


Question 46: Ethanol blending, and benzene management has shifted refinery octane and hydrogen balances. How are you managing hydrogen demand without giving away octane? Conversely, how are you, with excess hydrogen, maximizing availability for volume swell?
Praveen Gunaseelan (Vantage Point Consulting)
Mandated ethanol blending and limits on benzene in gasoline tend to lower the ceiling on the operating severity of catalytic reformers. While lower severity operation is theoretically desirable, in practice it can constrain H2 production, and impact regenerator operation due to lower coke make, which may require operational workarounds.
In instances where reformer derived H2 is a critical feed to other refinery units, the reduced reformer H2 make could be compensated from other sources, such as the refinery H2 plant (if present), pipeline H2 (if available), or installing an adsorption unit to recover H2 from refinery off gases. If these options are not feasible, and the reformer needs to be operated at high severity to meet refinery H2 needs, some octane giveaway may be an economical most favorable alternative.
Brad Palmer (ConocoPhillips)
In general, the lower demand for octane has cut Reformer severity which has increased C5+ yields, reduced coke make (increased catalyst stability and run life) and reduced hydrogen yields. For refineries with the flexibility of alternate hydrogen sources (pipelines or SMRs), the lower Reformer hydrogen yields has not been as difficult to manage; the hydrogen load on these alternative sources has been increased. For refineries with tight hydrogen supplies, this regulation, combined with the new reduced benzene requirements, has required the addition of hydrogen recovery projects and/or SMR revamps to meet hydrogen demand. Octane giveaway has been one of the knobs to optimize refinery economics.
Each refinery operates an LP model to determine the economic optimum given current constraints. In some cases, giving away octane has been an economic decision. Refineries have continued to make changes to reduce octane giveaway if economic to do so; changes have included blending more sweet naphtha and selling blend components on the market.
Most refineries are using hydrogen to meet the desulfurization and benzene saturation demands, but some refineries are operating FCC feed hydrotreaters more severely than required to maximize swell of cat feed. Again, the LP model is used to determine the most economic strategies for each refinery.
Roberto Amadei (Chemical & Energy Development srl)
1. Question 46 first point: How are you managing hydrogen demand without giving away octane? By carrying out a technology of ours it is possible, besides very hugely profitable, increasing by an important quantity the catalytic reforming hydrogen production.
As for the quantity: the gained catalytic reforming hydrogen production - net of isomerization use increase - is estimated to be comprised between around 28 and over 48 percent on catalytic reforming current hydrogen production, without any octane give-away.
In fact: when removing all or nearly all of the C6 molecules (shifting them to the isomerization process) from the catalytic reforming feed - as our technology does - it is experimentally proven, in 2 the refinery, that the catalytic reforming hydrogen production yield increases much, as indicated above, and this is in agreement with the theory in that the reforming feed quality, after the C6 removal, becomes much more favorable to the reforming dehydrogenation reactions, hydrogen producing, and much less favorable to the reforming hydrocracking reactions, hydrogen consuming.
The theory, somewhat deepened, can also give an idea of the hydrogen gain size. We show it in the following paragraphs a. and b.
a. Catalytic reforming reformate octane
Let’s start considering the aromatization catalytic reforming reactions:
1 naphthene = 1 aromatic + 3 H2 – 48-55 Mcal / kmol (1)
1 paraffin = 1 aromatic + 4 H2 – 60-65 Mcal / kmol (2)
As it is of common knowledge, the Δ octane [C8+ aromatics – C8+ (naphthenes + paraffins)] is much higher (around 4 times) in comparison with the [C6 aromatics – C6 naphthenes] one1. The heat absorbed in the catalytic reforming by a one molecule aromatization reaction, generating one aromatic molecule, is, as the above well-known reactions show, roughly speaking about same independently from the carbon atoms number contained in that molecule. Hence: with about the same heat absorption, the C8+ gets a Δ octane, from the aromatization of one molecule, about 4 times higher than the C6.
That is, with reference, as said, to one molecule: with about the same heat absorption, the C8+ gets a Δ octane 4 times higher than the C6 for a gasoline quantity as an average about 40% higher (because the relevant gasoline quantity, exactly the weight quantity, approximately the volume one, is proportional to the molecular weight). 1 As it is well known the C6 paraffins, passing through the catalytic reforming, do not practically increase their octane number. In fact, the C6 paraffins either crack or pass unconverted (exception made for a quota of the normal hexane, having a RON of about 25, pacifically established in a 10% maximum). The hydrocarbons C6, C7, C8, C9, etc. compete with one another in order to make use of the available heat. When the C6 is present it provides a modest octane increase by making use of a heat portion subtracted from the C7+ availability. When the C6 is present in order to increase the available heat, the only option is to increase the temperature.
