Question 51: What is your minimum Cloud Point Improvement that makes economic sense to apply catalytic dewaxing vs. traditional (i.e. additives and kero blending) Cold Flow Improvement methods?
Renata Szynarczuk (Criterion Catalysts & Technologies)
At low temperatures, products with “waxy” components start to crystallize and affect the flow characteristics of the product. To avoid problems of fuel supply to an engine or lubricating problems under low temperature conditions, diesel fuels and lube oils often have stringent specifications on flow properties at low temperature. To ensure that the various products meet low temperature flow properties, three main cold flow property specifications are used. Pour point (PP) is mainly used for base oils specifications. Cloud Point (CP) and Cold Filter Plugging Point (CFPP) are usually used for diesel fuels.
Small amounts of cold flow improvement additives can reduce the CFPP and PP significantly. However, CP cannot be significantly improved using additives and/or kerosene blending. With additives, CP can typically be improved to a maximum of 5-7°F (2-4°C) and for every 10 vol% of kero blended into the final product; the CP is reduced by ~2°F (1°C). Kerosene blending; however, is limited by other product specifications (e.g., Flash Point). Catalytic dewaxing is usually the most economically attractive solution for middle distillates if the CP improvement requirement is higher than 10 - 12°F (5-7°C).
Catalytic dewaxing is typically employed in either a single stage or in a two-stage configuration. In single stage dewaxing the dewaxing bed is part of the main hydrotreating section, and a base metal dewaxing catalyst (SDD-800) is used that can withstand the severe operating conditions that are encountered. Single stage dewaxing can provide a low-cost and flexible solution. For an existing unit, a number of key checks are required to ensure that a drop-in solution is feasible.
In Two Stage Dewaxing the feedstock is subject to hydrotreating in the first stage of the process after which it is separated from the gas phase containing H2S and ammonia and treated with the dewaxing catalyst in a second stage. This provides a cleaner environment (low sulphur and nitrogen), so that a high activity noble metal catalyst (SDD-821) can be used. Investment costs may be higher with this configuration, but product qualities and yields can be maximized.
In addition to the minimum required level of Cloud Point improvement, the attractiveness of the catalytic dewaxing solution is highly dependent on available unit hardware (number and size of reactors/beds, inter bed quench and appropriately sized separator and fractionation capability). Brian Watkins and Charles Olsen (ART) The target market for these products requires much more than several degrees decrease in cloud point below the value of the feed which is generally beyond that observed by hydrotreating alone. Whether the application of catalytic dewaxing makes sense needs to be balanced against the costs/benefits of other cold flow improvement methods.
The ability to improve the cold flow properties of the diesel in the hydrotreater using a dewaxing technology can have significant economic advantages that other options do not provide especially if the degree of cold flow improvement desired is high. Understanding the cold flow requirements is necessary to create an individually tailored process and avoid the pitfalls associated with inappropriate quantities of HDW catalyst such as yield losses and not having the flexibility to meet market demands.
The typical process of dewaxing utilizes a ZSM-5 type catalyst. The structure of ZSM-5 is such that only straight chained hydrocarbon molecules (normal paraffins or n-paraffins) fit inside the cage structure and are cracked into smaller, lighter molecules. These molecules have significantly lower cloud and pour point characteristics. One of the keys to successfully combining a dewaxing catalyst with an HDS system is an understanding of the tradeoffs between dewaxing and HDS activity as the amount of dewax catalysts changed. Similar to a hydrocracking reactor, as the temperatures are increased over the bed of HDW catalyst, the ability to break the n-paraffins increases. The extent of cloud point reduction at a given temperature depends on the LHSV over the dewax bed as well as the hydrogen partial pressure of the unit. Once the temperature is high enough to begin dewaxing, the kinetic response for converting the n-paraffins is linear for both catalyst systems.
As discussed earlier, the HDW function is to break the n-paraffins into smaller molecules in order create less waxy molecules in the finished diesel product. This chain breaking reaction does have the potential disadvantage that it can convert diesel boiling range material into naphtha and lighter materials. These materials, if the refinery is able to tolerate and utilize them, can be considered quite valuable as well. One concern is that if a significant volume of diesel is converted to lighter products, the downstream equipment might not be capable of handling excess light materials.
