the preheat train, chemical dispersants and antipolymerants are used. The properly selected dispersant will act upon the organic polymers by keeping them finely dispersed within the feed stream thus minimizing the risk of deposition on the exchanger surfaces. Likewise, dispersants can also prevent deposition of FeS by keeping them dispersed in the feed stream.
Antipolymerants act by disrupting the propagation and chain extending stages of the free radical polymerization reactions and by increasing the rate of termination. This will limit the rate of polymer growth within the preheat system. They will also minimize carbonyl formation which will in turn disrupt condensation polymerization reactions.
The key to monitoring the program effectiveness is through accurate monitoring of the preheat exchanger network. If the fouling results in a limitation of heat transfer efficiency, then a temperature survey of the exchanger network is carried out and this data is entered into a rigorous thermodynamic process model, such as Nalco’s MONITOR® program. This model will then use the plant data to calculate actual and normalized exchanger duties and heat transfer coefficients plus it will calculate the normalized furnace inlet temperature (NFIT). A successful antifoulant program will limit the decay in the NFIT and will generate significant returns for the refiner by improved energy efficiencies and optimized unit operation.
Robert Wade (ART)
We have not had success reducing fouling effects by adding antifoulants. It is our experience that adding antifoulants at best treats the symptom of the problem, and at worst further contributes to localized and downstream fouling. We recommend that the source of the fouling contaminant be identified through analysis and addressed at the source. If this is not possible then we revisit the basic design of the heat exchanger in question and ensure that it is operating in a shear controlled flow regime so that fouling effects are minimized