When, on the contrary, the C6 is removed automatically the reformate octane increases due to the strong octane upgrading become available for the C7+, particularly strong for the C8+, thanks just to the C6 absence: the C6 presence appears to be very harmful for the catalytic reforming performance.
The highest boiling hydrocarbon among all the C6 is cyclohexane, which has in the reforming a fair conversion rate (although lower than the C7+ naphthenes), but unfortunately with a relatively very modest octane increase potential. As a consequence it appears mandatory taking off the reforming feed as much C6 as it is possible. The more C6 is removed the better the reforming performance and the refinery economy.
The above theory that applies to any crude oil and to any refinery configuration is in agreement with the surprising refinery experimental results.
The conclusion is that the process variables being equal and being also equal the catalytic bed temperature and consequently the catalyst life, the reformate octane number increases much: in order to keep it constant it is mandatory decreasing much the catalytic bed temperature.
b. Catalytic reforming hydrogen production
The hydrocracking reactions are rather slow, and are very sensitive to temperature, hydrocracking rates decreasing rapidly with decreasing temperature. Hence, as a consequence of the C6 removal, the reformate octane being equal, the hydrocracking reactions, consuming hydrogen, decrease much - due to the much lower temperature - and much increases the hydrogen production.
But there are other important causes of the net catalytic reforming hydrogen production increase, among which the higher favorableness - that we will not discuss here - for the paraffin aromatization vs. the napthene one. The one paraffin molecule aromatization, as we can see from the (1) and (2) reactions above, produces 4/3 times more hydrogen, or 33% more, than the one naphthene molecule aromatization.
We are fully available for providing details and projects.
2. Question 46 second point: How are you, with excess hydrogen, maximizing availability for volume swell?
The maximum gasoline volume is obtained, in any situation, including the excess hydrogen case, by carrying out the above technology. The relevant gasoline volume gain is higher than 4 %vol on the reformate + isomerate part of the gasoline.
The reasons backing these experimental results, besides paragraph a. above, are the good C6 isomerate octane plus the C6 isomerate yield much higher and the C6 isomerate density much lower than the reformate ones.
As a general information about the technology, worth a few dozen million $ profit per refinery / year, we can yet say that its effects are all positive and, on top of the above, we mention:
- the strong gasoline octane number gain (changeable at will into an additional strong gasoline yield gain),
- the prevention of benzene formation in the refinery,
- the accomplishment with margin of the 0.62 %vol average benzene content USEPA requirement for the whole gasoline pool, resulting in saleable benzene requirement credits,
- the huge energy savings, besides the hydrogen availability gain one,
- the reduction of all the pollutants and greenhouse gas types of emissions, on top of the above, both in the gasoline production and consumption segments,
- the higher-octane gasoline production capacity increase,
- the improvement of the engine operation and maintenance.
As for the above point, we are fully available for providing details and projects.
Erik Myers (Valero)
Valero Reforming units are operating at minimum pressure and minimum chloride levels to maximize liquid volume yield for our low severity gasoline operations. Additionally, it is important to maintain continuous regenerator operation to achieve optimum yields. Modifying the regenerators for maximum coke turndown is important. On/Off catalyst circulation is preferred to circulating coked up catalyst when the regenerator is down. We also believe the key to optimum yields is replacing catalyst when economic, not based strictly on surface area.
Refineries with excess octane simply blend low octane naphtha to reduce gasoline octane give away.
Question 47: What success have you had with installing stabilizer feed chloride treaters in reducing the salt formation and frequency of water washes?
Erik Myers (Valero)
The use of promoted alumina versus zeolite in the liquid chloride treating has several issues. For aromatics operations a Zeolite chloride treater should be selected to protect the downstream aromatics solvent from organic and inorganic chloride. Organic chlorides are not picked up by alumina treating. In gasoline treating the use of promoted alumina is acceptable to Valero.