ART’s pilot testing has shown that there is a complex interaction between dewaxing and hydrotreating in ULSD applications. There is a balance between dewaxing activity and HDS activity which needs to be understood when designing a catalyst system. Furthermore, the liquid yield needs to be considered at both SOR and EOR as this is highly dependent on the amount of dewax catalyst in the system. ARTs technical services staff can work with refiners in order to provide the right catalyst system tailored for maximum refinery profit.
Question 52: Explain the differences with renewable and Biodiese. Can renewable feedstocks be co processed with traditional diesels in an existing ULSD (Ultra Low Sulfur Diesel) unit and how does this impact RIN's (Renewable Identification Number)?
Celeste McLeod (CRI)
Renewable Diesel is a hydrocarbon. Commercially, Renewable diesel is made from hydrotreating vegetable oil & animal fat. Renewable diesel must meet the specifications for diesel, either ASTM D 975 or D 396 in the United States.
Biodiesel refers to Fatty Alkyl Methyl Esters that have been produced by the transesterification of vegetable oils or animal fats. The resulting FAME or "Biodiesel" product still contains oxygen, and is therefore, an oxygenate. Specifications for biodiesel are contained in ASTM D 6751.
Renewable feedstocks, i.e., vegetable oil and animal fats, can be co-processed with traditional diesels in existing ULSD units. Catalyst selection is a consideration as these feeds contain oxygen. Additional considerations that should be reviewed during a management of change or equivalent process evaluation would include things such as:
•New feed components and side reactions
•Higher hydrogen consumption
•Accumulation of CO, CO2 and other lights in recycle gas
•Undesirable product cold flow properties depending on co-processing percentage in the feed
•Fouling resulting from poisons (Na, P, Cl) and diene formation and polymerization
•Coking and corrosion
How does this impact RIN's (Renewable Identification Number)? A RIN is a 38-character numeric code that corresponds to a volume of renewable fuel produced in or imported into the United States. RINs remain with the renewable fuel through the distribution system and ownership changes. The RFS mandates are prorated down to “obligated parties”—individual gasoline and diesel producers and/ or importers—based on their annual production and/or imports. Under the Renewable Fuels Standard (RFS2) only certain pathways qualify for RINs. The eligible pathways for generating RINs are defined by their renewable biomass feedstock input, the technology used for processing, and the resulting fuel products. Each pathway is assigned a D-code that groups all resulting pathway derived fuels together.
A table containing eligible feedstock, pathways and fuels produced is available at the EPA website: http://www.epa.gov/otaq/fuels/renewablefuels/new-pathways/rfs2-pathways-determinations.htmPertaining to vegetable oil neat processed and vegetable oil co-processed, they would each have to meet the prescribed greenhouse gas reduction, etc., excerpt from above website referenced table
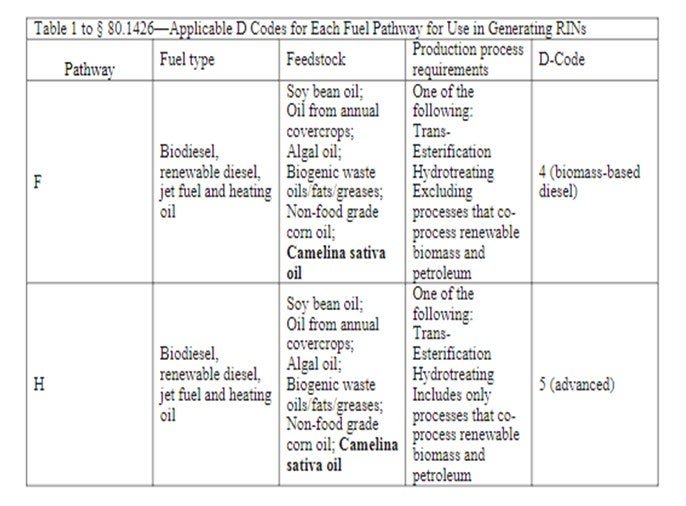
Operations that meet the 100% vegetable oil/animal fat processing requirements and greenhouse gas reduction would likely be successful in fuel registration for a D-Code of 4, allowing for generation of RIN’s once the facility registration process was completed and approved.