Since Zeolite chloride treating picks up organic and inorganic chlorides the zeolite beds spend faster which makes the use of zeolite relatively expensive. Organic chlorides decompose at reboiler temperatures (>250 Deg F) in the stabilizer reboiler circuit which is why HCl can be found in the off gas of a stabilizer tower downstream of an alumina chloride treater. Organic chloride can be formed from olefins generated at high temperatures in the Reforming reactors which is a simple equilibrium dependant on operating pressure and temperature in the last Reforming reactor. If a zeolite treater is used in front of the stabilizer column, Valero would expect little to no chloride corrosion or salt deposition and has achieved this at one refinery. Valero allows both the use of promoted alumina or zeolite based chloride treating on the liquid stream entering the stabilizer column. A process design detail often overlooked is that the liquid chloride treater should be installed inside the product separator’s liquid control valve (cold, high pressure, 100% liquid phase) so the treating is liquid phase not two phases. There is no advantage to installing the chloride treater in the hot position on the stabilizer liquid feed unless metal oxides are being used for chloride control.
Michael Windham (UOP).
UOP MOLSIV® sorbent PCL-100 is a molecular sieve designed for inorganic (HCl) and organic chloride removal from liquid hydrocarbon streams. It provides higher dynamic Chloride capacity than conventional doped alumina and metal oxide chemi-sorbents.
UOP PCL-100 will provide longer performance life than doped alumina's or metal oxide chemisorbents. As a molecular sieve, PCL-100 uses the entire particle for adsorption. Its unique structure combined with low coke formation, allows a higher rate of total chloride (inorganic and organic) removal and greater utilization of the sorbent bed. The end result is a higher breakthrough chloride capacity than conventional chemisorbents. True molecular sieve adsorption provides sharp mass transfer zones and removal to ultra-low concentrations of HCl and organic chlorides.
Brad Palmer (ConocoPhillips)
Stabilizer feed chloride treaters have been successfully installed and operated but not for reducing salt formation in the stabilizer. We do not operate our chloride traps as ammonium chloride salt filters; if they are performing this function, it is a side benefit. The best ammonium chloride prevention techniques are those that prevent nitrogen breakthrough to the Reformer, including robust naphtha hydrotreating, robust naphtha stripping, NHT catalyst change out before Si poisoning eliminates the denitrification activity, frequent Reformer feed nitrogen analyses and low-level nitrogen detection.
Chloride treaters have been installed on the stabilizer feed to reduce the amount of HCl and organic chlorides in order to reduce/prevent corrosion in the Stabilizer and downstream systems. HCl in the Stabilizer overhead vapor or liquid ends up being routed to Sat Gas units which are normally wet. Organic chlorides break down in the stabilizer reboiler and/or other downstream reboilers to produce HCl. The first wet points in downstream units are the most likely areas to corrode. Corrosion can potentially result in significant process safety incidents, especially in Sat Gas Units. Chloride treaters have been installed for corrosion prevention/reduction; hence, an improvement in process safety.
Jamie Chisamore (Johnson Matthey Catalysts)
Stabilizer feed chloride treaters can be an effective way to tackle salt formation problems that create the need for frequent water washes in the stabilizer tower. These beds tend to be quite large due to the volume of the feed stream and the liquid hourly space velocities required to overcome the diffusion limitations of treating a liquid stream. With this being said, the rate reductions often required to conduct water washes of the stabilizer tower may justify installing these chloride treaters.
Johnson Matthey Catalysts has a European refining customer that installed lead/lag chloride treaters at the inlet of their stabilizer column more than 10 years ago and has been able to eliminate the need for water washes between turnarounds. This particular customer had severe salt fouling in their stabilizer column that was requiring a water wash every two months. Prior to installing the beds they had worked to lower the nitrogen levels in the reformer feed naphtha, but were still unable to control the salt formation. They then began to work with the licensor of their unit who suggested using feed chloride treaters. They installed PURASPECJM TM 6250 and were able to eliminate the need for water washes between their two year turnaround periods.
Question 48: How does naphtha derived from tar sands and shale oil differ in respect to crude oil-based naphtha? What is the impact on reformer operation?
Praveen Gunaseelan (Vantage Point Consulting)
It is assumed that “shale oil” as mentioned in the question refers to oil produced from hydraulic fracturing of shale reservoirs, as opposed to oil produced from “oil shale” rock via retorting or other techniques. The composition of shale oils, let alone that of their naphtha cuts, is not reported in the public domain. Shale oils are known to be relatively light (40+ API) and sweet. Preliminary information for Bakken shale oil indicates that its properties are similar to conventional light oils, suggesting that its naphtha should have little to no impact on reformer operation.