Operations co-processing the vegetable oil/animal fat and greenhouse gas reduction would likely be successful in fuel registration for a D-Code of 5, allowing for generation of RIN’s once the facility registration process was completed and approved.
Approval of such lies with the US EPA, and refiner’s own evaluation group should determine the best application scenario for their purposes.
Brian Watkins and Charles Olsen (ART)
Fuels that are produced when vegetable oils or animal fats are processed in traditional refining processes are considered Co-Processed Renewable Diesel. Some common sources of renewable feeds are those produced for food grade oils such as soybean, rapeseed and other vegetable oils. The traditional process for introducing these sources into the diesel pool is to use the transesterification reaction for breaking the glycerol from the fatty acid chains.
Advanced Refining Technologies’ (ART) has completed extensive pilot plant studies demonstrating that these types of feeds can be co-processed with traditional diesels in an existing ULSD unit. Refiners who use the co-processing method would have exact knowledge of the bio-based fuels that are incorporated into the diesel pool which helps to ensure the finished blend quality.
One consequence of co-processing is an increase in n-paraffins in the diesel. This can be of significant value for ULSD as they have typical cetane numbers ranging from 95 to 110 which can provide a significant boost for those refiners processing feeds with lower cetane (i.e., FCC LCO’s). The typical diesel hydrotreater has only a small effect on cetane with cetane upgrade of about 2-4 numbers.
Another concern is that these renewable feed sources can include various contaminants. An analysis of several different bio feed sources has indicated the presence of contaminants such as sodium, calcium and phosphorus. Since these renewable feeds are derived from a biological source, they also contain a high concentration of oxygen. The oxygen content ranges from 10 to 15% and is entirely dependent on the length and degree of saturation of the fatty acid chains. This quantity of oxygen is important, as under normal hydrotreating conditions the oxygen will react with the hydrogen to form water. This water, if generated in a significant enough quantity, may cause problems such as weakening the catalyst support or redistribution of the active metals and loss of surface area. At the expected blending ratios of 10%, the oxygen content is around 1 to 1.5 wt%, and even if all of the oxygen is converted, this is unlikely to generate enough water to be a significant problem.
Due to the addition of unsaturated chains from the bio component, there is expected to be an increase in hydrogen consumption to saturate these C=C bonds. The co-processing of bio components may yield improvements in the diesel cetane due to the increase in n-paraffin content in the diesel product. These n-paraffins, however, raise concerns about the bio feed-based diesel cloud point. Normal paraffins have a significantly higher cloud point than other hydrocarbons of similar carbon number. Since hydrotreating converts the fatty acid chains into long chained n-paraffins, it is expected that the cloud point of the hydrotreated product will increase compared to the product using the SR base feed.
Tony Barnette (UOP)
Biodiesel (or FAME- Fatty Acid Methyl Ester) is a product of the transesterification of oils, fats, or grease, resulting in the production of FAME and glycerin byproduct. Renewable Diesel is the product of hydroprocessing of oils, fats, or greases, resulting in a full hydrocarbon product (first as n-paraffin, followed by isomerization to produce a high grade, isomerized hydrocarbon product with properties verisimilar to petroleum diesel. High value propane and naphtha by-products are also produced. Other important differences to note:
•The FAME product is an oxygenate, and as such, is not as stable as conventional diesel, or renewable diesel, both of which are full hydrocarbon products. As a result, FAME cannot be transported in existing diesel pipelines, whereas Renewable Diesel can, greatly reducing transportation costs of delivering this fuel.
•Renewable Diesel has both a higher cetane number and lower density than either FAME or petroleum diesel, allowing it to be an excellent blend stock into a refiner’s diesel pool. Blending of Renewable Diesel allows the blending of lower value feed stocks with higher density and low cetane number- creating a synergistic effect and increasing the value of these lower quality feeds.
•Renewable Diesel has higher RIN value (1.7 RIN/ gal for Renewable Diesel, vs 1.5 for FAME) due to higher energy content.
•Higher value byproducts (propane and naphtha for Renewable Diesel, vs. glycerol for FAME), which can be used within the refinery, with existing infrastructure for handling and transportation.
•Renewable Diesel cold flow properties generally can be adjusted to meet seasonal requirements, without the need to switch to higher cost feedstocks- a two stage process for producing Renewable Diesel can maintain excellent cold flow properties while processing lower grade feedstocks simply by adjusting the degree of isomerization in the second stage, whereas in production of FAME, cold flow properties are dependent on the feed used (i.e. when lower cloud point fuel is required, the process must switch to higher cost feeds that can produce FAME with lower cloud point).
Can renewable feedstocks be co processed with traditional diesels in an existing ULSD (Ultra Low Sulfur Diesel) unit and how does this impact RIN's (Renewable Identification Number)?
Tony Barnette (UOP)
Renewable feedstocks (such as vegetable or seed oils, used cooking oil, or animal fats) can be co-processed in an existing diesel hydrotreater unit with limitations. These limitations include:
•Byproducts- renewable feedstocks contain significant oxygen, and when hydroprocessed this oxygen forms either water or carbon dioxide in quantities that a typical distillate hydrotreater is not equipped to remove.
•Heat Release- renewable feed stocks have a much higher heat of reaction, resulting in much higher heat release in the reactor. Generally distillate hydrotreaters are not designed to handle this high heat release, and may exceed to units recycle or quench capacity. Higher temperatures that may result from this high heat release can also reduce catalyst life.
•Generally, without modifications, conventional distillate hydrotreater systems are only able to process ~ 5 vol% renewable feed stocks without modifications to handle the above issues.
Additionally, there are disadvantages to producing renewable fuel via co-processing, such as:
•While co-processing does not affect the value of the RIN for the product, Renewable Diesel produced by co-processing is not eligible for the current $1/ gallon blender tax credit.
Renewable Diesel product is not available as a neat product, and is therefore not available as a separate product that might be sold at a premium.
Question 53 How are you planning to meet Tier III gasoline specifications? Please share your strategies for post-treat and pre-treat options, feedstock selection and catalyst technology? Can renewable feedstocks be co-processed with traditional diesels in an existing ULSD (Ultra Low Sulfur Diesel) unit and how does this impact RIN's (Renewable Identification Number)?
Amit Kelkar and James Esteban (Criterion Catalysts & Technologies)
FCC gasoline has the highest sulfur content and is also the largest volume component of the mogas pool of a typical US refinery. Consequently, most refiners are evaluating options to reduce FCC gasoline sulfur (to 20 – 30 ppm) to comply with Tier III regulations. Options under consideration include FCC feed pretreat, FCC gasoline post-treat or a combination of the two.
The CAPEX for a grass-root FCC feed pretreat unit is significant; however, HDS performance of existing pretreat units often can be improved with minimal capital investment. In some cases, simply increasing operating severity while utilizing the best available catalyst technology such as Criterion’s CENTERA® technology provides an acceptable cycle life and meets desired product specifications. In addition, many refiners are considering the addition of reactor volume, improvement of catalyst utilization by installing new reactor internals, and hydrogen partial pressure improvements via compressor modifications, recycle gas scrubbers, membrane units, and higher purity hydrogen sources. These changes in modes of operation and modifications are critically tied to improvements in catalyst performance and technologies. In many refineries the drive to improve product yields and maximize profitability has made FCC feed pretreat a highly desirable and profitable process. \
Several refiners, working closely with Criterion, are evaluating options for improved operating severity of FCC feed pretreat units targeting maximum aromatics saturation for improved FCC yields and profitable hydrogen upgrade. Increased FCC pretreat severity results in improvement in the quality of the FCC feed leading to higher conversion and improved yields. This option is constrained by the pressure rating & H2 availability of the pretreat unit; low pressure pretreat units (< 800 psi) with MU H2 limits are not typically suitable for higher severity operation. It is important that the capabilities of each unit be carefully considered using resources such as catalyst manufacturers and technology licensors to evaluate potential for meeting refinery specific targets.