Tar sands bitumen originally contains negligible amounts of naphtha. However, traded tar sands-based crudes such as DilBit do contain naphtha-range material. In the case of DilBit, condensate (light-naphtha range, saturated) is typically added as a diluent, but is more suitable as olefin cracker feedstock than reformer feed. The upgrading of tar sands-derived bitumen in a refinery, however, can produce naphtha that is suitable as reformer feed. For instance, coker naphtha produced from upgrading of bitumen via delayed coking is relatively low octane, and is suitable as reformer feed provided it can be adequately pre-treated. Tar sands-derived coker naphtha typically contains higher levels of contaminants such as sulfur, nitrogen, olefins, and silica, and, therefore, requires more elaborate pre-treatment compared to crude oil-derived naphthas. The higher contaminant load in coker naphthas tends to shorten the life of reformer catalyst compared to straight run crude oil-derived naphthas.
Brad Palmer (ConocoPhillips)
Naphthas from raw shale oil have high levels of olefins and paraffins, with variable concentrations of napthenes and aromatics, depending on the retorting process used. They are high in nitrogen and arsenic, but tend to be low in sulfur. Severe hydrotreating is required to produce a Reformer quality feed, such that many of the aromatics and all the olefins are saturated. In general, hydrotreated shale-derived naphtha to the Reformer has lower N+2A than hydrotreated crude-oil-derived naphtha.
Naphthas from tar sands are also low in naphthenes; however, the amount of paraffins and aromatics varies widely depending on the type and amount of diluent used. Bitumen diluted with condensate (dilbit) is highly paraffinic and has low aromatics. Bitumen diluted with syncrude (synbit) from an up-grader is highly aromatic. These crudes are also high in nitrogen and sulfur and must be severely hydrotreated with the subsequent aromatic saturation. The N+2A of the hydrotreated tar-sands-derived naphtha to the Reformer can vary widely depending on the diluent, but in general is lower than hydrotreated crude-oil-derived naphtha.
Since these naphthas require severe hydrotreating to remove nitrogen and sulfur, one potential negative side effect is nitrogen breakthrough, ammonium chloride formation and the subsequent reliability issues associated with equipment fouling, water washing and potential for increased corrosion. An increase in feed contaminants such as arsenic will poison the naphtha hydrotreater catalysts faster and could break through and poison the Reformer catalyst as well.
In general, a more paraffinic feed will require higher severity to make the same octane, increase coke make (shorten run length or increase catalyst circulation) and will reduce C5+ and hydrogen yields. As a rough rule of thumb, lowering the feed N+2A 1 lv% will decrease C5+ yields by 0.25 lv% and hydrogen yields by 10 scfb. These yield adjustments can vary depending on other operating parameters but are directionally correct with the right order of magnitude.
Question 49: What criteria do you use to predict coke drum hot spots (leading to blowouts) during the cutting cycle? How do you modify your standard cutting procedure if you anticipate hot spots?
Gary Gianzon (Marathon Petroleum Company)
Coke drum hot spots can be difficult, if not impossible, to predict but there are operating practices that can help minimize the occurrence of coke drum hot spots/blowouts. The operating practices are as follows:
1. Set a target and monitor the coke drum inlet temperature. It is a guide to understanding the actual condition of feed to the drum. High furnace outlet/coke drum inlet temperature improves coke porosity. Our experience indicates that raising the temperature a few degrees can make a huge impact on reducing blowouts/hot drums.
2. Maintain forward flow of steam/water in the drum while transitioning to water quenching.
3. Develop a quench ramp program that fits your specific unit. Initial water rate should be low and gradually increase at a set interval. Pressure override is needed to prevent over-pressuring of the drum during quench.
4. Install a “totalizer” that measures the total volume of water used during water quench and provide guidelines on minimum water usage during drum quench. Low water usage can indicate water has channeled through the coke bed.
5. Install a “totalizer” that measures the total volume of sour water produced during quenching and provide guidelines on minimum sour water make from the blowdown settling drum.
6. Ensure that all the coke drum skin temperatures are below target before draining water out of the drum.
7. Fill the drum with water above the coke bed to the High-level shutoff switch. This ensures that the coke bed as well as the drum metal has been cooled.
8. Install and use controlled back pressure quench either by raising the quench drum settling drum pressure or through a back pressure controller.
If a hot spot is encountered while drilling the pilot hole, our coke cutter follows the “decoking hot spot” procedure to minimize/reduce coke emissions during decoking. Here are the excerpts from our decoking for hot spot procedure:
1. Follow normal procedure for drilling of pilot hole, noting all areas of emissions.
2. De-coke the drum in 5 ft. increments versus 10 ft. during normal cutting procedure.
3. If emissions are encountered, raise the drill steam and allow water to run until emission is diminished
Rajkumar Ghosh (Indian Oil Corporation)
Coke drum hot spots refer to a situation where some part of the Coke bed remain hot, while the other parts get quenched. As soon as this hot part of the coke bed comes in contact with coke cutting water during drilling or cutting operation, it results in sudden vaporization and steam blowout. In our cokers, we have sometimes experienced very hot drums which resulted in blowout during cutting cycle and also observed red hot coke coming out of the drum along with coke cutting water.
Hot drums typically occur because of partially coked hydrocarbon resid collapsing at the top of the coke bed after drum switch and encasing the 800°F hot coke. This pitch type resid prevents quenching some part of the coke bed within the coke drum. When high pressure coke cutting water hits the unquenched part of the bed, the water explosively vaporizes, pressurizing the coke drum and lifting coke out of the coke drum. Hot drums can also occur in the lower portion of the coke bed for variety of reasons viz., poor coke drum warm-up, resid is poorly coked due to low temperatures, formation of large amount of shot coke (which cannot be avoided if feed is of quality that leads to shot coke), shorter cycle length with less quenching duration or improper steaming leading to channeling during water cooling.
Following guidelines are used for predicting coke drum hot spot:
1. Correlating the amount of quench water collected in the blowdown system can help predicting hot spot. Sour water production for 28 ft diameter coke drum is estimated to be 208 m3 /hr based on heat balance. Unusual reduction in sour water generation (below 70%) is a key indicator to poor quench and possible hot spot.
2. Premature water level buildup in the drum during water cooling step indicates less vaporization of water, thereby pointing to inadequate cooling and possibilities of hotspot. Less than normal water consumption could be another key indicator to hot spot.
3. longer time required for depressurization of coke drum after coke bed is filled with water also indicates improper cooling and possibilities of hot spot.
4. Any disturbance or interruption in a particular Coke Drum cycle is a precursor for Hot Spot. Short duration heater tripping, momentary loss of feed to heater/ coke Drum, variations in coke drum inlet temperature and variation in feed quality during the course of coke drum filling are some of the reasons for hot spot in coke bed.
Precautions like drum partial fill & drain, water-over or soaking can be employed to mitigate hot spots.
The frequency and severity of hot spots can be reduced by:
a. Increased COT near the end of cycle helps reducing quantity of uncoked resid at the top of the drum. This is also part of the action for reducing coke VCM.
b. Maintaining adequate Steam Stripping. The rate of steam and the duration of stripping will allow the coking reactions to complete since steam does not greatly cool the coke bed (it is actually heated as it moves through the bed). Higher steam rates will keep flow channels open and reduce channeling in the coke bed.
c. Ensuring that the Coke bed is positively submerged under water towards the completion of quench cycle. Coke drum skin temperatures are closely monitored for:
a. Increased COT near the end of cycle helps reducing quantity of uncoked resid at the top of the drum. This is also part of the action for reducing coke VCM.
b. Maintaining adequate Steam Stripping. The rate of steam and the duration of stripping will allow the coking reactions to complete since steam does not greatly cool the coke bed (it is actually heated as it moves through the bed). Higher steam rates will keep flow channels open and reduce channeling in the coke bed.
c. Ensuring that the Coke bed is positively submerged under water towards the completion of quench cycle. Coke drum skin temperatures are closely monitored for this purpose. Reliable coke drum level indicators play a crucial role. In case hot spot is anticipated, the coke bed should be allowed to soak for at least an hour before going ahead for draining activity.
As per our experience, steam blow-outs generally happen during the initial pilot hole drill of the coke bed. If it is predicted that the coke drum may have hot spots, we go slow to drill the pilot hole @2-3 fpm for such types of drums against normal of 5-8 fpm. During cutting operation, if the operator spots more than normal amount of steam coming out from the reactor, the drill stem is taken up by about 1 meter from that point and left there for 10- 15 minutes for cooling of that region.