CAPEX for a new FCC gasoline post-treat unit is lower than for pretreat and can be an attractive option. Post-treat technologies which reduce FCC gasoline sulfur content while minimizing olefin saturation & octane loss are well established in the industry. Octane loss is expected to increase at the higher severity required for Tier III; recent developments in post-treat catalyst can be employed to minimize this loss.
In conclusion, a strategy to comply with Tier III regulations depends on each refinery’s unique configuration and constraints. In most cases, it involves a combination of changes to both post-treat & pretreat operation.
A paper presented at the Annual Meeting earlier this year discussed this issue in more detail (Tier III Capital Avoidance with Catalytic Solutions by Patrick Gripka, et al.)
Roberto Amadei (Chemical & Energy Development srl.)
A pre-condition, to be carried out, of the optimum strategy for compliance with Tier III gasoline specifications and ULSD renewable and traditional diesel specifications is the following (already outlined above in our response to Question #7).
The naphtha catalytic reforming unit has to be partially unloaded, by subtracting from its traditional feedstock the higher-boiling C6 hydrocarbons, including naphthenes, benzene and hexane. Typically, the optimum allocation of this material unloaded from reforming is the isomerization unit. The deriving set-up of reforming and isomerization has the potential of generating value in several ways and in no way destroys any value. However, also in case of an allocation of the above material different from the isomerization, its unloading from reforming keeps a significant potential of generating value.
The main components of the optimum reforming + isomerization set-up generated value are the following:
-hydrogen net production gain,
-gasoline yield gain,
Gasoline octane number gain, changeable at will into an additional gasoline yield gain, compliance, with margin, with the most severe limits of gasoline benzene content in the world, such as the 0.62 vol% content required by the United States Environmental Protection Agency, in the US resulting in saleable benzene content credits, significant energy savings, besides the hydrogen net availability gain one, emissions reduction of all of the pollutants and greenhouse gases types, in addition to the above, both in the gasoline production and consumption segments, also carrying out a gasoline lifecycle emission reduction of a few percent of CO2 equivalent, higher octane gasoline production capacity increase, improvement of the engine operation and maintenance.
The case study experimental results pointing out the above and the relevant theoretical explanation can for instance be seen in PTQ and Digital Refining 2013 Q1, article “Improved hydrogen yield in catalytic reforming”, or in “Gasoline Processes”, 2011 NPRA Q&A and Technology Forum.
Referring to the particular North America case, we premise that the optimum set-up of reforming and isomerization carries out the production of gasoline and hydrogen in lieu of fuel gas. With this due premise, we can conclusively deduce that the above hydrogen gain is much more convenient than the hydrogen production carried out by means of special units consuming the cheap shale gas (SMR).
Precisely, neglecting here the gasoline-fuel gas replacement value, said hydrogen production gain is over three times cheaper, as far as the variable (operating) costs alone are concerned. In fact, in the case of the optimum reforming-isomerization set-up, the shale gas should be used, for combustion in the furnaces, in order to replace the fuel gas not produced anymore by reforming. In such a way the rate of substitution of fuel gas by shale gas is 1:1. On the contrary, any use of shale gas for producing hydrogen would require the consumption of more than 3 units of shale gas (taking into account all the energy flows, both consumed and produced by the SMR unit) per each unit of produced hydrogen (rate of substitution: >3:1).
Moreover, depending on the specific refineries, the relevant hydrogen gain can even avoid the capital costs of either installations or revamps or even duplications of the special, highly energy consuming, hydrogen generation units.
The FCC-gasoline pre-treat and post-treat options and the ULSD processes consume hydrogen and energy. Furthermore, the FCC-gasoline post-treating causes reduction of the FCC-gasoline octane number and yield, due to saturation of high-octane olefins.
It is apparent that the optimum set-up of reforming and isomerization, as it provides hydrogen gain, reduction of energy consumption and gasoline octane plus yield gain, counteracts the FCC pre-treat and post-treat options and the ULSD processes negative effects. Plus, it provides additional very low sulfur combined reformate-isomerate gasoline blending component, due to its yield gain, thus allowing a higher sulfur content of the FCC-gasoline for a given full gasoline sulfur content: this allows to carry out a further reduction of the FCC pre-treat and/or post-treat options negative effects.