Eberhard Lucke (Commonwealth E&C)
I don’t know of any criteria that can be applied to predict hot spots during the cutting cycle. Typically refiners apply certain measures during the quench cycle to potentially identify irregularities like potential hot spots. They monitor drum temperatures and drum pressure to manipulate the quench rate controller. Some operations also use a totalizer for quench water used to fill up the drum to compare to historical data for normal operation. I don’t see any benefit in adjusting your cutting procedure when knowing about a hot spot. The bigger concern is to make sure that no operations personnel (or other personnel) is in the vicinity of the top head opening during these critical stages of the drum operation. Also, consider installing a top head system that includes a containment dome with a vent connection to a safe place to minimize the hazard of hitting a hot spot during quenching or cutting.
Question 50: What unique challenges do you consider when designing a safety instrumented system (SIS) for a delayed coker heater?
Jeff Lewellen (HollyFrontier)
We are currently constructing two new delayed coker heaters at our El Dorado, Kansas facility. As part of this project, we have performed both a process hazard analysis (PHA) and a safety integrity level (SIL) evaluation. We incorporated experience from other recent heater installations during these evaluations. However, unique differences were noted during these evaluations including:
• Heater tube fouling rates commonly resulting in tube skin temperature increases of 1o F to 4o F per day.
• Higher frequency of loss of process flow incidents
• Daily/hourly changes in heater conditions due to drum cycle impacts
• On line heater pass decoking – spalling
• Frequent off line heater pass decoking – steam air decoking/pigging.
• Bringing a heater off line or restarting a heater while the balance of the unit remains running.
• Multiple velocity steam injection locations into the process coils. However, this is common with other “heavy oil” units.
• Double fired coils utilizing shared burner rows with individual pass controls.
• Extended loss of charge heater firing resulting with incomplete coking of the residuum/asphalt in the coke drum.
With the analysis teams consisting of instrumentation/controls, operations, maintenance, engineering, and outside experts discussing these issues, a better design has evolved. Some of the key issues found through these evaluations include:
• As with all of the facility’s heater designs – the teams utilized API Recommended Practices for Fired Heaters 556 and ANSI/ISA-84.00.01 with good engineering practices to implemented adequate instrumentation and controls to maintain safe operation of the heater.
• Ensure adequate equipment and administrative systems are in place to test SIS to maintain reliability.
• Considered the controls and SIS for all of the operation and startup/shutdown modes. This included spalling, steam-air decoking, and shutdown/startup of heater with the unit online or offline.
• Evaluated best practice instrumentation in the design, level of redundancy, and control technologies to optimize for both shutdown and inadvertent heater trip situations.
• When the heater is shut down by the SIS, insured equipment is designed to help the operator quickly recognize the trip has occurred, understand the cause of the trip, correct the condition, and restart the heater if safe to do so. This includes o Utilize Instrumentation/alarm prioritization for “first out” information.
o Facility design and logical location of HMI panels to aid in operator response.
o Complete and detailed operator and maintenance training on these systems.
o an adequate administrative system (including levels of authority) to implement “By-Pass” of the SIS and/or DCS in the event of a malfunction or for maintenance of system.
• Evaluated operating procedures and equipment is in place to safely address the consequences of a heater trip that cannot or should not be restarted those results in a un-coked residuum (tar ball) event.
Gary Gianzon (Marathon Petroleum Company)
The coker heater is unique since a loss of heat or heater shutdown can result in an unconverted pitch “tar ball” in the drum which in itself creates its own hazard. That being said, MPC’s SIS philosophy is to get the heater in the safe state and address the “tar ball” issue through our emergency procedure.
The flame of the low duty burners used in the coker heater can be hard to detect and have caused unnecessary heater trips. This issue was remedied by using a combination of flame rods and flame scanners to prevent spurious shutdowns or trips. We also have emergency steam automatically activated on low pass flow to prevent the heater tubes from plugging which is unique to the coker furnace SIS.
Question 51: What are some steps that can be taken to minimize re-foaming while steaming to the fractionator? Are there ways to predict re-foaming tendencies of different crudes or coker feed properties?
Rajkumar Ghosh (Indian Oil Corporation)
After a Coke drum is taken offline, it is steam stripped with ‘little’ steam @ 2.5 t/hr for 30 minutes (28 ft drum) to main fractionator to recover volatile hydrocarbon material. During this period, following conditions exist in the offline coke drum:
a. As the coke drum is filled to near its maximum allowable level, there is less vapor space available in the drum.
b. Presence of 8 – 10 ft high foam front
c. Reduced system pressure, as the fresh coke drum is yet to achieve its normal Coke drum outlet temperatures
Under these conditions, sudden drop in pressure of the off-line Coke drum results in re-foaming phenomenon. This can happen due to:
a. Switch over being done to a relatively cold Coke drum. Because of the changeover at a lower temperature and very less vapour generation, the overall system pressure comes down till the fresh Coke drum has achieved the overhead temperature of at least 420o C.
b. Very quick changeover in less than 30 minutes in a Coker having 4 or less drums would result in lower system pressure immediately after switching over.
c. If there is condensate carryover into the Coke drum along with ‘little’ steam, it results in immediate pressure surge and subsequent abrupt lowering of pressure. This also enhances chances of refoaming.
The steps taken at our cokers to minimize re-foaming while steaming to the fractionators are:
a. Proper heating of the drum is ensured to avoid switching to the cold drum. We monitor the Coke
drum Skin temperatures and our standard is to maintain these temperatures in the range of 260 –
280 degrees C
b. Condensate draining is done religiously prior to putting in steam to the offline coke drum.
c. The quantity of steam being put into the Coke Drum, to be done in-line with Licensors’. recommendations or based on firm procedures based on experience. Lower steam rate can cause faster rate of pressure decline in the drum and also blockage of channels in the coke bed while higher steam rate can lift the foam front of the filled drum towards fractionator.
d. Antifoam injection is started approx. 4 hrs before switchover and is continued in the off-line coke drum till it is lined up to blowdown. If frequent problems of refoaming are being faced, the rate of antifoam injection can be increased during switch over.
e. In case high system pressure drop is observed during and after the drum switch, we have a practice of increasing the set point for Wet Gas compressor suction pressure thereby reducing the RPM of Compressor.
Generally, we ensure that rate of pressure fall in the offline coke drum is in the range of 0.22 to 0.25 psi/ min immediately after switching over. This ensures that refoaming doesn’t take place. (Trend of coke drum pressures during a typical switch over is attached)
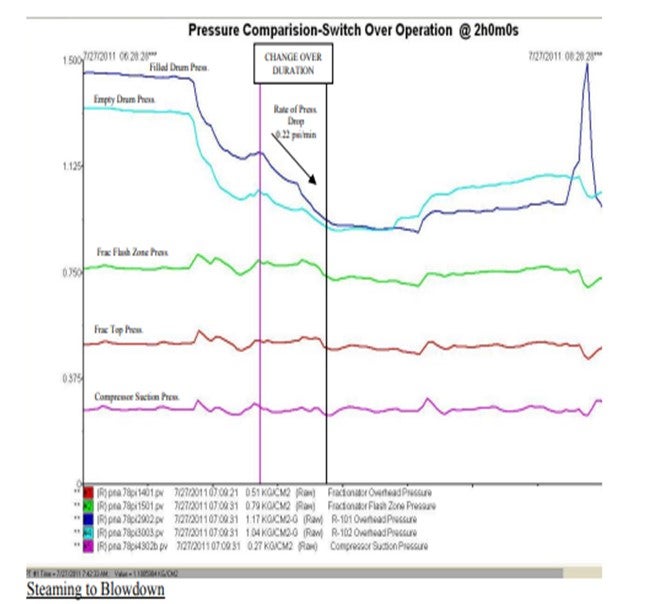
Once steaming towards fractionator is over, the steam and residual vapour from the drum overhead is routed to the blowdown system and steaming is ramped up to 16-18 t/hr (28 ft Coke Drum) and continued for 1.0 hr. If this switch to blowdown is performed too fast, it might again cause a sudden pressure drop in the drum which might uplift the foam bed. In some instances, foam and other material have been sucked in the drum vapour line and caused severe fouling and plugging.
As there is a significant pressure difference between the offline drum (1.1 kg/cm2 g) and the blowdown system (0.3 kg/cm2 g), care is to be taken by the operator to perform the switch to the blowdown system smoothly. We advice our operators to fully close one of the Vapour valves prior to opening the Blowdown valve. Further, the blowdown valve opening is to be manipulated through ‘inching’ control for slowing depressurizing the Coke drum pressure to Blow down.