The two last paragraphs outline the qualitative aspect of the matter. As far as the quantities in play are concerned, HOP (Hydrogen-Optimization) analyzes and optimizes the operation and any asset of the specific refinery as a function of the specific refinery plant structure, refinery supply slate and predicted upcoming additional needs of hydrogen, energy, gasoline octane and gasoline yield, also providing alternative cases results.
Here we owe an explanation: HOP is an Alliance established between Chemical & Energy Development and Prometheus, rendered very suitable by the worldwide hydrogen thirst that deserves the maximum management efficiency. Chemical & Energy Development brings to the new Alliance its deep knowledge and practice of the specific, above indicated, technology and Prometheus brings to the new Alliance its deep knowledge and practice of planning and optimization procedures and of refinery engineering design.
The resulting project gives a substantial contribution to meeting the Tier III gasoline and ULSD specifications.
Brian Watkins and Charles Olsen (ART)
Tier 3 gasoline regulations are requiring that all gasoline is to contain ≤10 wppm sulfur. This is a decrease from the current Tier 2 regulations of ≤30 wppm sulfur. This change in product sulfur can have a dramatic impact on refiners that have a significant portion of their gasoline pool generated from the FCC. In order to meet this regulation, there are only two methodologies to reduce the gasoline sulfur pool, and both involve some level of hydrotreating, either before or after the FCC unit. The process of hydrotreating after the FCC unit however can have a dramatic effect on the gasoline octane levels due to the additional olefin saturation that occurs when removing the last amount of sulfur.
For those refiners that utilize a hydrotreater in front of their FCC unit, changes to that operation need to be considered in order to meet the desired product targets. To address these needs, Advanced Refining Technologies LLC (ART) utilizes the ApARTTM Catalyst System for FCC pretreatment. This technology is designed to provide significant increases in HDS conversion while at the same time providing significant upgrading of FCC feedstock quality and increasing yields. In essence, an ApARTTM Catalyst System is a staged bed of high activity NiMo and CoMo catalysts where the relative quantities of each catalyst can be optimized to meet individual refiner’s goals and constraints. ART has continued to develop a better understanding of the reactions and kinetics involved in FCC pretreating, and through its relationship with Grace Davison Refining Technologies, a detailed understanding of the effects of hydrotreating on FCC unit performance.
It is clear that FCC pretreating plays an important part in reducing the sulfur content of FCC products. ART has completed many studies looking into the effects of hydrotreating on FCC performance and the quality of the FCC products. The work demonstrates that reducing the sulfur in FCC gasoline and LCO simply requires a reduction in the sulfur of the FCC feed by increasing the severity of the pretreater. Figure 1 shows the relationship between FCC feed sulfur and the resulting sulfur of the FCC gasoline.
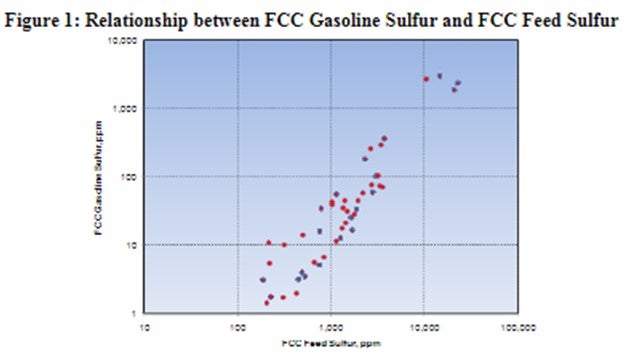
This data was generated using a variety of FCC feeds that had been hydrotreated over several types of catalysts and catalyst systems. As can be seen in the chart, there is a good correlation between FCC feed sulfur and the corresponding FCC gasoline sulfur. In this case, the sulfur content in the FCC gasoline is roughly 100 times less than the sulfur in the feed to the FCC. Increasing the severity of the pretreater operation in order to drive down the product sulfur will tend to move the catalyst towards more of a PNA mode of operation. This mode of operation, while beneficial to the FCC in many ways, can impact the cycle length due to the increased temperatures. Both the hydrotreating catalyst system and the operating strategy for the pretreater are critical to providing the highest quality feed for the FCC. Driving the hydrotreater to remove nitrogen and PNA's improves FCC product value when targeting gasoline production, but this needs to be balanced against the increased costs of higher hydrogen consumption and shorter cycle length that result from this mode of operation. Use of tailored ApARTTM catalyst systems with 586DX and AT795 can optimize the FCC in order to produce not only high-quality feeds to the FCC but also lower sulfur products resulting in less impact on downstream hydrotreating. This tailoring can also be beneficial if the FCC products are able to be blended directly without hydrotreating, as they can be driven towards lower sulfur and higher gravity products allowing the refiner to be able to blend these fuels directly without sacrificing the FCC gasoline octane level.