As a best practice, we have configured the layout of Vapour valves, Blowdown valves and the Coke Drum pressure local indication in such a way that the valves can be operated with an eye to the pressure indication.

Prediction for refoaming
Data for predicting refoaming tendencies based on Coker feed properties is not available with us. However, it’s understood that Coke drums experiencing the following aspects may end up having tendency to refoam :
a. Sharp variations in feed quality w.r.t. API gravity
b. Light oil ingress in Coker feed through slop route
Jeff Lewellen (HollyFrontier)
I concur with the primary answer, and would like to highlight two additional events when foaming can be a significant issue:
o Establishing steam (or re-establishing feed) to a drum following a charge heater upset may result in a sudden foaming event. Typically, this occurs following loss of charge heater firing, or during startup and shutdown activities.
o Our experience indicates heavier unit feeds allow us to operate at lower heater outlet and drum outlet temperatures while maintaining same coke qualities. However, the lower drum outlet temperature tends to form a shorter, denser, and more stable foam layer that is more difficult to collapse. Higher antifoam rates are required to prevent a foaming event.
Eberhard Lucke (Commonwealth E&C)
Foaming during the steam stripping step can occur for different reasons. The most typical mechanism I know of is foam entrainment when depressurizing the drum from fractionator to blowdown pressure. This can be controlled by the speed of depressurization (slow opening of blowdown valve). In general, to see foaming during the steam stripping step there has to be significant amount of hydrocarbon liquid sitting on top of the coke bed. Not going into all the details of what feed properties influence this behavior, all residues that coke relatively slowly (typically high aromatic coke content, low asphaltenic coke content) will leave more liquid on top of the coke bed after switching the coke drum. This liquid layer can foam up if for example the steam stripping flow rate is ramped up too fast, or if there is condensate entering the drum with the steam that spontaneously vaporizes and causes a peak in steam flow through the coke drum. One idea to think about: ramp up the coil outlet temperature for 15-30 minutes at the end of the drum cycle to boil off more of the liquid from the coke bed.
Question 52: What test methods do you use to predict desalter performance when switching to new crudes or a blend of several crudes?
Tom Collins (Forum Energy Technologies)
Standard laboratory tests include salt, BS&W, gravity, viscosity, and filterable solids. The amount, and type of filterable solids can be the greatest factor to consider for interface emulsion resolution and predicting performance. Testing can be time consuming, and of little value without correlations to commercial desalter performance, so a database of crude types and properties is advantages. Typically blending of several crudes in a charge tank may offer little time to accurately predict performance, in which case the best tool is experience. Along with the standard analytical tests, during operation, volts & amps should be monitored, along with interface emulsion composition and effluent water quality, all of which can be used to predict desalter performance, and help establish a data base.
Question 53: Asphaltenes are known to destabilize at higher crude preheat temperature. Are there guidelines that can be used to determine the maximum allowable desalter operating temperature before the asphaltenes begin to drop out of solution and come out in the desalter brine?
George Duggan (Baker Hughes)
We have worked with several refining organizations that process heavy Canadian crude oils to find the optimum desalter operating temperature. In general, raising temperature reduces the crude oil viscosity, which is desirable from a Stokes’ Law stand point as it makes breaking the emulsion easier. On the other hand, asphaltenes are destabilized by factors such as solvency, pH, and increasing temperature. In some crude blends, maximizing desalter temperature in an effort to improve oil-water separation has led to asphaltene destabilization, which contributes to emulsion band growth and negates the benefit of lower viscosity. The degree of asphaltene destabilization required to induce desalting problems is often less than that required to manifest itself as heat exchanger fouling. As mentioned on the previous question, our ASIT technology is used to gauge the relative potential for asphaltene instability in crude blending, both for desalting concerns and preheat fouling. Efforts to optimize desalter temperature have led to operating temperature targets to balance the needs for reducing viscosity with minimizing asphaltene instability effects. The customers who have gone this route consider the optimum temperature target confidential information, so we are not at liberty to share specific operating targets. For refiners who are unable to stay at or below the target temperature, we apply asphaltene stabilizer chemicals in the crude storage tanks to improve asphaltene stability and prevent desalter emulsion problems.