Question 55: What operating procedures and designs are you using to increase the safety of drilling coke drums?
Question 56: How will the recently announced EPA requirement to depressure coke drums to below 2 Psig prior to venting to atmosphere regulations impact your coker operation and design?
John Metka (Foster Wheeler USA Corporation)
In order to meet the potential requirement of less than 2 psig prior to atmospheric venting, new units designed to operate at low coke drum pressure will require additional equipment such as an ejector system discharging to a condenser (potentially the blowdown or fractionator overhead condenser) to adequately reduce the coke drum pressure prior to atmospheric venting. Vent gas compressors utilized in higher pressure coke drum units that route the blowdown vent gas to the Wet Gas Compressor (enclosed blowdown system) will have to reflect the new requirements to ensure that the equipment is designed to be capable of reducing the coke drum pressure to the required level.
Existing low-pressure units may need to be revamped to include an ejector system installed on the coke drums or the Blowdown Settling Drum. Steam requirements, air-cooler condensing capacity and sour water capability will also need to be reviewed. Higher pressure units with vent gas or flare gas compression will require evaluation to determine equipment capability to achieve the lower pressure and subsequent modifications if deemed necessary.
Time taken to depressure the drums will have to be considered while setting total cycle time.
Question 57: If vacuum tower bottom feed to the Coker unit drops below unit minimum, what are your operating options available?
Srini Srivatsan (Foster Wheeler USA Corporation)
(Similar question answered by Srini in 2013 as a panelist)
Increasing amount of tight oil production with hardly any residual fraction will lead to challenges in filling up the Delayed Coking Unit (DCU). In order to maintain the DCU capacity, external purchase of HFO or VR will have to be made. If purchasing external feed is not an option and if you have multiple drums, you may have to shut a module down. The DCU could also be operated in turndown mode with or without high recycle.
FCC slurry oil, if available, could be sent to the DCU as feed. Since only a small portion of the FCC slurry / decant oil converts to coke, the remaining portion goes through the coker along with the rest of the cracked VR and mostly ends up with the HCGO. If the HCGO is sent directly to the FCC without hydrotreating, this may create a recycle stream that could become difficult to handle due to build-up of refractory type material. Hence, we typically limit this amount to approximately 10% of feed and depending on the limitations of the coking unit, you may have to cut back on vacuum residue (VR) feed rate.
Question 58: What are you using for velocity medium in coker heaters? Are you using boiler feed water?
Jack Buckley (Foster Wheeler USA Corporation)
Foster Wheeler has used BFW as velocity media. Our order of preference for Velocity media is:
1.Steam
2.Condensate
3.BFW
If Condensate or BFW is used as velocity media or to clear the heater tubes during a heater trip, precautions must be taken as excess free water has the potential to overpressure the heater during a shut-in condition. The injection must also be stopped after a period of time after the heater trips because water will flow to the coke drums or back to the fractionator (coke drum by-passed) if the injection is continued too long.
Question 59: What is your experience processing a crude oil that has been treated with a pour point depressant and/or wax dispersant agent?
Dennis Haynes (NALCO Champion)
Some laboratory simulation experience has demonstrated that there were no adverse impacts to processing crude oil treated with a certain pour point depressant relative to oil-water separation for desalting. Due to these additives containing proprietary components, having differences between providers, and due to the potential of new variants being introduced to address increasing issues from Tight Oils, it is uncertain that past experience will equal future experience.